1
Q
is a single, double, triple bond considered a single electron domain?
A
yes
2
Q
single bond length and strength
A
longest weakest
3
Q
double bond length and strength
A
shorter stronger
4
Q
triple bond length and strength
A
shortest strongest
5
Q
When do covalent bonds occur?
A
when atomic orbitals overlap
6
Q
sigma-bond
A
head on/ direct overlap
7
Q
where is electron density concentrated in a sigma-bond?
A
between atoms
8
Q
examples of sigma overlap
A
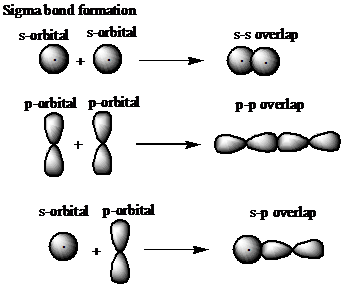
9
Q
pi-bond
A
sideways overlap
10
Q
where is electron density concentrated in a pi-bond?
A
above and below atoms
11
Q
examples of pi overlap
A
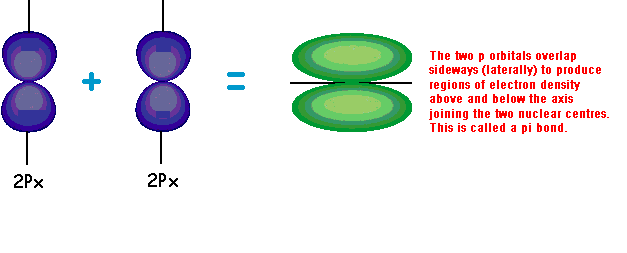
12
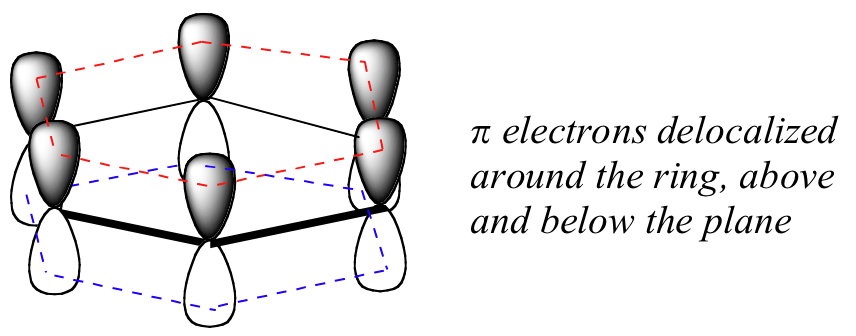
Q
benzene bond structure
A
- 6 C-H sigma bonds
- 6 C-C sigma bonds
- remaining p-orbitals overlap sideways
- 6 electrons delocalize throughout pi-system
- VERY STABLE