dynamic equilibrium
forward and reverse reactions occur at the same rate… no net change in concentrations of products or reactants
entropy
measure of the distribution of energy throughout a system or between a system and its environment
at equilibrium…
reaction will reach equilibrium when the system’s entropy is at a maximum or ΔG is at a minimum
law of mass action
if a system is at equilibrium at a constant temperature, then following ratio is constant
Kf/Kr = Kc = Keq
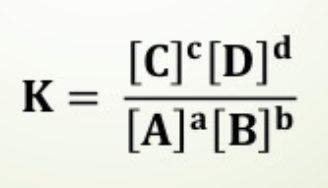
equilibrium expression
for aA + bB ⇔ cC + dD,
Kc= [C]c[D]d / [A]a[B]b
*exponents equal stoichiometric coefficients UNLIKE in rate laws
reaction quotient, Q
“timer” to indicate how far the reaction has proceeded toward equilibrium… utility of Q is comparison that can be made between Q at any moment and known Keq for reaction at a particular temperature
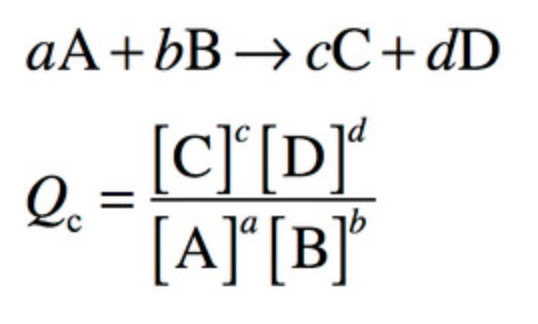
Q and Keq
- Q < Keq: ΔG -, forward reaction has not yet reached equilibrium
- forward rate of reaction is increased
- Q > Keq: ΔG +, forward reation has exceeded equilibrium
- rate of reverse reaction increased to restore equilibrium
- Q = Keq: ΔG=0, reaction is at dynamic equilibrium
things to remember about equlibrium/law of mass action
- concentration of pure solids and pure liquids do not appear in the equilibrium constant expression
- Keq is temperature dependent
- larger Keq leads to farther right equilibrium position
- if Keq is for forward reaction, 1/Keq is for reverse reaction
equilibrium equations
- Keq=[products]/[reactants]
- Keq with a large positive exponent indicates a reaction that goes almost to completion
- Keq with a large negative exponent indicates that the amount of reactant that has reacted is neglibible compared to the amount of reactant that remains *use this instead of ICE
le chatelier’s principle
if a stress if applied to a system, the system shifts to relieve that applied stress
- stress can be change in concentration, pressure, volume, temperature, etc.
change in concentration
changing concentration of either product or reactant results in Qc ≠ Keq
changes in pressure (and volume)
only chemical reactions w/ at least one gaseous species will be affected by pressure and volume
- when a system is compressed, increase in total pressure is correlated with increase in partial pressures of each gas in the system
- when pressure is increased, system moves toward whichever side has the lower total number of moles of gas… due to ideal gas law, which states that there is direct relationship between number of moles of ags and pressure of gas
changes in temperature
changing temperature changes Keq, not Qc (like pressure and concentration)
- direction system moves to restore equilibrium is determined by enthaply of reaction…
- if reaction is endothermic (∆H>0), heat functions as reactant
- if reaction is exothermic (∆<0), heat functions as a product
kinetic product
forms at lower tempertaures (with smaller heat transfer)
- less free energy needed for kinetic product to form
- kinetic product forms faster and is less stable
thermodynamic product
forms at higher temperatures (with larger heat transfer)
- greater stability and more negative ∆G
- forms slower and higher stability


