How are Metal Oxides formed?
WHat are redox reactions(Oxidation and Reduction)?
- Metals react with oxygen in the air to produce metal oxides
- Oxidation and reduction involve the addition or removal of oxygen from a substance and are called redox reactions
- Oxidation is a reaction in which:
- Oxygen is added to an element or a compound
- Reduction is a reaction in which:
- Oxygen is removed from an element or a compound
- A common example is the reaction with red-brown copper metal to produce black copper oxide:
2Cu + O2 ⟶ 2CuO
- In this reaction copper metal has been oxidised since oxygen has been added to it
- Another example is the reaction of zinc oxide with carbon:
ZnO + C ⟶ Zn + CO
- In this reaction the zinc oxide has been reduced since it has lost oxygen. The carbon atom has been oxidised since it has gained oxygen
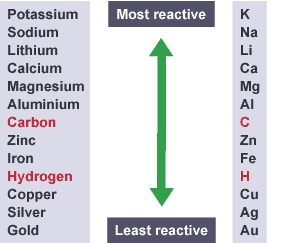
What is The reactivity series?
- The tendency of a metal to lose electrons is a measure of how reactive the metal is
- A metal that is high up on the series loses electrons easily and is thus more reactive than one which is lower down on the series
- Note that although carbon and hydrogen are nonmetals, they are included in the series as they are useful in extracting metals from their oxides by reduction processes
What happens when Metals react with Water?
metal + water → metal hydroxide + hydrogen
- Some metals react with water
- Metals above hydrogen in the reactivity series will react with water, but the reaction may be very slow
- Metals that react with cold water form a metal hydroxide and hydrogen gas:
For example calcium:
Ca + 2H2O → Ca(OH)2 + H2
calcium + water → calcium hydroxide + hydrogen
Name the Reactivity series
- There are several reactivity series mnemonics to help you remember the order of the metals
- One that we like goes as follows: “
Please
send
lions,
cats,
monkeys
and
cute
zebras
into
hot
countries
signed
Gordon”
How do Metals react with Acids?
- Only the metals below hydrogen in the reactivity series will not react with acids
- When acids and metals react, the hydrogen atom in the acid is replaced by the metal atom to produce a salt and hydrogen gas:
metal + acid → metal salt + hydrogen
- For example iron:
Fe + 2HCI → FeCl2 + H2
iron + hydrochloric acid → iron(II)chloride + hydrogen
- In both these types of reactions (water and acids) the metals are becoming positive ions
- The reactivity of the metals is related to their tendency to become an ion
- The more reactive the metal the more easily it becomes an ion (by losing electrons)
Why do Non-metals appear in the Reactivity Series?
- Why do non-metals appear in the reactivity series of metals?
- A reactivity series will usually contain the elements carbon and hydrogen
- This is beause these elements play different roles in our understanding the reactions of metals and our ability to predict how metals can be extracted from their ores
- From the reactions with water and acids we have seen that whether a reaction takes place depends on the position of the metal in the reactivity series relative to hydrogen
- A reaction takes place if the metal is able to displace hydrogen from water or acids
- Carbon is a cheap reducing agent which can be used to remove oxygen from metal oxide ores
- Placing carbon in the reactivity series allows us to see whether a metal oxide can be reduced or not by carbon
- Metals below carbon can be extracted by heating the oxide with carbon
- Metals higher than carbon have to be extracted by other methods, such as electrolysis
What is a Displacement reaction?
- The reactivity of metals decreases going down the reactivity series.
- This means that a more reactive metal will displace a less reactive metal from its compounds
- Two examples are:
- Reacting a metal with a metal oxide (by heating)
- Reacting a metal with an aqueous solution of a metal compound
- For example it is possible to reduce copper(II) oxide by heating it with zinc.
- The reducing agent in the reaction is zinc:
Zn + CuO → ZnO + Cu
zinc + copper oxide → zinc oxide + copper

What happens in displacement reactions between metals & Aqueous soloutions?
- This is easily seen as the more reactive metal slowly disappears from the solution, displacing the less reactive metal
- For example, magnesium is a reactive metal and can displace copper from a copper sulfate solution:
Mg + CuSO4→ MgSO4 + Cu
- The blue color of the CuSO4 solution fades as colorless magnesium sulfate solution is formed.
- Copper coats the surface of the magnesium and also forms solid metal which falls to the bottom of the beaker

What are Redox reactions?
balanced equation for the reaction between magnesium and copper sulfate solution can be written in terms of the ions involved:
Mg(s) + Cu2+(aq) + SO42-(aq) → Mg2+(aq) + SO42-(aq) + Cu(s)
Sulfate ions, SO42-, appear on both sides of the equation, but they do not take part in the reaction. The equation can be rewritten without them:
Mg(s) + Cu2+(aq) → Mg2+(aq) + Cu(s)
This equation is an example of a balanced ionic equation. It can be split into two half equations :
Mg(s) → Mg2+(aq) + 2e- (oxidation)
Cu2+(aq) + 2e- → Cu(s) (reduction)
Notice that:
- magnesium atoms lose electrons - they are oxidised
- copper ions gain electrons - they are reduced
Reduction and oxidation happen at the same time, so the reactions are called redox reactions.
Oxidation is the ______ of Electrons, Reduction is _______ of electrons
Oxidation is the loss of electrons, and reduction is the gain of electrons.
It helps to remember OIL RIG - oxidation is loss of electrons, and reduction is gain of electrons.
Displacement reactions are just one example of redox reactions. Electrolysis reactions are also redox reactions.
How do we extract Metals?
- The Earth’s crust contains metals and metal compounds such as gold, copper, iron oxide and aluminium oxide
- Useful metals are often chemically combined with other substances forming ores
- A metal ore is a rock that contains enough of the metal to make it worthwhile extracting
- They have to be extracted from their ores through processes such as electrolysis, using a blast furnace or by reacting with more reactive material
- In many cases the ore is an oxide of the metal, therefore the extraction of these metals is a reduction process since oxygen is being removed
- Common examples of oxide ores are iron and aluminium ores which are called haematite and bauxite respectively
- Unreactive metals do not have to be extracted chemically as they are often found as the uncombined element
- This occurs as they do not easily react with other substances due to their chemical stability
- Examples include gold and platinum which can both be mined directly from the Earth’s crust
How are Elements more reactive then Carbon extracted?
How are elements less reactive than Carbon but more reactive than silver Extracted?
How are Elements less copper extracted?
- The most reactive metals are at the top of the series
- The tendency to become oxidised is thus linked to how reactive a metal is and therefore its position on the reactivity series
- Metals higher up are therefore less resistant to oxidation than the metals placed lower down which are more resistant to oxidation
- The position of the metal on the reactivity series determines the method of extraction
- Higher placed metals (above carbon) have to be extracted using electrolysis as they are too reactive and cannot be reduced by carbon
- Lower placed metals can be extracted by heating with carbon which reduces them

How is Iron Extracted?
Extracting iron
Iron(III) oxide is reduced to molten iron when it reacts with carbon. One of the products is carbon monoxide:
iron(III) oxide + carbon → iron + carbon monoxide
Fe2O3(s) + 3C(s) → 2Fe(l) + 3CO(g)
This method of extraction works because carbon is more reactive than iron, so it can displace iron from iron compounds. Extracting a metal by heating with carbon is cheaper than using electrolysis.
How is Aluminium Extracted?
Extracting aluminium
Aluminium is more reactive than carbon so it must be extracted from its compounds using electrolysis. Even though aluminium is more abundant than iron in the Earth’s crust, aluminium is more expensive than iron. This is mainly because of the large amounts of electrical energy used in the extraction process.
Electrolysis of aluminium oxideThe electrolyte
Aluminium ore is treated to produce pure aluminium oxide. The electrolytes used in electrolysis are ionic compounds:
- in the molten state, or
- dissolved in water
Aluminium oxide is insoluble in water, so it must be molten to act as an electrolyte. However, the melting point of aluminium oxide is high. A lot of energy must be transferred to break its strong ionic bonds, and this is expensive. To reduce costs, powdered aluminium oxide is dissolved in molten cryolite. This ionic compound melts at a lower temperature than aluminium oxide, reducing costs. However, significant amounts of energy are required to melt the cryolite.
The electrolysis process
The diagram shows an electrolysis cell used to extract aluminium. Both electrodes are made of graphite, a form of carbon with a high melting point and which conducts electricity.
During electrolysis:
- at the cathode, aluminium ions gain electrons and form aluminium atoms
- at the anode, oxide ions lose electrons and form oxygen gas
The oxygen reacts with the carbon anodes, forming carbon dioxide. So the anodes are gradually oxidised. They must be replaced frequently, adding to the cost of producing aluminium.
Worked example - Higher
Explain, with the help of a half equation, how oxide ions are oxidised during the electrolysis of aluminium oxide.
The half equation is: 2O2- → O2 + 4e-. It shows that oxide ions lose electrons, and oxidation is loss of electrons.

What are Acids?
Acids
Acids form acidic solutions in water. Acids produce hydrogen ions, H+ in aqueous solution. For example:
HCl(aq) → H+(aq) + Cl-(aq)
Acidic solutions have pH values less than 7.
What are Alkalis?
Alkalis form alkaline solutions in water. Alkalis produce hydroxide ions, OH- in aqueous solution. For example:
NaOH(aq) → Na+(aq) + OH-(aq)
Alkaline solutions have pH values greater than 7.
What are Neutral Soloutions?
A neutral solution is neither acidic, nor alkaline. A neutral solution has a pH value of 7.
What is the pH scale?
The pH scale measures the acidity or alkalinity of a solution. The pH of a solution can be measured using a pH probe, or estimated using universal indicator and a colour chart.
Universal indicator is one example of an acid-alkali indicator. Indicators show whether a solution is acidic, neutral (pH 7) or alkaline. The table shows the colours for litmus paper.

What happens when Acids with Metals?
- Only metals above hydrogen in the reactivity series will react with dilute acids.
- The more reactive the metal then the more vigorous the reaction will be.
- Metals that are placed high on the reactivity series such as potassium and sodium are very dangerous and react explosively with acids.
- When acids react with metals they form a salt and hydrogen gas:
- The general equation is:
metal + acid ⟶ salt + hydrogen
- Some examples of metal-acid reactions and their equations are given below:

Are Metal & Acid Reactions Redox reactions?
- Metal-acid reactions are redox reactions
- Redox means reduction and oxidation at the same time
- If we analyse the ionic equation for the reaction between zinc and hydrochloric acid:
Zn + 2HCl ⟶ ZnCl2 + H2
- The ionic equation is:
Zn + 2H+⟶ Zn2+ + H2
- This equation can be further split into two half equations illustrating oxidation and reduction individually:
Zn → Zn2+ + 2e–
2H+ + 2e–→ H2
- The zinc atoms are thus oxidised as they lose electrons.
- The hydrogen ions are thus reduced as they gain electrons.
- Both reactions are occurring at the same time and in the same reaction chamber hence it is a redox reaction
What is a Base?
Bases and alkalis
A base is any substance that reacts with an acid to form a salt and water only. This means that metal oxides and metal hydroxides are bases.
Bases that are soluble in water are called alkalis and they dissolve in water to form alkaline solutions. For example:
- copper oxide is a base, but it is not an alkali because it is insoluble in water
- sodium hydroxide is a base, and it dissolves in water so it is also an alkali
What are Neutralisation reactions?
A neutralisation reaction is a reaction between an acid and a base. Remember:
- acids in solution are sources of hydrogen ions, H+
- alkalis in solution are sources of hydroxide ions, OH-
In acid-alkali neutralisation reactions, hydrogen ions from the acid react with hydroxide ions from the alkali:
H+(aq) + OH-(aq) → H2O(l)
Pure water is neutral (its pH is 7). A neutral solution can be produced if the correct amounts of acid and alkali react together. The change in pH during a neutralisation reaction can be measured using a pH probe and meter, or estimated using universal indicator solution and a pH colour chart.
An acid-alkali neutralisation is the reaction between hydrogen ions and hydroxide ions, forming water.
What happens when Metal Hydroxides and Metal oxides react with an acid?
Reactions of Acids with Metal Oxides and Metal Hydroxides
- Metal oxides and metal hydroxides act as bases
- When they react with acid, a neutralisation reaction occurs
- In all acid-base neutralisation reactions, salt and water are produced
- The following are some specific examples of reactions between acids and metal oxides / hydroxides:
2HCl + CuO ⟶ CuCl2 + H2O
H2SO4 + 2NaOH ⟶ Na2SO4 + 2H2O
HNO3 + KOH ⟶ KNO3 + H2O

What happens when Acids react with Metal Carbonates?
- Acids will react with metal carbonates to form the corresponding metal salt, carbon dioxide and water
- These reactions are easily distinguishable from acid – metal oxide/hydroxide reactions due to the presence of effervescence caused by the carbon dioxide gas
The following are some specific examples of reactions between acids and metal carbonates:
2HCl + Na2CO3 ⟶ 2NaCl + H2O + CO2
Reactions with carbonates
A salt, water and carbon dioxide are produced when acids react with carbonates. In general:
Acid + carbonate → salt + water + carbon dioxide
For example:
Hydrochloric acid + copper carbonate → copper chloride + water + carbon dioxide
2HCl(aq) + CuCO3(s) → CuCl2(aq) + H2O(l) + CO2(g)
H2SO4 + CaCO3⟶ CaSO4 + H2O + CO2














