To calculate the atomic mass of a number, what do you need to know?
The number of stable isotopes of the element, the mass of each isotope, and the natural percent abundance of each isotope.
define the term atomic number of an element?
the number of protons in the nucleus of an atom
What is an Atom?
- Atoms are the smallest particles of an element that have the chemical properties of that element.
- All atoms consist of three particles, called protons, neutrons and electrons.
- Each atom has a nucleus at approximately the centre of its volume consisting of the two types of particles called protons and neutrons.
- The nucleus of an atom is positively charged.
- Electrons are distributed around the volume of the atom of which they are a part in “shells”, “orbits” or “energy levels”
In 1913, Niels Bohr suggested an explanation for the emission spectrum by proposing a model for the hydrogen atom. What did he propose?
- He proposed that electrons of specific energy move around the central nucleus in circular orbits or energy levels.
- Electrons cannot exist between these orbits
- Although an electron cannot lose energy while orbiting a nucleus, it could be given excess energy (by a flame or electric current) and then move to a higher orbit. If this happens, the electron has moved from the ground state (lowest energy level) to an excited state.
- When it drops back down to a lower, more stable orbit, this excess energy is given out as a photon or quantum of light. This is seen as a line of a particular colour on the visible spectrum.
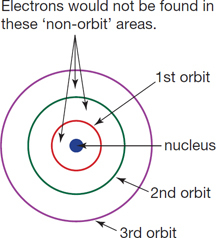
What are 5 Problems with the Bohr Model?
- It violates the Heisenberg Uncertainty Principle because it considers electrons to have both a known radius and orbit.
- The Bohr Model provides an incorrect value for the ground state orbital angular momentum.
- It makes poor predictions regarding the spectra of larger atoms.
- It does not predict the relative intensities of spectral lines.
- The Bohr Model does not explain fine structure and hyperfine structure in spectral lines.
- It does not explain the Zeeman Effect.
List of 5 important facts about Compounds.
- Compounds consist of molecules formed from atoms of 2 or more different elements bound together chemically.
- Compounds can be broken down into a simpler type of matter (elements) by chemical means; not by physical means
- Compounds always contains the same ratio of component elements.
- Compounds have properties different from their component elements
- e.g. the compound water (H2O) is a liquid at room temperature and pressure and has different chemical properties from those of the two elements, hydrogen (H2) and oxygen (O2), from which it is formed.
- Compounds can be represented using chemical formulae.
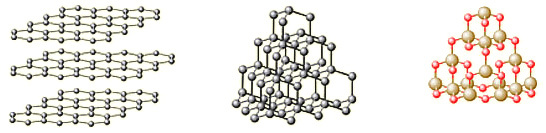
- Draw a diagram to show the arrangement of carbon atoms in a diamond crystal.

Do all covalent bonds conduct electricity?
NO (except graphite and graphene)
- Diamond, graphite and silicon dioxide are all examples of giant covalent structures. What does the word giant mean in this context?
- it continues on and on in three dimensions.
- Draw a diagram or diagrams to show the arrangement of carbon atoms in a graphite crystal.

Give 3 examples of a giant covalent structure
- Graphite
- Diamond
- SiO2
What is electrical current?
The movement of charged particles such as ions or electrons in a circuit.
Whose experiments were to find the quantity of charge carried by an electron?
Robert A. Millikan started his work on electron charge in 1906 and continued for seven years. His 1913 article announcing the determination of the electron’s charge is a classic and Millikan received the Nobel Prize for his efforts.
- What is electron configuration?
- The arrangement of electrons in the shells is called the atom’s electron configuration
- The electron capacity of each shell is limited.
- The maximum number of electrons that each shell can hold is 2n2 where n is the shell number or energy level.
- Electron shells are filled in order from the nucleus, starting with the K shell, so that the electrons are in their lowest possible energy levels (or ground state).
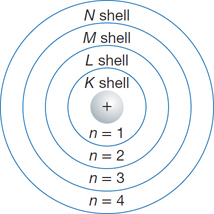
What are some Key Facts about Electrons?
- Electrons exist in the volume of atoms that surrounds the nucleus (see the diagram of an atom, on the right) and not in the nucleus of atoms - which is where the protons and neutrons are located.
- Electrons are therefore subatomic particles but they are not nucleons.
- Electrons have a negative charge of -1.
- An electron has a mass approximately 1/1836 of the mass of a proton, so a relative mass of 1/1836 ≈ 0.0005 ≈ zero. Electrons therefore account for very little of the total mass of the atom of which they are a part.
- The number of electrons in an atom is the same as the number of protons in the atom. That is because atoms are charge neutral, and protons have a charge of +1 while electrons have a charge of -1 so there must be the same number of each in order for the whole atom to be charge neutral. Recall that the number of protons in an atom is the atomic number of that atom (element).
How do you know how many electrons are in an element based upon what you see in the periodic table?
Check the atomic number, there are equal protons and electrons
When was the electron discovered? and
Who discovered the electron?
The electron was discovered in 1897 by British physicist J.J. Thomson who was working with Cathode Rays at the Cavendish Laboratory, Cambridge University.
Are electrons positively charged or negatively charged?
Negatively charged.
Elements are different because they contain different numbers of _______.
protons
Elements of the same period have the same number of
Electron shells
Elements such as O2 and Br2 are…
Diatomic molecules
Elements with the same group have the same number of…
Electrons in the outer shell
List 4 important facts about Chemical Elements.
- Elements consist of only one type of atom - which may, or may not be joined together to form molecules or large structures, so …
- Elements can exist either as atoms (e.g. argon) or as molecules (e.g., nitrogen)
- Elements cannot be broken down into a simpler type of matter by either physical or chemical techniques - though some larger elements break-down spontaneously due to being radioactive.
- Elements are listed in the periodic table.
What is meant by Energy State in Chemistry?
The position and energy of an electron described by its quantum numbers is known as its energy state.






