What is the mobile phase?
Where molecules can move
What is the mobile phase always?
Always liquid or gas
What is the stationary phase?
Where molecules can’t move
What is the stationary phase always?
Solid, or a liquid on solid support
What does the distance that each substance moves up the plate by depend on?
Substance’s solubility in mobile phase and its retention by stationary phase
State 3 types of chromatography
- Thin-Layer Chromatography
- Column Chromatography
- Gas Chromatography
State what the stationary phase in thin-layer chromatography is
thin layer of silica (silicon dioxide) or alumina (aluminium oxide) fixed to glass or metal plate
When handling the plate in TLC, what should you do and why?
Wear gloves to avoid contamination by substances on your hand
Describe a method for thin-layer chromatography (TLC)
- Draw line in pencil near bottom of TLC plate (the baseline) & put small drop of each mixture on line
- Allow spots to dry
- Place plate in beaker with small volume of solvent (mobile phase)
- Solvent level must below baseline
- As solvent moves up plate, it carrier substances in mixture with it
- Some chemicals will be carried faster than other and and travel further up plate
- Remove plate from beaker when solvent has nearly reached top of plate
- Before it evaporates, use a pencil to make how far solvent travelled up plate (solvent front)
- Place plate in fume cupboard to dry
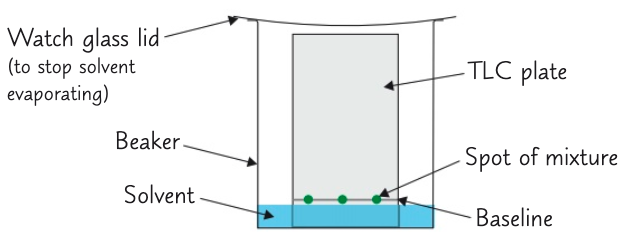
Why must the solvent level be below the baseline?
So solvent doesn’t dissolve samples away
Why should you place a TLC plate in fume cupboard to dry?
Fume cupboard will prevent any toxic or flammable fumes from escaping into room
What is the result produced from TLC called?

Chromatograph
Name 2 substances you can use to reveal colourless chemicals
- UV Light
- Iodine
Describe how you can use UV light to reveal colourless chemicals
- Many TLC plates have special fluorescent dye added to silica or alumina layer that glows when UV light shines on it
- Can put plate under UV lamp and draw around dark patches to show where spots of chemical are
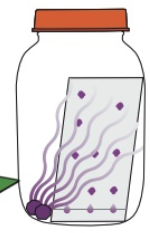
Describe how you can use iodine to reveal colourless chemicals
- Expose chromatogram to iodine vapour
- i.e. leave plate in sealed jar with some iodine crystals
- Iodine vapour is a location agent - sticks to chemicals on plate and they’ll show up as brown/purple spots
How can you find the number of chemicals present in a TLC chromatograph?
Count no. of spots formed on plate
How can you find out what each chemical is from TLC?
By calculating a Rf value
State the formula to calculate a Rf value
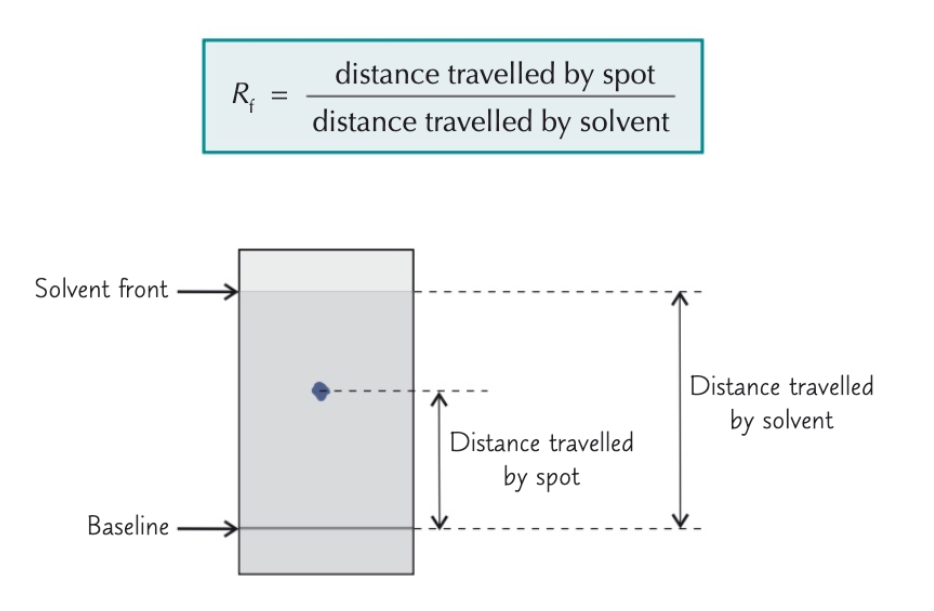
Once you’ve calculated a Rf value, what do you do next?
Look up Rf values in table of standard Rf values to identify what the substance is
Name 3 factors that can change Rf values
If composition of TLC plate, solvent or temperature changes even slightly = get different Rf values
As it’s hard to keep conditions ideal in TLC, what can you do to combat the differences in Rf values?
∴ if you suspect mixture contains e.g. chlorophyll best to put spot of chlorophyll on baseline of same plate as mixture and run them both at same time
What is column chromatography used for?
Used for purifying an organic product
(Done to separate product from unreacted chemicals and by-products)
Describe column chromatography (method)
- Involves packing glass columns with slurry of absorbent material e.g. aluminium oxide, coated with water
- Mixture to be separated is added to top of column and allowed to drain down into slurry
- Solvent is then run slowly and continually through column
What is the stationary phase in column chromatography?
Water-coated particles of Al2O3







