Describe how you would test for alcohols
Adding sodium metal to pure alcohol = fizzing as it gives off H2 gas
Describe how you would test for primary, secondary, tertiary alcohols
- Add 10 drops of alcohol to 2 cm3 of acidified potassium dichromate solution in test tube
- Warm mixture gently in hot water bath
- Watch for colour change
- Primary: Orange solution turns green as aldehyde forms
- Secondary: Orange solution turns green as ketone forms
- Tertiary: Nothing happens
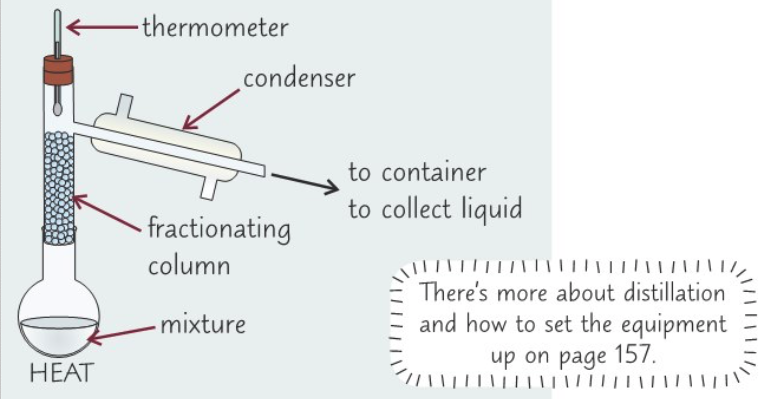
Describe how you could test for primary alcohol (differentiate it from a secondary alcohol)
- Add excess alcohol to 2 cm3 of acidified potassium dichromate solution in round bottomed flask
- Set up flask as part of distillation apparatus
- Gently heat flask
- Alcohol will be oxidised & product will be distilled off immediately = so you can collect it (if product produced aldehyde, then solution was a primary alcohol)
Name 3 reagents you can use to test for an aldehyde or ketone
- Fehling’s solution
- Benedict’s solution
- Tollens’ reagent
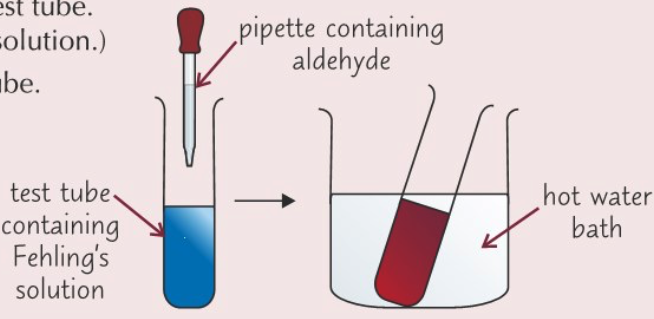
Describe a test for an aldehyde or ketone using Fehling’s (or Benedict’s) solution
- Add 2 cm3 of Fehling’s (or Benedict’s) solution to test tube
- Add 5 drops of aldehyde or ketone to test tube
- Put test tube in hot water bath to warm it for few minutes
- Aldehyde: Blue solution will give brick red precipitate
- Ketone: Nothing
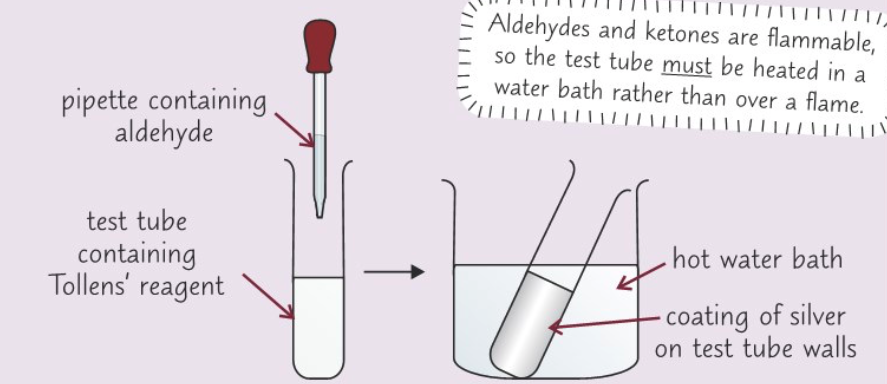
Describe a test for an aldehyde or ketone using Tollens’ reagent
- Put 2 cm3 of 0.1 mol dm-3 silver nitrate solution in test tube
- Add few drops of dilute sodium hydroxide to solution
- Light brown precipitate should form
- Add drops of dilute ammonia solution until brown precipitate dissolves completely
- Place test tube in a hot water bath and add 10 drops of aldehyde or ketone
- Aldehyde: silver mirror forms on walls of test tubes
- Ketone: Nothing
Why is a silver mirror produced when Tollens’ reagent is added to an aldehyde?
Aldehyde reduced Ag+ ions to silver atoms
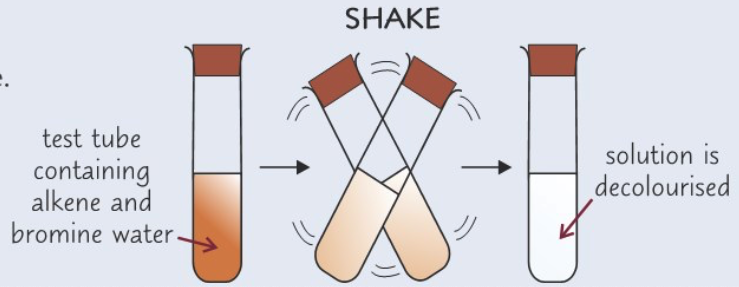
Describe the test for alkenes
- Add 2 cm3 of solution you want to test to test tube
- Add 2 cm3 of bromine water to test tube
- Shake test tube
- Alkene: Solution will go from orange to colourless
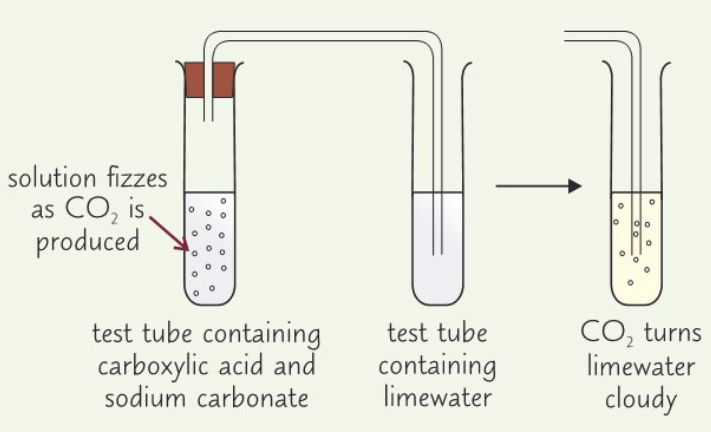
Describe the test for carboxylic acids
- Add 2 cm3 of solution you want to test to test tube
- Add 1 small spatula of solid sodium carbonate
- If solution begins to fizz, bubble gas that it produces through some limewater in 2nd test tube
- Carboxylic Acid: Solution will fizz & CO2 gas produced will turn limewater cloudy
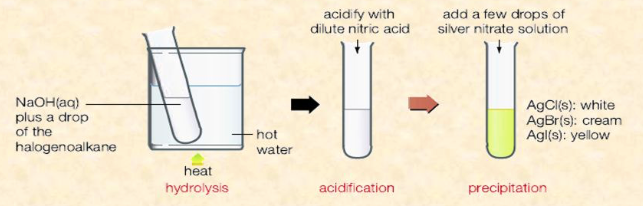
Describe the test for halogenoalkanes
- Add halogenoalkane (R—X) and sodium hydroxide to test tube
- Warm it by placing it in hot water bath
- Add dilute nitric acid and silver nitrate solution to test tube to acidify its contents
- Halogenoalkane: Precipitate of AgX
What is mass spectrometry used for?
To find Mr of compound
Molecular ion (M+) is formed when…
a molecule loses an electron
M+ produces ____ _____ ____ on mass spectrum of compound
molecular ion peak
Mass/charge (m/z) value of molecular ion peak = …
molecular mass of compound
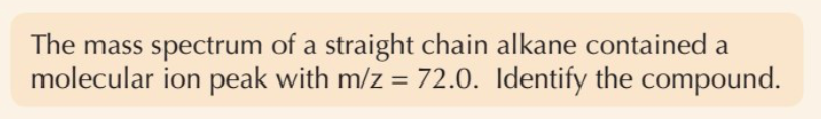

What does high resolution mass spectrometry measure?
Measure atomic and molecular masses extremely accurately
What is high resolution mass spectrometry used for?
Identifying compounds that have same Mr when rounded to nearest whole number

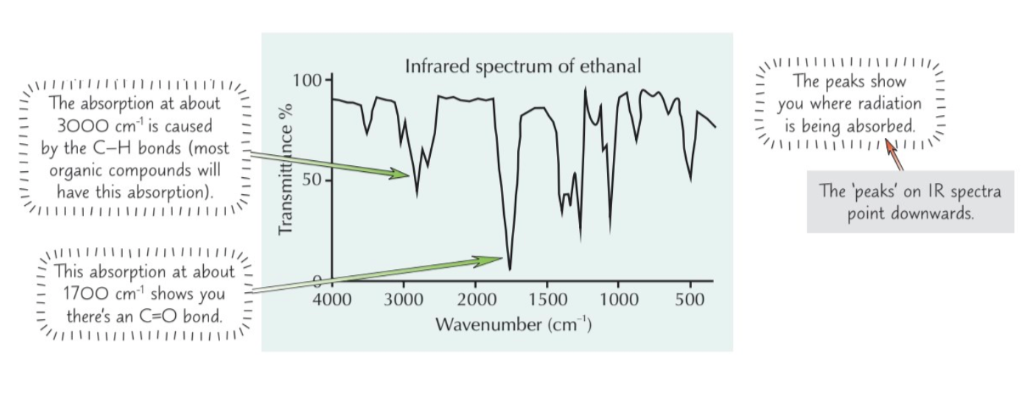
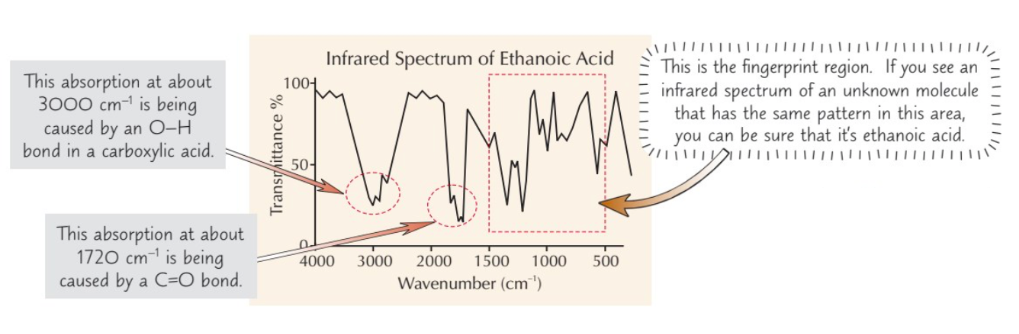
Describe how infrared spectroscopy works
- Beam of IR radiation passed through sample of chemical
- IR radiation absorbed by covalent bonds in molecules = ↑ vibrational energy
- Bonds between different atoms absorb different frequencies of IR radiation
- Bonds in different places in molecules absorb different frequencies too (e.g. O-H group in alcohol and O-H in carboxylic acid absorb different frequencies)
What does an infrared spectrometer produce & what does this show?
Infrared spectrometer produces graph that shows what frequencies of radiation molecules are absorbing
What can a graph produced by an infrared spectrometer be used for?
To identify functional groups in molecule
The fingerprint region is between ______ and ______
500 cm-1 and 1500cm-1
Fingerprint region is _____ to a particular compound
Unique
Describe how you can use the fingerprint region to identify a molecule
- Can use computer database to check this region of unknown compound’s IR spectrum against known compounds
- If it matches one of them = know what molecule it is
Infrared spectroscopy can also be used to find out….
how pure compound is & identify any impurities





