How is protein (enzyme) activity regulated?
SHORT TERM
- Change in substrate or product concentration (fast)
- Change in protein conformation (longer) :
- Proteolytic cleavage (irreversible)
- Covalent Modification (e.g phosphorylation)
- Allosteric regulation
LONG TERM
- Changing rate of protein degradation (ubiquitin-proteasome pathway)
- Changing rate of protein synthesis (enzyme induction)
What are isoenzymes?
Enzymes that catalyse the same reaction but have different kinetic properties, e.g hexokinase and glucokinase
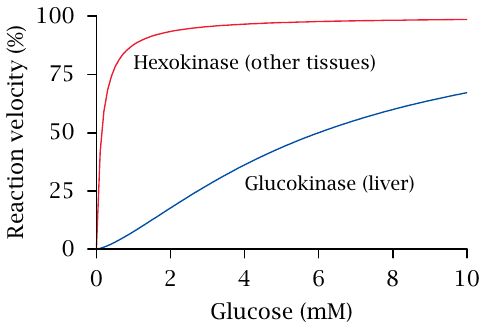
What is the difference between hexokinase and glucokinase?
- Glucokinase has a higher Km, so lower affinity, so pathway only activated when blood glucose levels are high
What is product inhibition?
Accumulation of a product leads to inhibition of the forward reaction.
e.g glucose-6-phosphate inhibits hexokinase activity
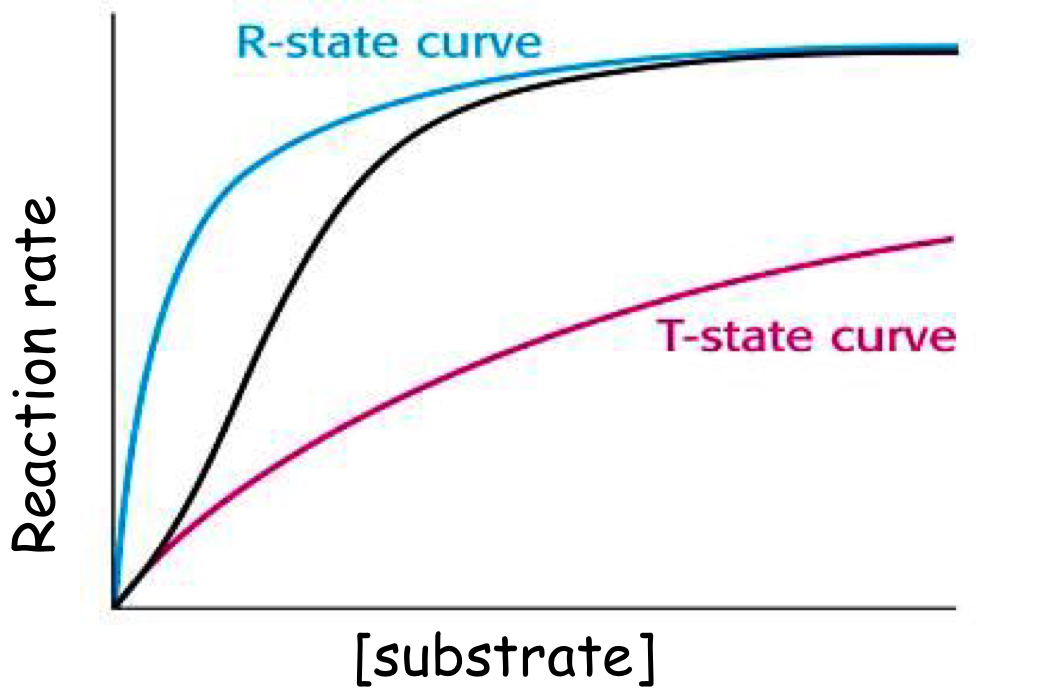
What is one of the main differences between a multisubunit (allosteric) enzyme and a single subunit enzyme?
- Allosteric show sigmoidal relationship
- Simple show rectangular parabola
Draw a graph to show the relationship between substrate concentration and reaction rate in an allosteric enzyme?
Substrate binding to one subunit causes a conformtational change, making subsequent binding easier
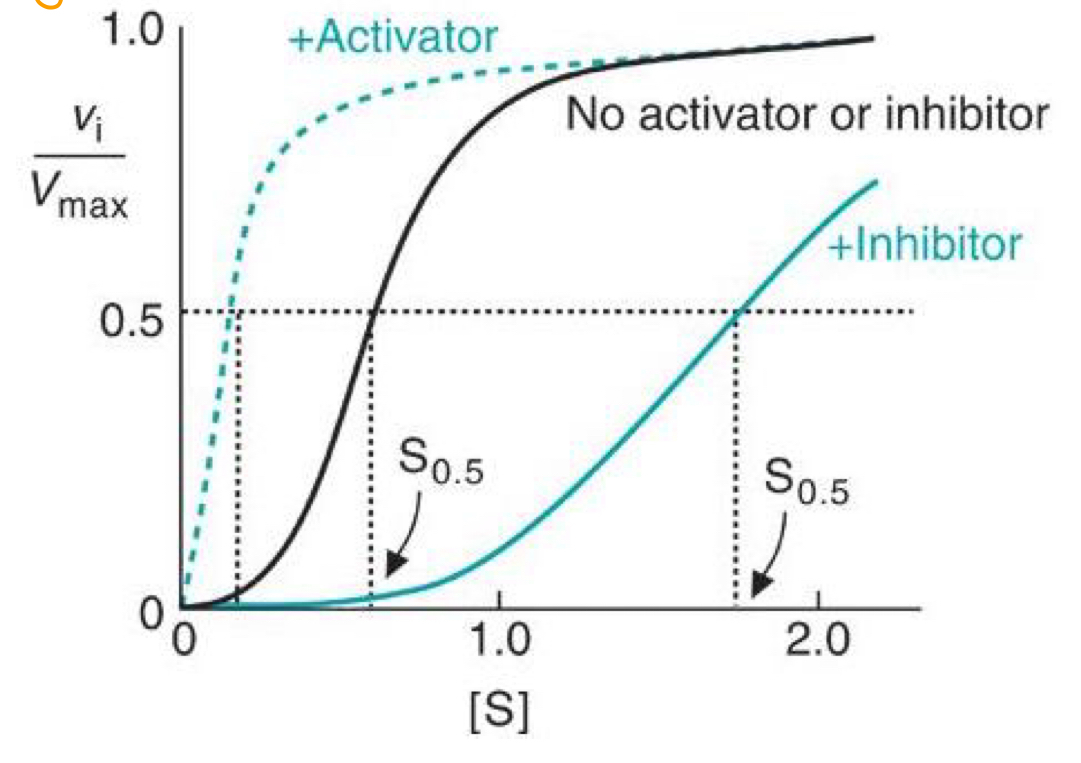
What is an allosteric activator and inhibitor?
Bind to enzyme at a site that is not the active site, which induces a conformational change
- Activator: Stabilises R state
- Inhibitor: Stabilises T state, e.g 2,3 BPG, products of pathway
Draw a graph of Vi/Vmax against [Substrate], with and without an allosteric activator.
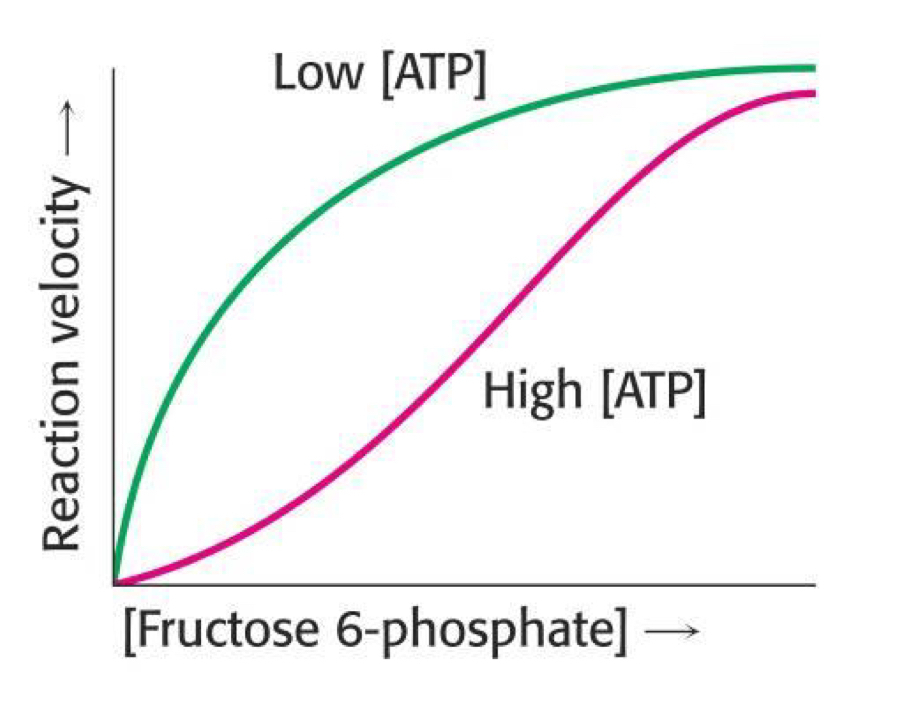
What is the role of phospho-fructokinase and what are the allosteric activators and inhibitors?
- Allosterically regulated to set the pace of glycolysis
Activators: AMP, Fructose 2,6-Bisphosphate
Inhibitors: ATP, Citrate, H+
Draw a graph of reaction velocity against [phosphofructokinase]
What are some covalent modifications that can affect enzyne activity?
- Acetylation
- Carboxylation
- Phosphorylation
- Sulfation
- Ubiquitination
What enzymes are involved in phosphorylation/dephosphorylation?
Protein Kinases - Add terminal phosphate from ATP to OH groups on Ser, Thr, Tyr
Protein Phosphatases - Hydrolysis reactions to remove phosphate
How does phosphorylating an enzyme affect its activity?
- Adds 2 negative charges
- Phosphate can form H bonds
- Conformational change, especially if near the active site
- Links energy status to metabolism, using ATP
- Allows for amplification affects, e.g PKA
What is reciprocal regulation?
e.g Glycogenolysis and Glycogenesis
When one path way is active, the other is inhibited
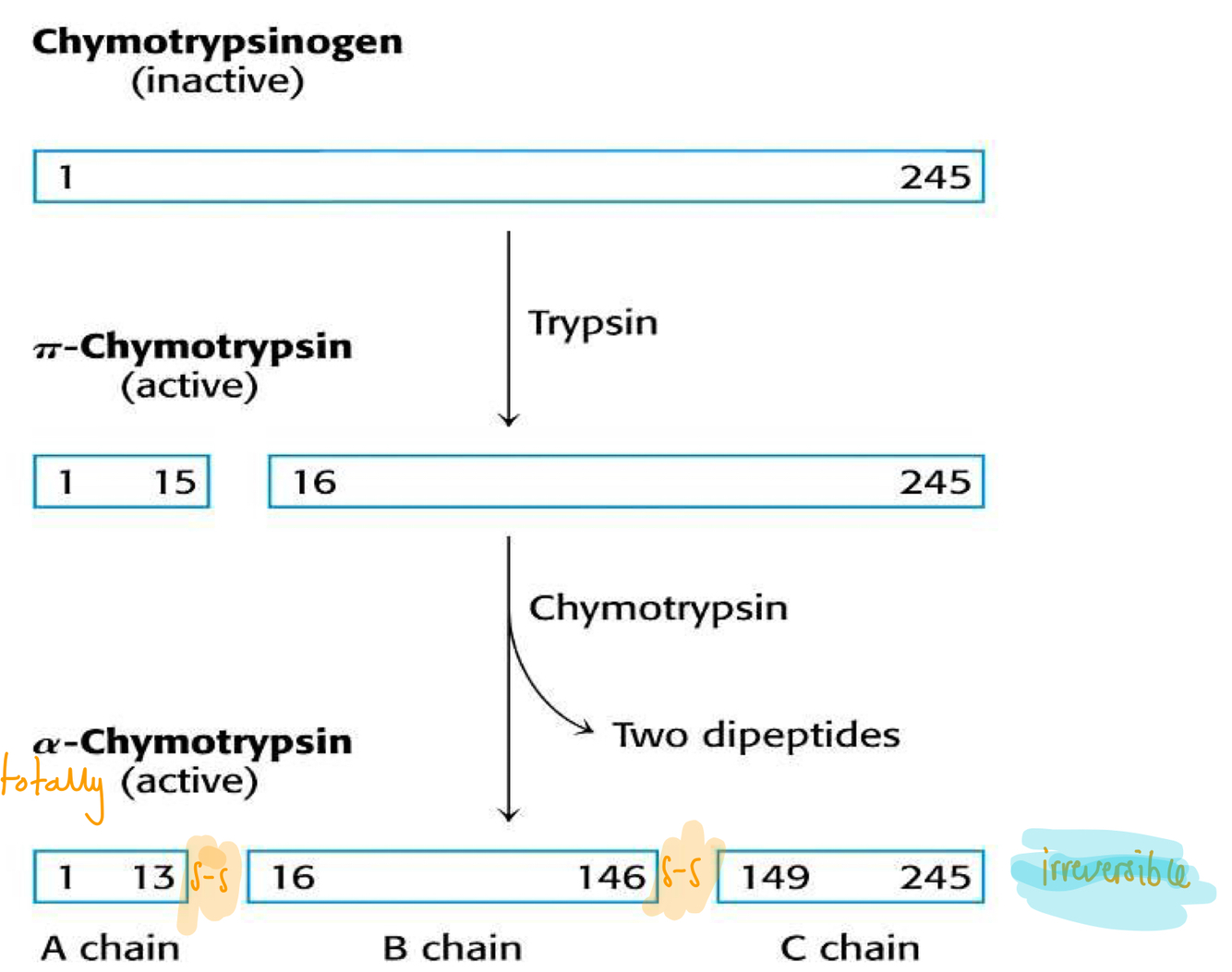
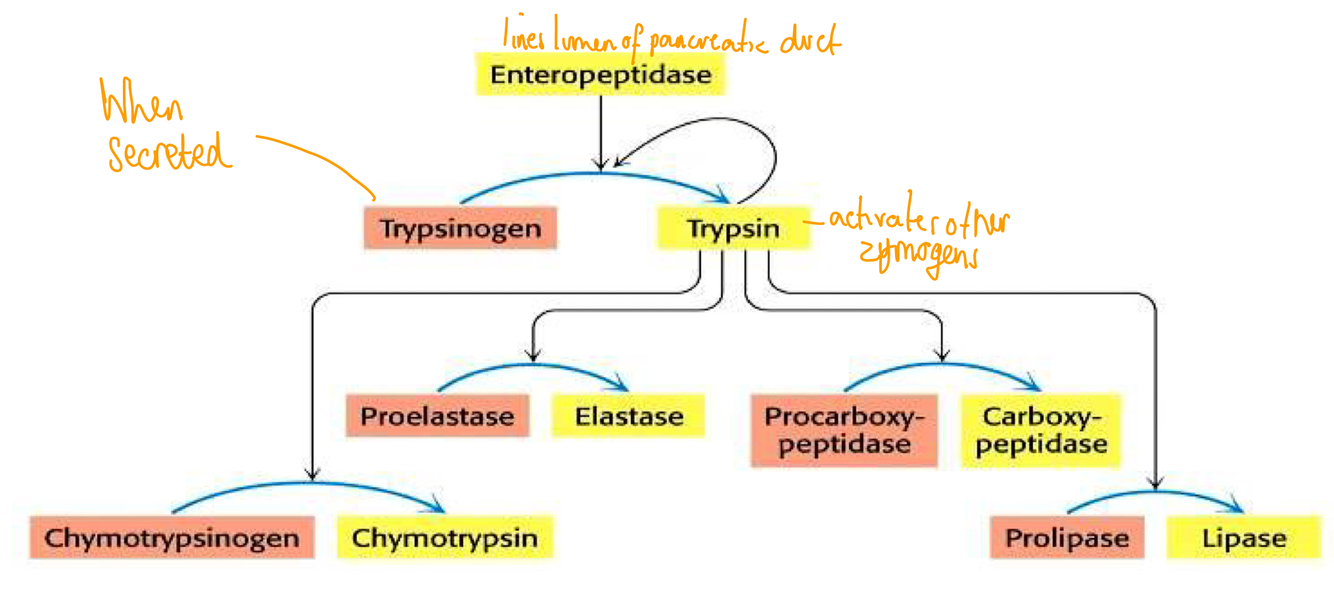
Explain how proteolytic cleavage can affect enzyme activity?
- Digestive enzymes synthesised as zymogens (inactive as active site blocked)
- Cleavage
- Fully active protein
Give some examples of zymogens/
Apoptosis mediated by captases, that were initially procaptases

What is the proteolytic cleavage of chymotrypsinogen?
How is proteolytic cleavage activated?
Trypsin is a protease and cleaves zymogens
If proteolytic cleavage is irreversible, how is the enzyme switched off?
- Degradation
- Irreversible binding of inhibitors to active site, especially active site of trypsin
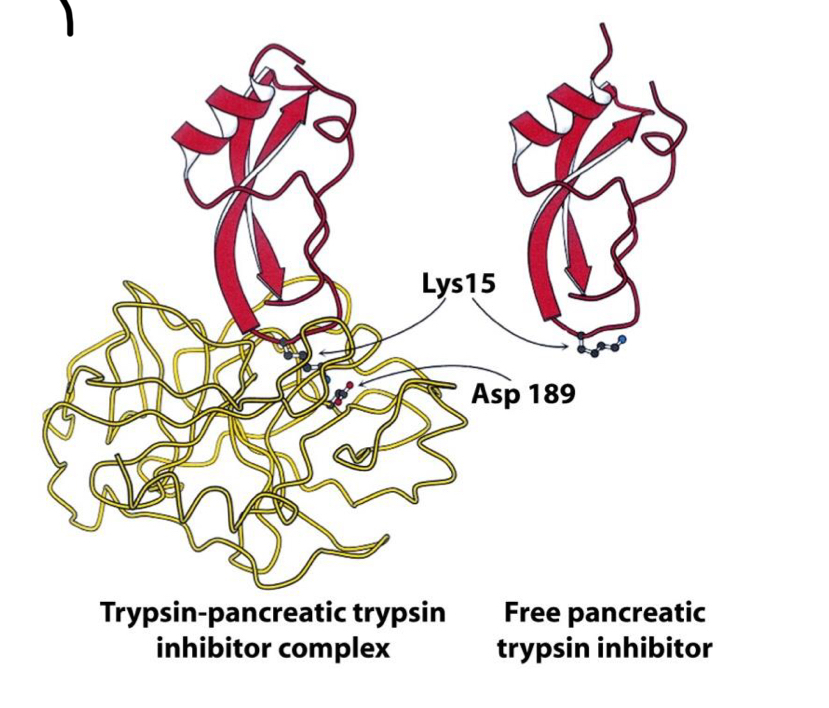
How can trypsin inhibitors lead to disease?
- Deficiency of a1antitrypsin
- Can’t switch off trypsin and therefore other proteases
- Elastase destroys alveolar walls
Why are cascades important?
Allow amplification of regulatory signals
What are the key features of regulation?
- Can be irreversible or reversible
- Regulation can occur on a range of time scales, seconds to days
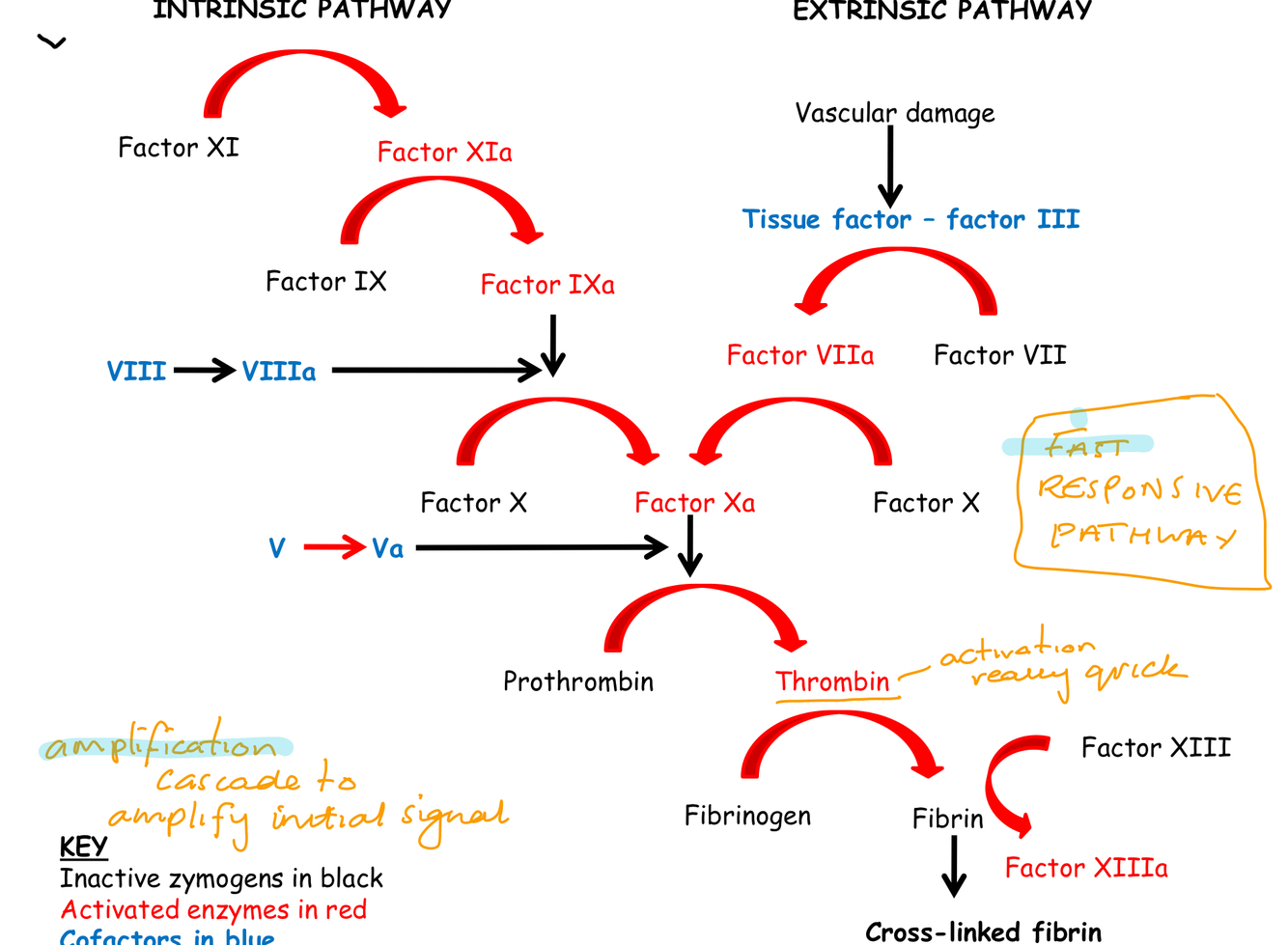
Summarise the blood clotting cascade and why the cascade is important?
Cascade important as normally little factors in the blood
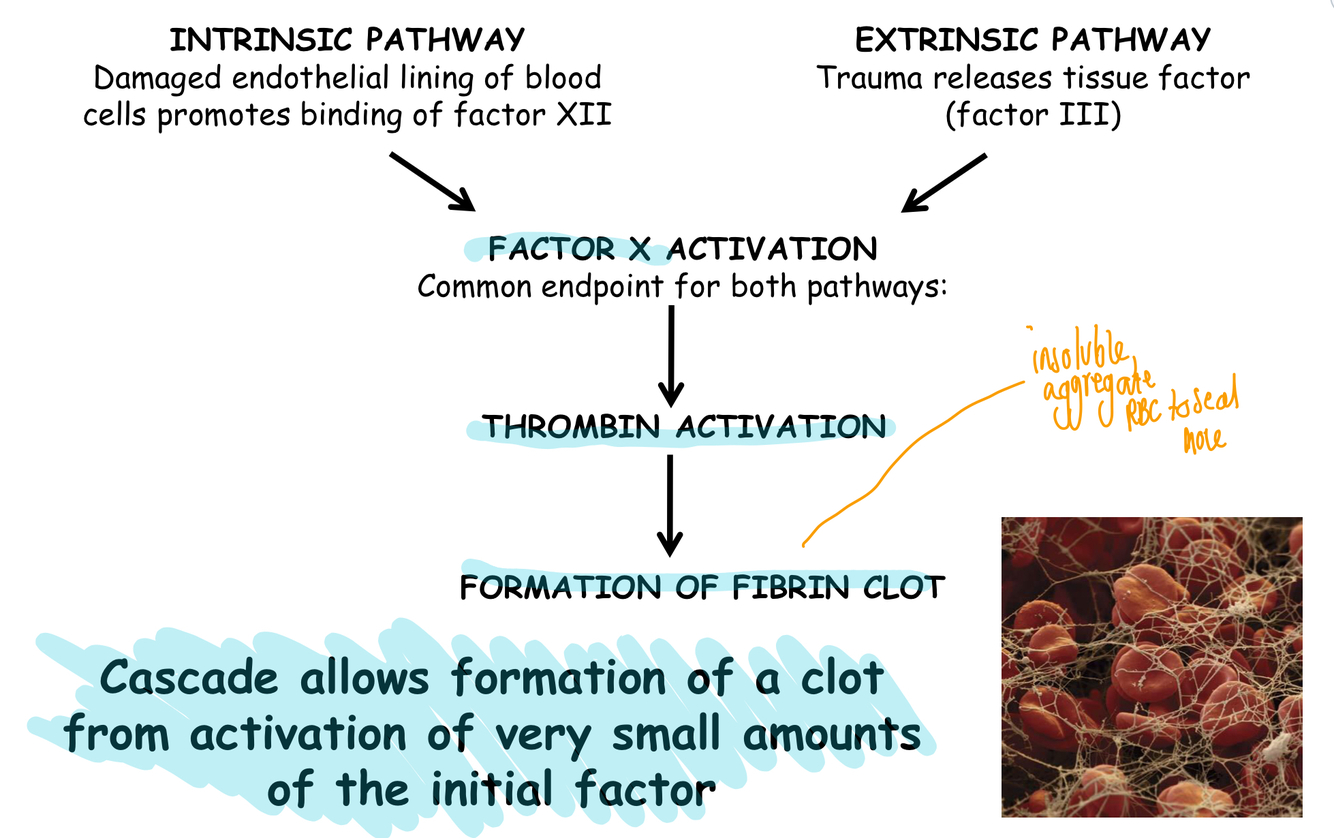
What are the key features of the clotting cascade?
- Fast responsive pathway
- Initial signal amplified by cascade
- Thrombin produce quickly which activates fibrinogen cleavage