Explain atoms
- All matter is made up of tiny particles called atoms
- Smallest particle still characterizing a chemical element
- Made up of subatomic particles
- 4 atomic theories developed over time
(Billiard Balls → Raisin Bun → Solar System → Energy level)
What are subatomic particles?
- Particles that make up the atom (including electrons, neutrons, and protons)
Deescribe the components of an atom
[The atom’s nucleus is made up of protons and neutrons, which altogether make up most mass of the atom]
- Protons (p+) can NEVER change, otherwise is a different element
- Neutrons (no) can change
- Electrons (e-) organized into energy levels, outside nucleus, can change
What is an isotope? How do you write an isotope?
Atoms of the same element with different # of neutrons
Written as Name-Mass #
mass # ELEMENT SYMBOL
atomic #
Describe nuclear notation
[How to find # of protons? / What does the atomic mass number represent? / How to find neutrons?]
Atomic Number = # of protons & electrons, states what element
Mass number = # of protons & Netrons
of neutrons = mass # – atomic #
What types of elements can be found on the periodic table? Where can they be found, and what properties do they have?
Metals
- Left side of staircase
- Properties; ductile, malleable, conductive
Non-metals
- Right side of staircase
- Properties; dull, non-conductive
Metalloids
- On staircase
- Properties; brittle, some shiny, some conductive, poor conductors
What are ions?
Atoms that have gained or lost electrons to become stable & postively or negatively charged
Cations (+):
- Positive ions (loss of electrons)
- Metals
- End is ion
Anions (-):
- Negative ions (gain electrons)
- Non-metals
- End is -ide (ie. Fluoride)
What elements are diatomic in their most natural state?
Iodine, bromine, chlorine, fluorine, oxygen, nitrogen, hydrogen, phosphorus (4), sulfur (8)
I2 Br2 Cl2 F2 O2 N2 H2 P4 S8
I bring clay for our new house 4pouring 8sidewalks
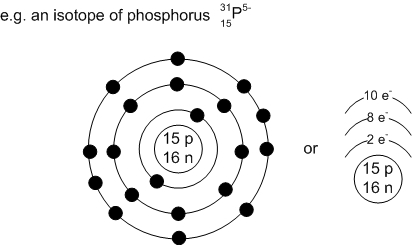
What does a Bohr’s Energy level diagram include? What are valence electrons?
Includes: atomic symbol, # of protons, # of neutrons, # of electrons, and energy levels
Energy level organization:
1st energy level: 2 electrons (max)
2nd energy level: 8 electrons (max)
3rd energy level: 8 electrons (max)
(*288)
Valence electrons: electrons located on outermost shell
- Group # atom belongs to = # of electrons on outermost shell*
- Period # atom belongs to = # of energy levels*
What happens when electrons absorb a specific quantity of energy? What happens when they emitt that same quantity?
Absorb specific quantity of energy: move ↑UP to higher energy level
Emitt same quantity: move ← BACK to original energy level
What does a lewis dot / electron dot diagram include? Is there a difference between atom diagrams & ion diagrams?
Atom:
Includes: atomic symbol, # of valence electrons
ie:
☻
Na
Ion:
Includes: atomic symbol, # of valence electrons, square [] brackets (charge is displayed outside)
ie:
☻☻
[ ☻☻ Na ☻☻] +
☻☻
Explain the sections of the periodic table
(ie. Which groups are the most reactive?, When you go down a group, it becomes ______?, What properties do these sections have?)
When you go down a group, it becomes more reactive
Alkali metals (group 1) & Halogens (group 17) are the most reactive
Alkali metals (group 1): very soft, malleable, ductile, react easily with other substances
Alkaline Earth Metals (group 2): malleable, ductile, react with oxygen to form oxides
Transition metals (group 3-12)
Halogens (group 17): react with metallic elements, forms salts
Noble gases / Inert gases (group 18), unlikely to take part in chemical reactions
What is the octet rule? Any exceptions?
Octet rule: have 8 electrons on outer shell to become stable by gaining, losing, or sharing electrons.
[EXCEPTION: Hydrogen, Lithium, Beryllium only need 2 electrons (Helium is the closest noble gas)]
Describe ionic compounds (properties, naming, formation, electrons, etc.)
- Metal (+) and Non-metal (–)
- Metal named first, non-metal second, 2nd element is -ide
- Attracted by opposite charges (positive and negative)
- Transfer valence electrons
- Crystal Lattice is produced as solid; crystal shapes with flat sides as regular repeating pattern of ions
- High melting points - lot of energy required to break strong bonds
- Non-conductive when solid (non electrolyte) unable to seperate into + & - charges,
- If melted/dissolved in solution becomes conductive (electrolyte) can seperate into opposite charges
Describe covalent (molecular) compounds (properties, naming, formation, electrons, etc.)
- Non-metal & non-metal
- Element named first goes first, 2nd element ending is -ide
- Uses prefixes (mono, di, tri, penta, etc.), never use mono for 1st element
- Share valence electrons
- Low melting points - attractive forces between molecules are weak, easy to overcome
- Non-electrolytes - never conduct electricity, do not seperate into positive and negative ions as solid or dissolved.
- Intramolecular bond (within molecules)
Desrcibe the different types of ionic compounds.
Binary ionic: just 2 elements
Multivalent ionic: metal elements that have more than 1 possible charge, when writing the name of the formula use roman numerals (stock system) to indicate charge (I,II,III,IV,V, etc.)
Polyatomic: more than 2 elements, ending does not change, listed as second unless NH4
What happens to the metal and the non-metal when the compound is formed?
Metals:
Tend to lose valence electrons, become positive
Non-metals:
Tend to gain valence electrons, become negative
List the properties of acids and bases
Acids:
- Taste sour
- Corrode metals
- Turn litmus red
- pH lower than 7
- contain H+ (aq) (hydrogen) ions
Bases:
- Taste bitter
- Feel slippery (“soapy”)
- Turn litmus blue
- pH higher than 7
- contain OH- (aq) (hydroxide) ions
The more ions in an acid the more ____ it is
The more ions in an acid the more acidic it is
[More ions create a stronger acid]
What does Arrhenius’ theory of acids and bases state?
- Only behave as acids/bases when dissolved in water (otherwise just regular compounds)
- Must always be written with (aq)
- Both conduct electricity (electrolytes), (solutions must contain freely moving positive and negative ions)
How would you write the chemical formula for water? Is it ionic or covalent? (Include IUPAC name)
Water is covalent
IUPAC name:
dihydrogen monoxide
Chemical formula
H2O
List the (seven) properties of water
Intermolecular bonds: hydrogen bond between water are the strongest overall type of intermolecular bond (forces of attraction between molecutes
High BP & MP: lots of energy is required to increase temperature and break bonds
Surface tension: sticks strongly together (forms droplets)
Ice floats in water: less dense, forms large open pattern of polar molecules when frozen
Concave meniscus: sticks to itself + other surfaces because of large forces of attraction between molecules (like the sides of the container)
Capilliary action: draws water up
Polarity: bent, electrons pulled more strongly to O (slight -), less with H (slight +)
What is a physical change? What illustrates that it has occured?
Change to a substance where the chemical composition stays the same
- Shape change
- Phase change: moving from solid liquid, or gas
- Size change: increasing/decreasing
What is a chemical reaction? What evidence supports that it has occured?
When one or more substances change to form new substances
Evidence of a chem. reaction:
- Energy change: can be observed as teperature change, light emission, sound emission, electricity.
- Odour change: in some chem. reactions, the odour of the products differs from the odour of the reactants. (ie. odour of food after cooking)
- Colour change: in some chem. reactions, the colour of the product(s) differ from the colour of the reactants (ie. substances in bleach react with coloured compounds to form colourless compounds)
- Formation of a gas: gases are formed in solute chemical reactions (When the reaction takes place in a solution, you can observe the gas being formed as tiny bubbles)
- Formation of a solid (precipitate) in solution: in some chem. reactions, a substance is formed that is insoluble & comes out of solution as solid (ie. soap scum)


