All physiological processes are based on…?
-Chemical interactions
What are the 6 elements the body is composed of?
99% of the human body is made of 6 elements. -Carbon, C -Hydrogen, H -Oxygen, O -Nitrogen, N -Phosphorus, P -Sulfur, S
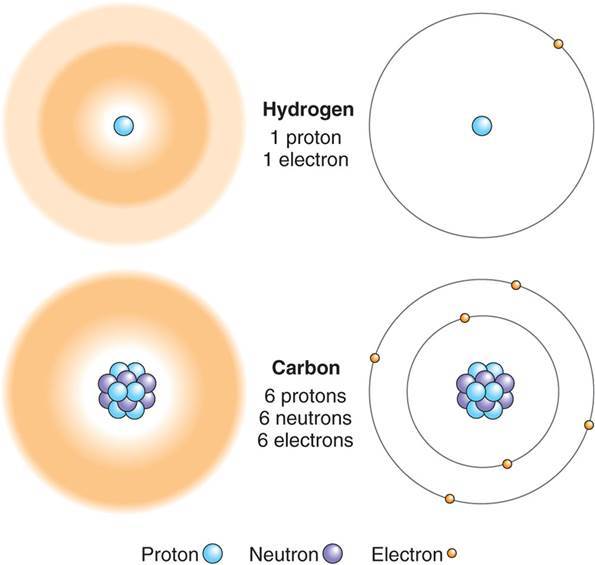
What are atoms and what do they contain?
-Atoms are the smallest unit of an element, they contain protons (+ charged, in nucleus), neutrons (uncharged, in nucleus), and electrons (- charged, orbiting nucleus).
What are chemical bonds?
-Strong force of attraction holding atoms together in a molecule or crystal as a result of sharing or transfering electrons.
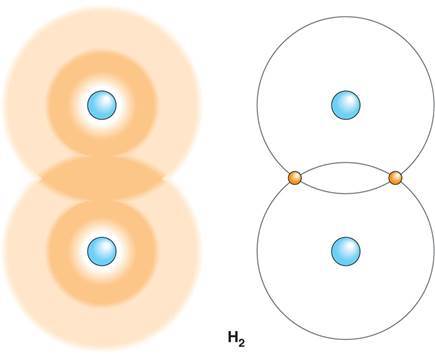
What are covalent bonds?
- A chemical bond formed by the sharing of valence electrons between atoms.
- Can be nonpolar: electrons are shared equally
- Can be polar: electrons are not shared equally
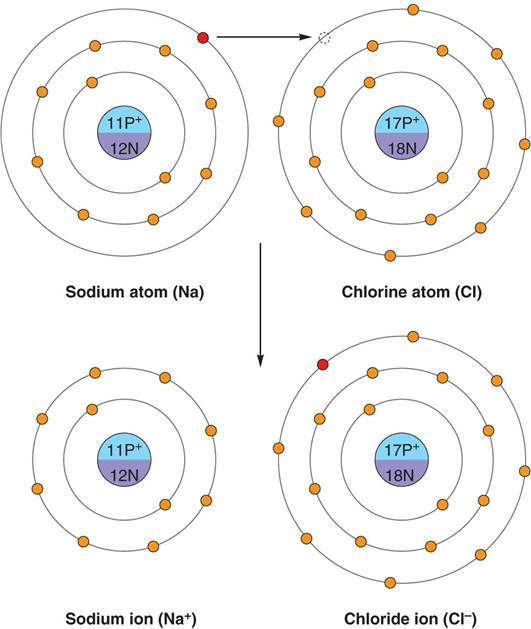
What are ionic bonds?
-A chemical bond in which an atom donates an electron to another atom to achieve filled outer shells for both atoms. -Electron donor becomes + charged (cation) -Electron receiver becomes - charged (anion)
How much of the human body is composed of water?
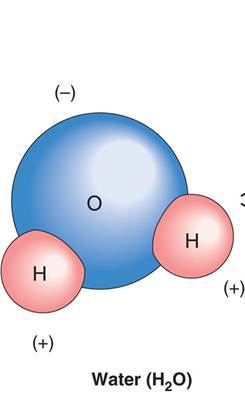
-65% of the human body is made up of water and other fluids
What are some valuable properties of water?
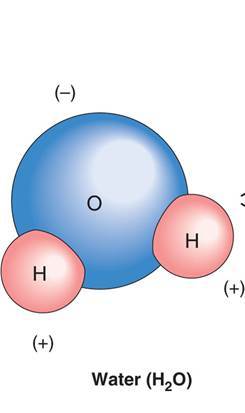
- Good solvent (most substances dissolve in it)
- Polar molecule
What are molecules that dissolved readily in water called? How about molecules that do not dissolve readily in water?
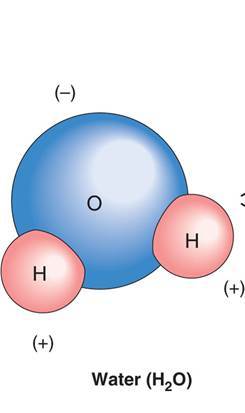
- Molecules that dissolve readily in the water are Hydrophilic, tend to be ionic compounds
- Molecules that do not dissolve readily in water are Hydrophobic.
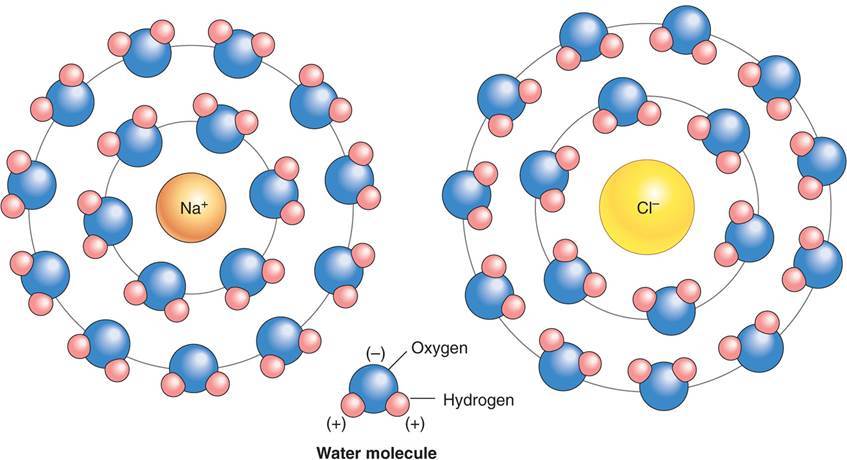
What are ions? What happens when an ionic compound is placed water?
- Ions are atoms that are electrically charged as a resulting of “gaining” or “losing” an electron.
- When ionic compounds are placed in water, they tend to dissociate and revert back to their constuient ions.
- Negative side of water is attracted to (+) cation (atom that lost electrion)
- Postive side of water is attracted to (-) anion (atom that gained electron)
What is a Hydrogen Bond?

- (Intermolecular interaction) It is a weak bond formed between two polar molecules based on opposite charged attracting.
- Happens between water molecules, amino acids in a protein –> shape, and it holds the two strands of the DNA molecule together.
What is an acid? What makes a solution acidic?
-An acid is a chemical species that donates protons or hydrogen ions and/or accepts electrons. A solution is acidic when more H+ ions than OH- ions are present. pH is <7.
What are Bases? When is a solution Basic?
-An aqueous substance that can accept hydronium ions. A solution is basic when there ar more OH- ions than H+ ions. The pH would be >7.
When is a solution neutral?
-When H+ ions are equal to OH- ions. The pH = 7
What are Buffers? What do they do? How does it apply to blood?
- Solutions used to stabilize an optimum pH of a liquid.
- Buffers neutralize H+ concentration in solutions.
-In blood, pH is stablized by bicarbonate (HCO3-) and carbonic acid (H2CO3) system:
HCO3- + H+ <–> H2CO3 <–> H2O + CO2
- If blood falls below pH 7.35, condition is called Acidosis (too little base)
- If blood falls below pH 7.45, condition is called Alkalosis (too little acid)
What are Organic Molecules? What is a crucial element for Organic Molecules? Why is it crucial?
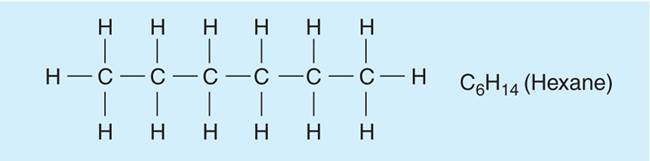
- Organic molecules are molecules that contain Carbon (C) and Hydrogen (H)
- Carbon, an important element, has four outer electrons and forms 4 bonds.
- Carbon forms the backbone of all organisms on earth in terms of…
- Proteins
- Carbohydrates
- Lipids
- Nucleic acids
- Chains and rings of carbons bonded to other atoms.
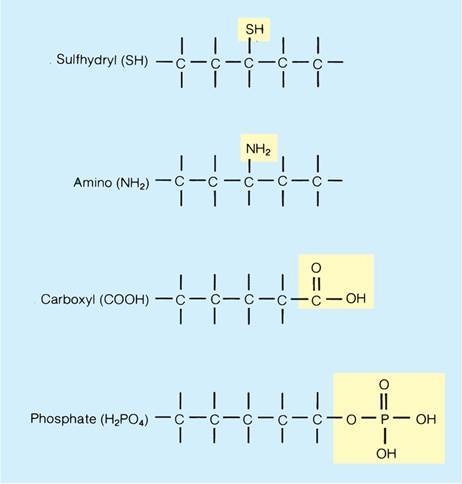
What are functional groups? How does Carbon play into this?
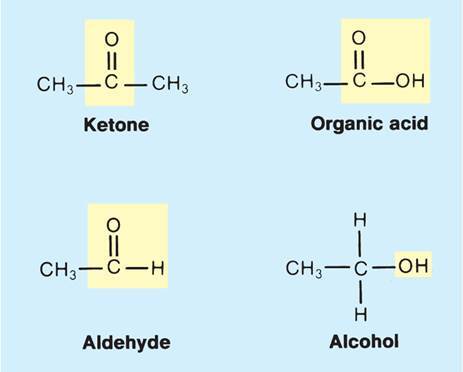
- A group of atoms reacting as one: a group of atoms that reacts as a single unit and determines the properties and structure of a class of compounds.
- Carbon forms the backbone for these reactive groups.
- These classes of molecules are named after their functional group.
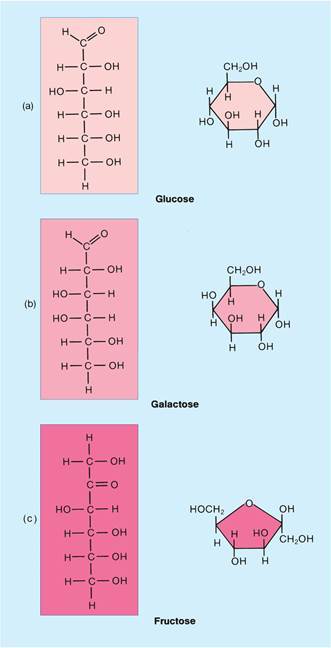
What are Carbohydrates? What function do they serve in our body? Give examples of these carbohydrates.
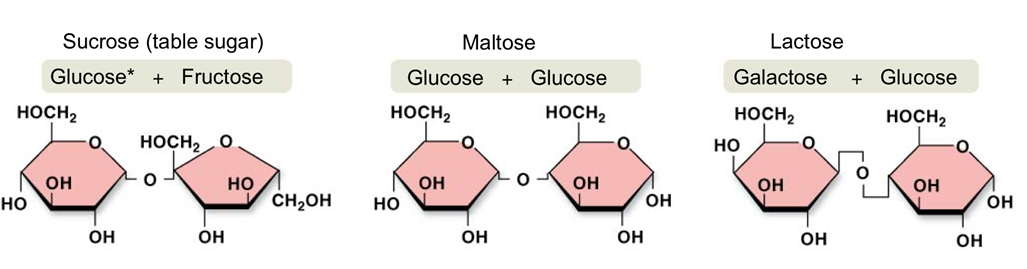
- Carbohydrates are organic molecules such as sugars and starch that contain carbon, hydrogen, and oxygen.
- Serve as the primary source of energy in our bodies.
- A simpler form of a carbohydrate is a monosaccharide (meaning “mono” = single, “sacchar” meaning sugar). Some monosaccharide sugars include: glucose, fructose, galactose.
- A disaccharide is two monosacchardies joined by a covalent bond: sucrose, maltose, lactose
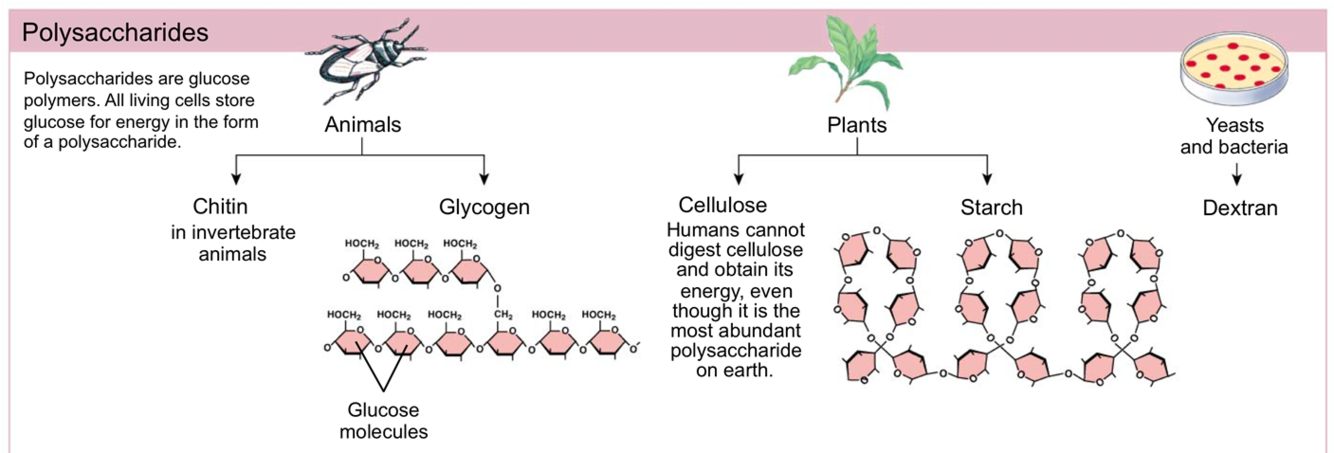
- A polysaccharide is many monosacchardies joined together: starch.
What is Glycogen? What is Cellulose?
- Glycogen is a polysaccharide made in liver cells and muscle cells of animals to store sugar in a cell.
- Cellulose is a polysaccharide made by plants that is not digestible by humans.
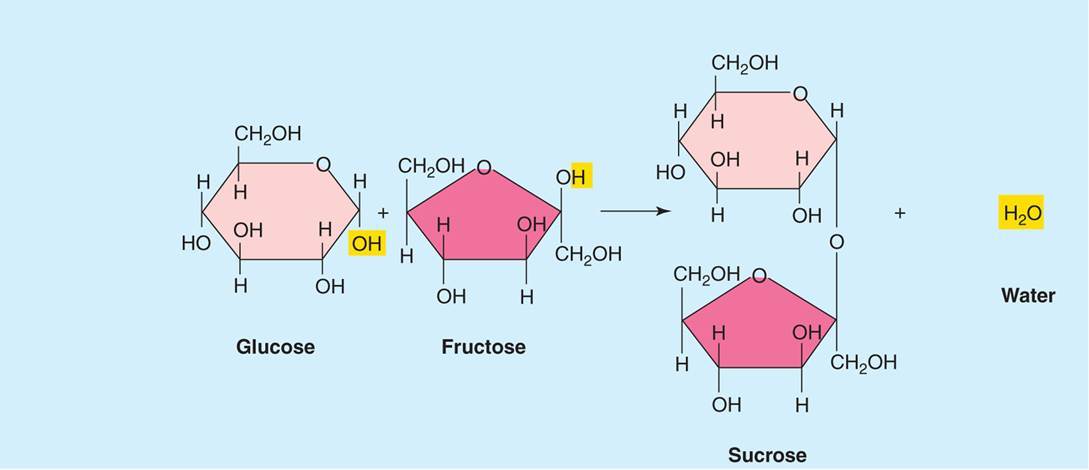
How are bonds that hold monosaccharides together are formed?
- Monosaccharide covalent bonds are formed by dehydration synthesis.
- Dehydration synthesis is when hydrogen is removed from one molecule and hydroxyl is removed from another to form water. This inturn causes the monomers to join together.
How are bonds broken between molecules such as monosaccharides?
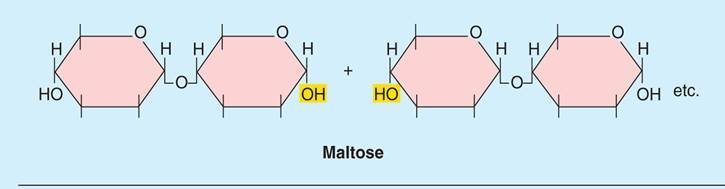
- Hydrolysis is a chemical reaction or process in which a chemical compound reacts with water to break a bond between its molecules.
- Water is split and used to complete individual molecules such as monosaccharides.
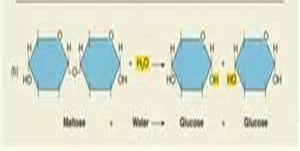
What does Dehydration Synthesis and Hydrolysis do to help with Physiological Functioning?
-Dehydration synthesis and hydrolysis help by building/breaking fats, proteins, and nucleic acids.
What are lipids? Are they soluble in water? What are common types of lipids?
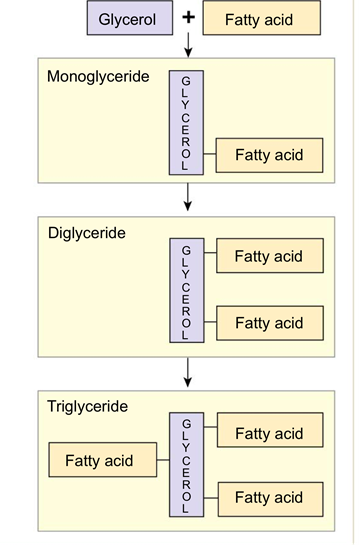
- They are nonpolar hydrocarbon chains and rings.
- They tend to be insoluble in water or polar substances.
- Fats, oils, fatty acids, phospholipids, steroids, waxes. Triglycerides are made up of glycerol and three molecules of fatty acids.
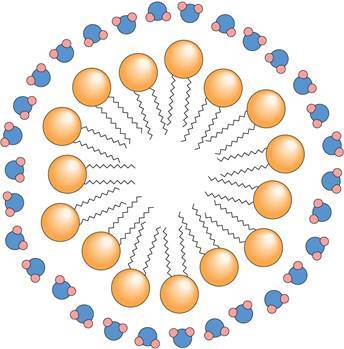
What are phospholipids? Where can they be located? How are they positioned? How do they help transport lipids throughout the body?
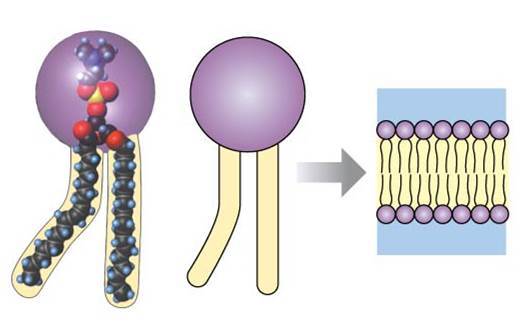
- They are lipids (nonpolar hydrocarbon chains and rings) combined with a polar phosphate group.
- They are located on the cell membrane, forming a bilayer which has hydrophilic phosphates poing outward on each side and hydrophobic fatty acids and glycerol pointing inward.
- The phospholipids form micelles (which are like tiny balls of molecules) to help suspend and transport lipids in aqueous environment of body.