Phytosomes
Phytosome is a complex of a natural active ingredient and a phospholipid.
This complex results from the reaction of stoichiometric amounts of phospholipid with the selected polyphenol (like simple flavonoids ) in a nonpolar solvent.
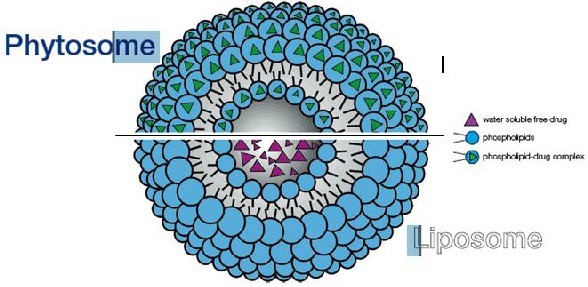
Differences between Lyposomes and phytosomes
- In liposomes no chemical bond is formed and the phosphatidylcholine molecules surround the water soluble substance.
- In a liposome, the material is simply emulsified.
- In phytosomes the phosphatidylcholine and the plant components actually form a 1:1 or a 2:1 molecular complex depending on the substance(s) complexes, involving chemical bonds.
- This difference results in better absorption of phytosomes than liposomes showing better bioavailability.
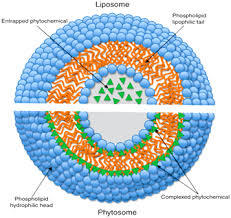
Lyposomes VS. phytosomes
- Liposomes are much bigger than phytosomes
- There is the formation of new bonds in phytosomes, whereas no chemical bond is formed in liposomes.
- In liposomes, there are hundreds or thousands of PC ( phosphatidylcholine ) molecules surrounding the water-soluble compounds. Conversely, the molar ratio of PC ( phosphatidylcholine ) and natural ingredients is 1:1 or 2:1 for phytosomes depends on the chemical bond forming material
- In liposomes the natural active ingredients are dissolved in the medium or wrapped by the membrane. In phytosomes the active ingredients are anchored through chemical bonds to the polar head of phospholipids.
- In liposomes, the ingredients are dissolved in the central part of the cavity, with limited possibility of molecular interaction between the surrounding lipid and a hydrophilic substance.
- In a Phytosome® the ingredient is dispersed into lecithin, a dietary surfactant and can be compared to an integral part of the lipid membrane.
Cubosomes
Cubosomes are nanoparticles but instead of the solid particles, cubosomes are self- assembled liquid crystalline particles of certain surfactant with proper ratio of water with a microstructure that provides unique properties of practical interest.
Cubosomes consist of honeycombed (cavernous) structures separating two internal aqueous channels and a large interfacial area.
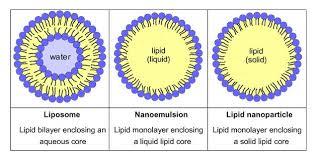
Liposome vs Nanoemulsion vs Lipid nanoparticle
- Liposome: lipid bilayer enclosing an aqueous core
- Nanoemulsion: lipid monolayer enclosing a liquid-lipid core
- Lipid nanoparticle: lipid monolayer enclosing a solid-lipid core
Polymersomes
- Polymersomes are nanostructures composed of amphiphilic block copolymers that have a size range from 50 nm to 5 μm and encapsulate drugs inside the vesicle membrane.
- They are capable of encapsulating hydrophobic and hydrophilic drugs and they can be surface functionalized.
- Polymersomes share many similarities with liposomes, but are **more stable and less permeable to small water-soluble molecules than liposomes. **
Reverse micelles
Phase inversion of particle-stabilized emulsions from oil in water to water in oil can be achieved either by
variation of the particle hydrophobicity (transitional) or by variation of the oil/water ratio (catastrophic).
Application of a liposome
LN (liposomal nanomedicines can be used to deliver cancer cell-killing drugs into tumours
Emulsion
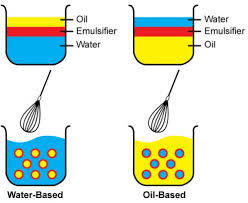
Emulsion – is a mixture of two or more liquids that are normally immiscible.
In an emulsion, one liquid (the dispersed phase) is dispersed in the other (the continuous phase)
Emulsifying Agents
Emulsifying Agents are the substances added to an emulsion to prevent the coalescence of the globules of the dispersed phase.
These agents have both a hydrophilic and a lipophilic part in their chemical structure. (i.e. amphiphiles)
All emulsifying agents concentrate at and are adsorbed onto the oil:water interface to provide a protective barrier around the dispersed droplets.
In addition to this protective barrier, emulsifiers **stabilize the emulsion by reducing the interfacial tension of the system. **
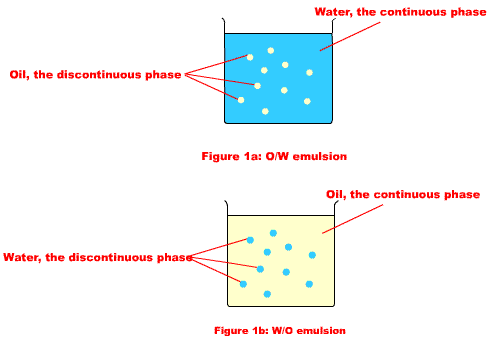
Classification of emulsions
- Based on dispersed phase:
- Oil in Water (O/W): Oil droplets dispersed in water
- Water in Oil (W/O): Water droplets dispersed in oil
- Based on size of liquid droplets:
- 0.2 – 50 mm Macroemulsions (Kinetically Stable)
- 5-200 nm Microemulsions (Thermodynamically Stable)
Common Emulsifying Agents
- Surfactants:
- Anionic: sodium stearate, potassium laurate
- Nonionic: polyglycol, fatty acid esters, lecithin.
- Cationic: Quaternary ammonium salts
- Solids: Finely divided solids with amphiphilic properties such as soot, ** silica, and clay. **
- For microemulsion only surfactantas are know to be active as emulsifier.
- For macroemulsion, a greater variety of components such as surfactants, polymers, and colloidal particles (or nanoparticle) can be active as emulsifiers.
Bancroft’s rule for emulstions states:
“The phase in which an emulsifier is more soluble constitutes the continuous phase.”
- In Oil in Water emulsions – use emulsifying agents that are more soluble in water than in oil (High HLB surfactants).
- In Water in Oil emulsions – use emulsifying agents that are more soluble in oil than in water (Low HLB surfactants).
What affects the type of emulsion?
- the ratio of the oil to water(non-polar to polar)phase;
- •the chemical properties and the concentration of the emulsification agent;
- the temperature; the presence of **additives; **
- for solid particles as the stabilizing agents (Pickeringemulsions) the wetting conditions (contact angles of the oil and water phases on the solid)
Many synthetic and herbal drugs possess the problem of poor oral bioavailability, and the reason are: $$
- their very low water solubility
- poor permeation through the biological membrane.
- Poorly soluble drugs have suffered from low bioavailability and inefficacy in therapy due to their low dissolution profile in biological fluid.
- Without a proper level of drug concentration in the gastrointestinal (GI) fluid, the drugs cannot be effectively transported by the epithelia of the GI tract, resulting in **low systemic absorption. **
- Several plant actives in spite having potent in vitro pharmacological activities have failed to demonstrate similar in vivo response.
although most bioactive molecules of plants are biologically polar or water-soluble, they are difficult to pass through the lipid-rich biological membrane and be absorbed by human, the reasons of which include: $$
1) large molecular weight,
**2) low lipid solubility **
The interaction had been atributed to formation of hydrogen bond and/or hydrophobic interaction between the two molecules.
Shapes of aggregates ($$often)
Basic shapes:
Micelles’ shapes:
Superstructures:
Basic shapes:
- spheres
- cylinders
- bilayers
- vesicles
micelles’ shapes:** spheres and cylinders**
Superstructures:
- micellar crystals
- lamellar phases
- bicontinuous networks
Benefits of phyto-phospholipid complexation
- Enhanced bioavailability: (+) facilitaing membrane permeation, (+) Improving solubility
- Reduce Dose –> Enhancing systemic absorption
- Value addition (+)lipid profile improvement (+)Hepato protection
- Hepatic/Tumor Targeting –> passive targeting
- Sustained action –> modified release profile
- Safe –> Non toxic and non mutagenic (FDA approved)
- Are most bioactive molecules of plants polar or water-soluble?
- Why these bioactive molecules of plants are difficult to pass through the lipid-rich biological membrane and be absrobed?
- Why phytosomes are more bioavailable than conventional herbal extracts?
- Yes,
- (1) large molecular weight, (2) low lipid solubility (3) formation of H(+) bond and/or hydrophobic interactions between drug and lipid (phyto-phospholipid complex). Van der Waals forces.
- Phytosomes have enhanced capacity to cross the lipoidal biomembrane and reach the systemic circulation

Cochleates:
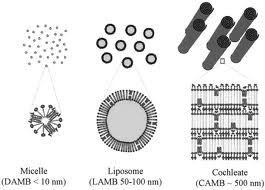
phospholipid-calcium precipitates derived from the interaction of anionic lipid vesicles with divalent cations such as calcium.
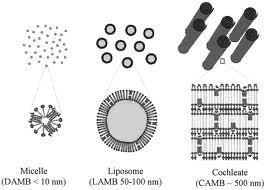
Cochleates have a defined multilayered structure consisting of a solid, lipid bilayer sheet rolled up in a spiral.
Cochleates were first described by Papahadjopoulos in 1975 as an intermediate in the preparation of large unilamellar liposomes,
They have the ability to deliver a variety of drugs, such as antifungal agents, polypeptides, proteins, vaccines, oligonucleotides and genes.
What kind of drugs can be encapsulated in cochleates?
Cochleates contain both hydrophobic and hydrophilic surfaces, they are suitable to encapsulate both hydrophobic drugs like amphotericin B and clofazimine and hydrophillic drugs like doxorubicin.
- Hydrophilic molecules can be accommodated between the lipid bilayers
- Hydrophobic molecules can be accommodated in the inner hydrophobic space
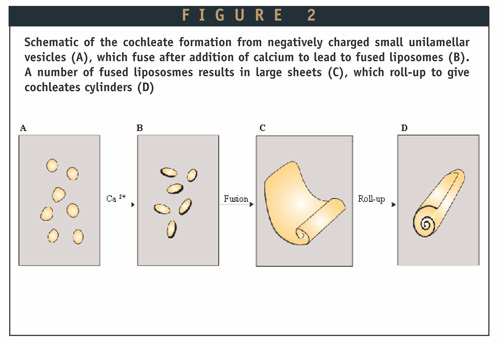
Cochleates formation.
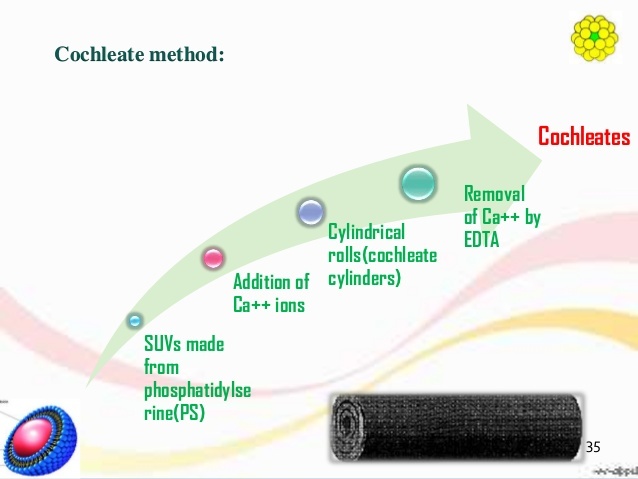
- Cochleates are formed as a result of the condensation of small unilamellar negatively charged liposomes.
- In the presence of calcium, the small phosphatidylserine (PS) liposomes fuse and form large sheets. These sheets have hydrophobic surfaces and, in order to minimize their interactions with water, tend to roll-up into the cigar-like cochleate.
- For cochleates, the molar ratio of divalent cations and phospholipids is:
- Ca2+ function in the cochleate
- Amphotericin B is an an;fungal drug often used intravenously for systemic fungal infections. (Fungilin, Fungizone, Abelcet, AmBisome, Fungisome, Amphocil, Amphotec).
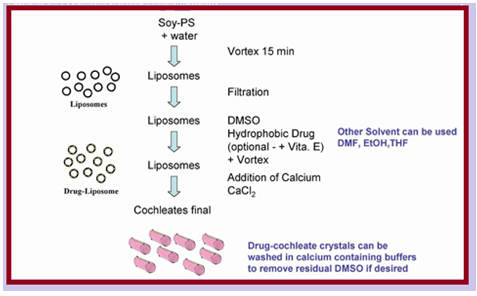
Describe synthesis
- For cochleates, the molar ratio of divalent cations and phospholipids is 1:2.
- Ca2+ maintains the cochleate in its rolled form, and bridges each successive layer through ionic interaction.
- The loading efficacy of the cochleates depends upon the (2): $$$
- the particle size of the drug-cochleate complex depends on (1): $$$
- The main components of cochleates are (2) $$
- The loading efficacy of the cochleates depends upon the physical chemistry of the drug to encapsulate
- the particle size of the drug-cochleate complex depends on the process used to encapsulate.
- The main components of cochleates are phosphatidylserine (PS) and calcium, two natural compounds.






