What is a buffer solution?
- Resist changes in pH upon addition of small amounts of strong acid or strong base
- Resistive action is a result of the equilibrium which is set up between the weak acid and the salt
What do buffer solutions consist of?
- Weak acid and it’s salt(conjugate base) or a weak base and it’s salt(conjugate acid)
- Can also be composed of mixtures of salts of polyprotic acids
What is essential for both conjugate species in a buffer?
•Have to be comparable(or same) concentrations
What is the equation for a buffer solution?
.

In a buffer solution, what does HA represent?
•Weak acid
In a buffer solution, what does H+ represent?
•Hydrogen ion component of the dissolved salt
In a buffer solution, what does A- represent?
•The anion component of the salt
Write the dissociation constant for a weak acid
.
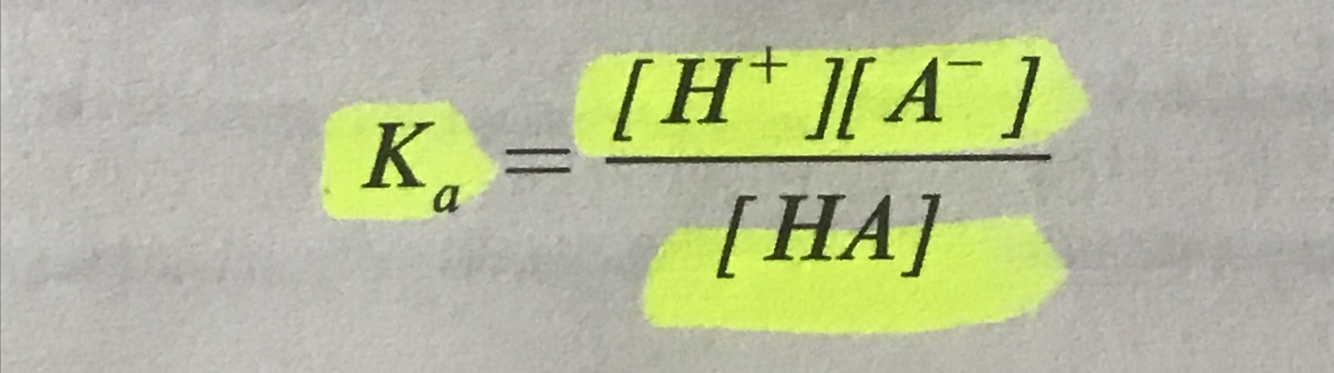
What occurs when an alkali is added to the standard acid buffer solution?
•The hydronium ions mop it up- the ions are regenerated as the equilibrium moves to the right and some of the acid is broken down into hydronium ions and anions
What occurs when an acid is added to the standard acid buffer solution?
•The anions combine with the substance and pH is once again restored
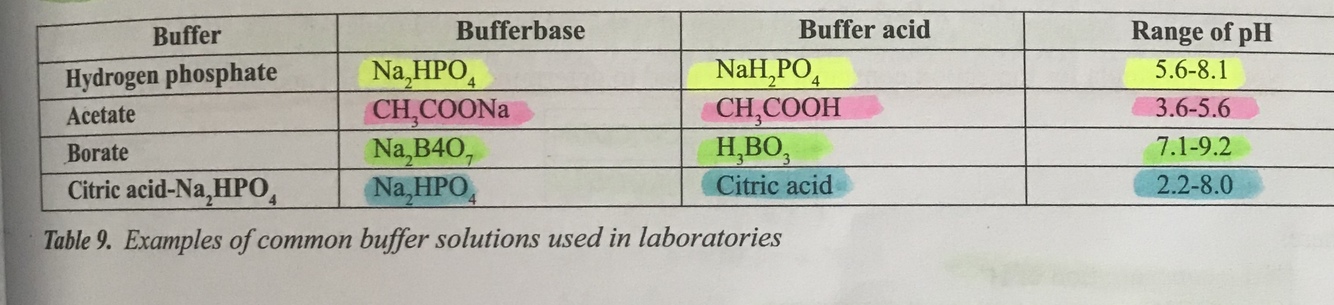
What are the common buffer solutions used in laboratories?
.
Why are buffers important for enzymes?
- Keep the right pH for enzymes to function-enzymes only work under very precise conditions
- If pH strays too far from margin, enzymes slow or stop working and organism dies
What are the applications for buffer solutions industrially?
- Useful In fermentation processes
- Setting the correct conditions for the dyes used in colouring fabrics
Illustrate a buffer solution of acetic acid and sodium acetate in action
.

Illustrate the Henderson hasslebach equation for an acid buffer
.

Illustrate the Henderson hasslebach equation for an basic buffer
.

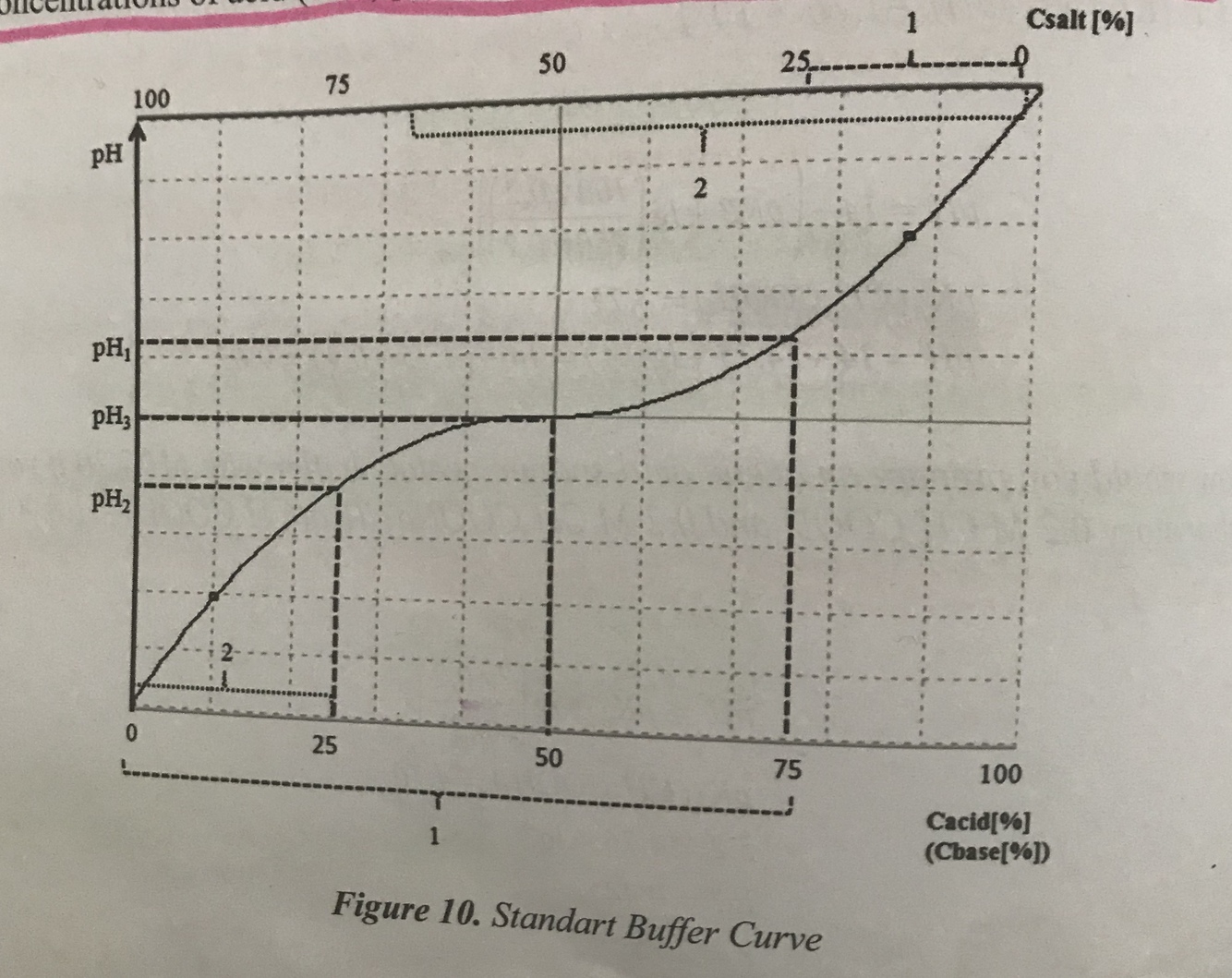
Describe the buffer curve
- Vertical axis shows the pH of the buffered solution
- Horizontal axis shows the composition of the buffer(on the left hand side of the plot, most of the buffer is in the form of salt and on the right side, most of the buffer is in the form of acid-or base)
- As acid is added-pH decreases and the buffer shifts towards greater acid concentration
- As base is added, pH increases and buffer shifts toward greater salt concentration
- Slope of curve flattest where pH3 is equal to pK value for the buffer-buffering capacity greatest here because a shift in relative concentrations of acid(or base) produces only a small change in pH of the solution
- At pH values higher than pH1- slope of curve much higher and a shift in the relative concentrations of acid(base) produces a large change in pH of the solution
Draw the buffer curve
.
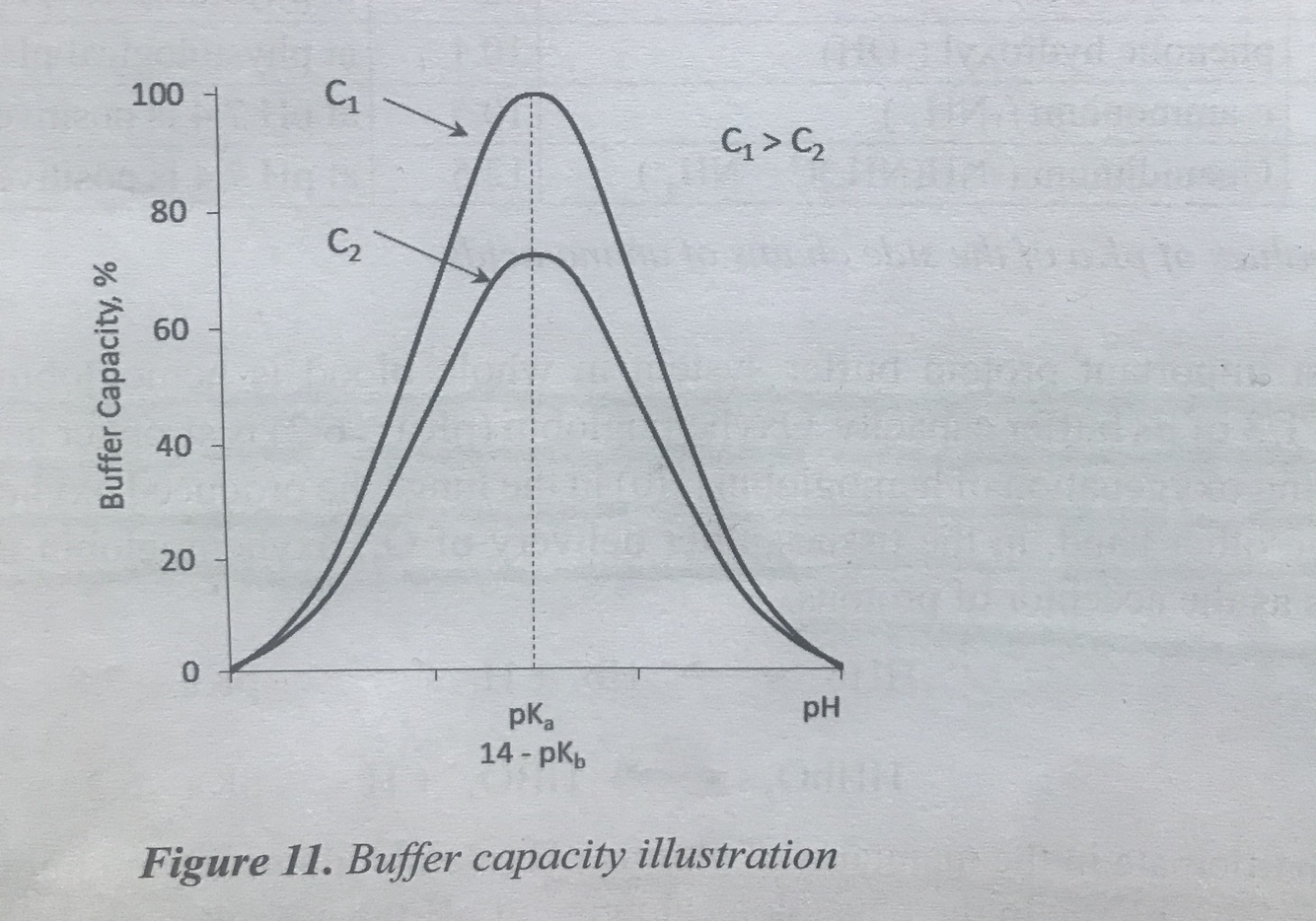
Describe buffer capacity?
- The ability of a buffer to suppress changes in pH
- The amount of an acid or base that can be added to a volume of a buffer solution before pH changes significantly
- Buffering capacity increases as the molar concentration(molarity) of the buffer salt/acid solution increases
- The closer the buffered pH is to the pKa, the greater the buffering capacity
- When buffer is diluted- it’s capacity decreases but pH does not change
What factors determine the buffering capacity of a solution?
- The concentration of the buffer (the higher the concentration. The larger the buffering capacity)
- How close the pKa of the buffer is compared to the pH of the solution(the closer it is, the greater the buffer capacity
Illustrate the buffer capacity
.
What are the most important buffer systems within an organism?
- Hydrogen carbonate
- Proteins
- Hydrogen phosphate
Describe hydrogen carbonate as a buffer
- Main buffer of extracellular fluids- makes more than half the buffer capacity of blood
- Consists of HCO3- and H2CO3
What causes the buffer effect of proteins?
•Caused by their amphoteric character




