Define ionic bonding
Electrostatic force of attraction between oppositely charged ions formed by electron transfer
Ionic Bonding
Metal atoms ___ electrons to form ___ ions
Metal atoms lose electrons to form +ve ions
Ionic Bonding
Non-metal atoms ____ electrons to form ___ ions
Non-metal atoms gain electrons to form -ve ions
Simplest ions are…
single atoms which have lost/gained electrons to make full outer shell
What are compound ions?
Ions that are made up of groups of atoms with an overall charge
State the formula for a sulfate ion
SO42-
State the formula for a hydroxide ion
OH-
State the formula for a nitrate ion
NO3-
State the formula for a carbonate ion
CO32-
State the formula for an ammonium ion
NH4+
Name the structure of ionic crystals
Giant Ionic Lattice
Sodium nitrate contains Na+ (1+) and NO3- (1-) ions. State the fomula of the sodium nitrate.

Magnesium chloride contains Mg2+ (2+) and Cl- (1-) ions. State the fomula of the magnesium chloride.
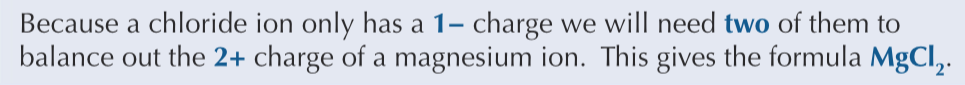
Name 3 physical properties of ionic compounds
- Conduct electricity only when they’re molten or dissolved
- High melting points
- Tend to dissolve in water
Why can ions conduct electricity when they’re molten or dissolved?
∵ ions in liquid are free to move and carry a charge
Why can’t ions conduct electricity when they’re in a solid?
∵ ions are in fixed position by strong ionic bonds
Why do ionic compounds have high melting points?
- Giant ionic lattices
- Strong electrostatic forces of attraction between oppositely charged ions
- Takes a lot of energy to overcome these forces
Why do ionic compounds tend to dissolve in water?
- Water molecules are polar
- Part of molecule has a small negative charge and other bits have small positive charges
- Charged parts pull ions away from lattice = causing it dissolve
Ionic bonding is stronger and melting points are higher when ions are… (2x)
smaller and/ or have higher charges
When do molecules form and how are they held together?
- Form when 2 or more atoms bond together
- Held together by strong covalent bonds
What do single covalent bonds contain?
Shared pair of electrons
Describe covalent bonding
- Two atoms share electrons so they’ve both got full outer shells
- Both postive nuclei are attracted electrostatically to shared electrons

Multiple covalent bonds contain…
multiple shared pairs of electrons
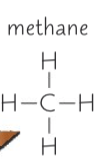
Draw methane, represent the covalent bonds by drawing lines