What do we need to have a reaction take place
- Enough force in the collision 2. the right orientation
Ectothermic
If the products have less energy than the reactants
Endothermic
If the products have more energy than the reactants
Equilibrium
when the rate of the foreword reaction equals the rate of the reverse reaction
If k>1,
It favors the products
If k
it favors the reactants
Le chateliers principle
It is possible to shift an equilibrium to favor the reactants or the products
How does heat affect equilibrium
If heat is added, the equilibrium will shift to the side with the heat. If we remove it, it will shift away
How does adding substances affect equilibrium
If you add a substance, the equilibrium will shift away from it, if you remove it, than twoards
How does pressure affect equilibrium
only for gasses) increasing the pressure shifts the equalibrium to the side with fewer moles of gas.
In heat+H2S+I2 = 2Hi + S Adding more s
Shifts left
In heat+H2S+I2 = 2Hi + S Cooling the system
Shifts left
In heat+H2S+I2 = 2Hi + S Increasing the pressure
Shifts left
In heat+H2S+I2 = 2Hi + S Removing h2s
Shifts left
In heat+H2S+I2 = 2Hi + S Increasing the volume
Shifts right
What does a catalyst do
It lowers the activation energy
Ways to speed up a reaction
- Heat it up 2. Particle size. (Increase the surface area) 3. Add a catalyst (speeds up a reaction by lowering the activation energy - not used up in the reaction 4. Increase the concentration 5. Shrink the volume (primarily with gasses)
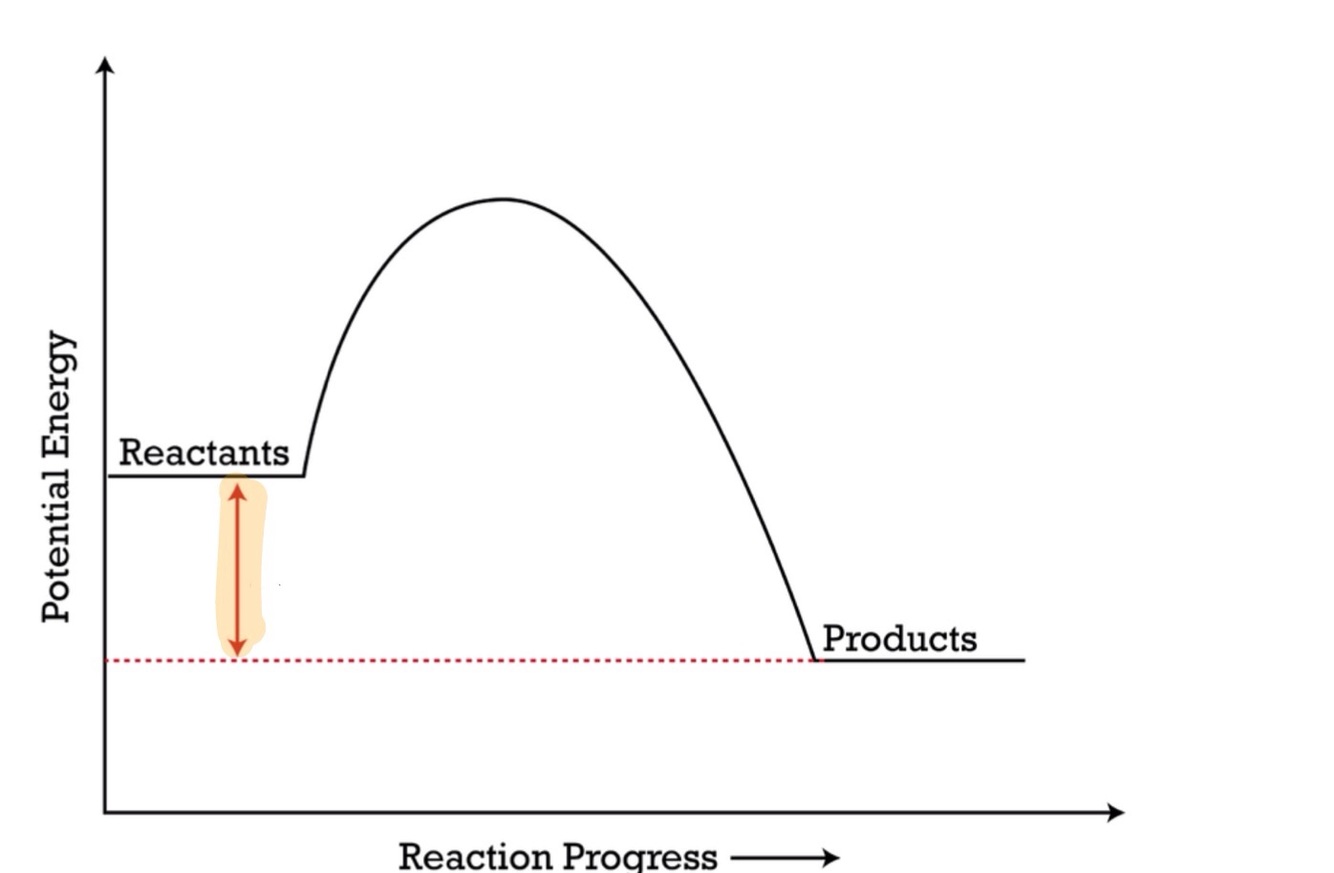
Delta h (enthalpy
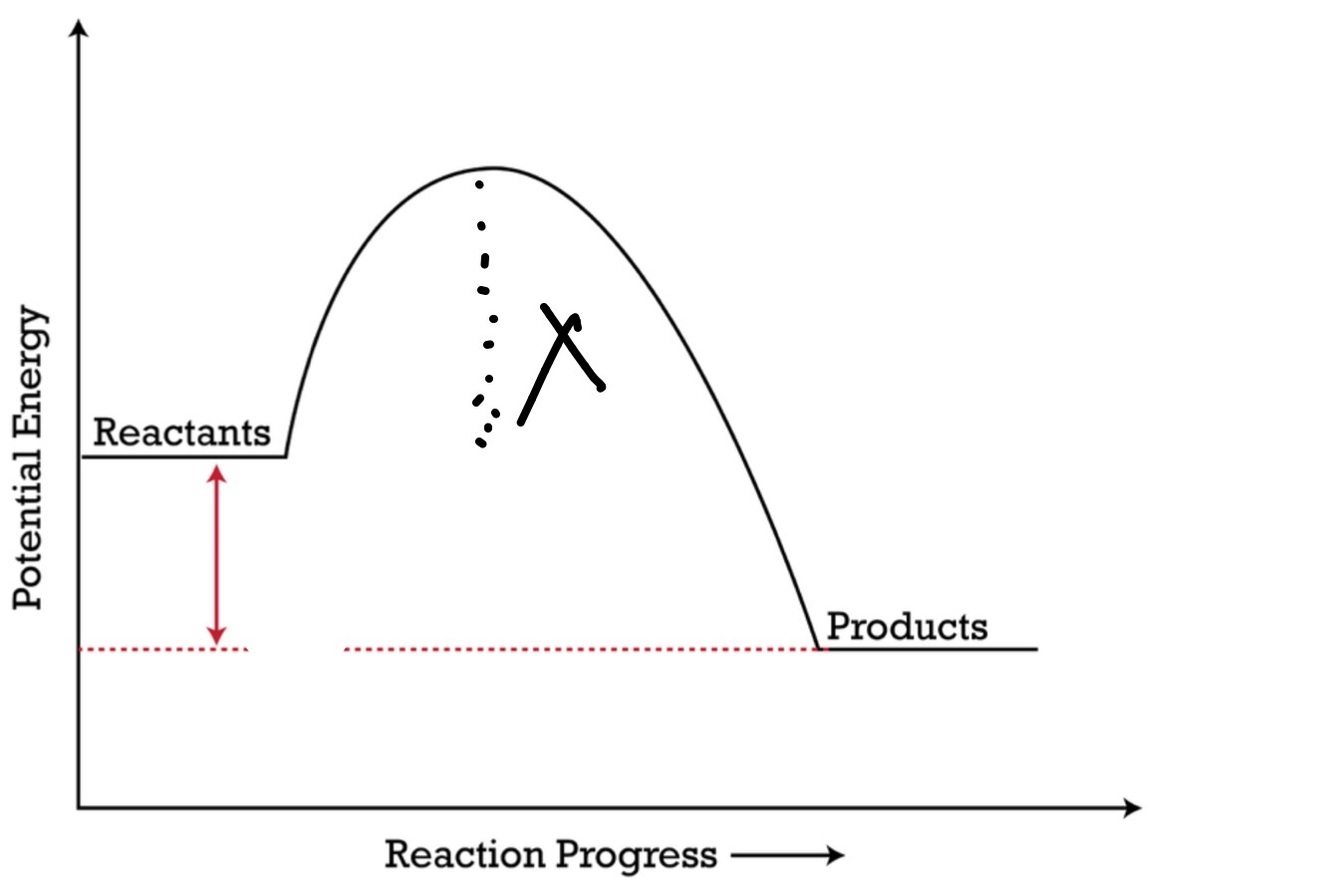
Ea. activation energy
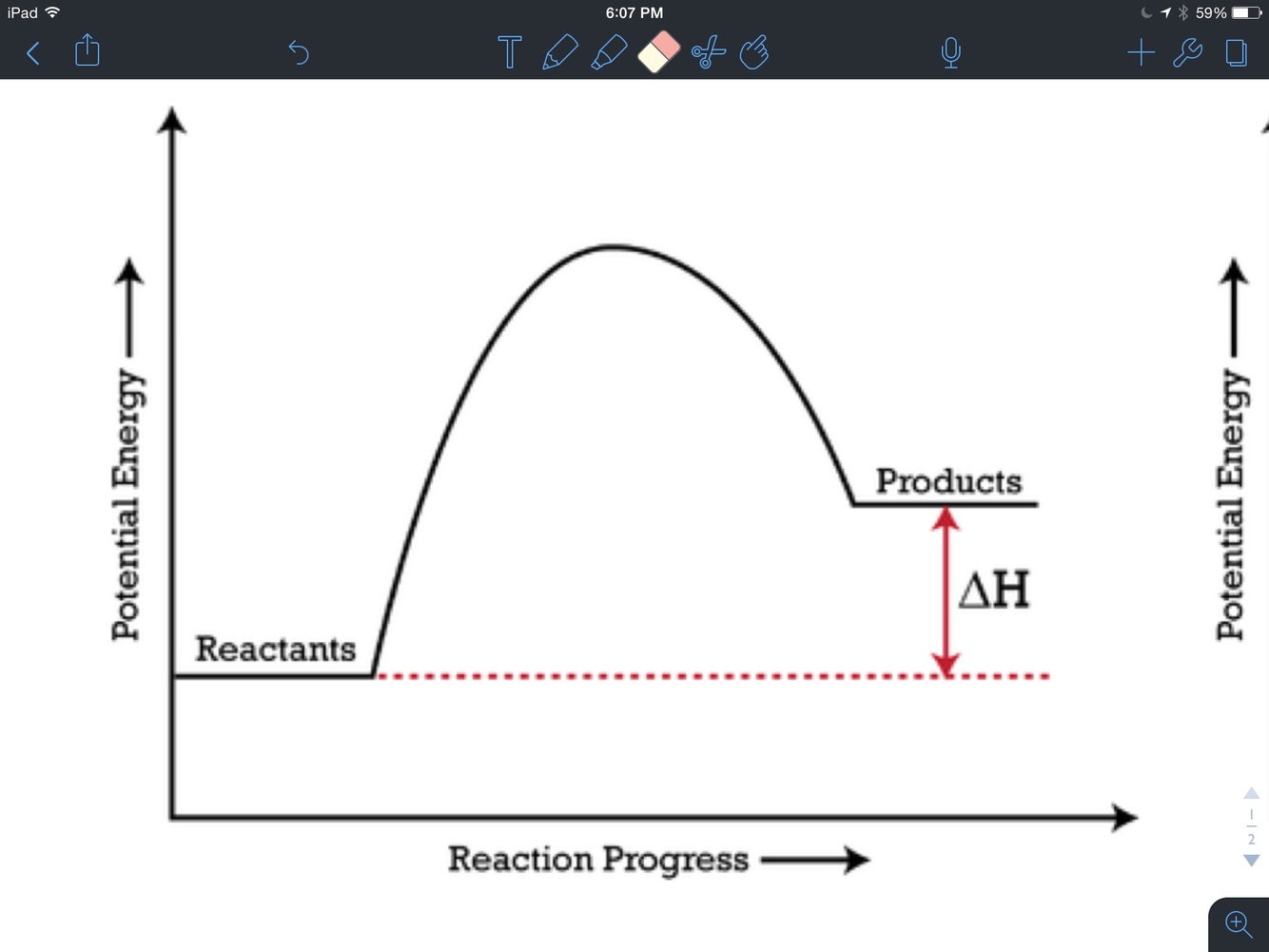
Endothermic
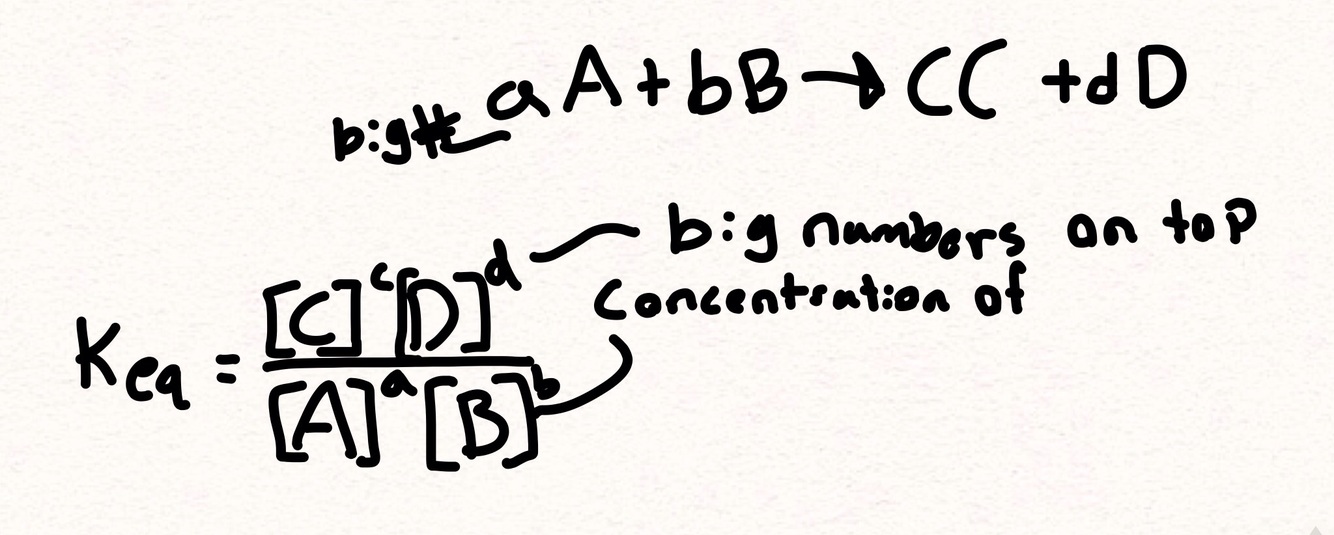
What is the equilibrium consistent formula
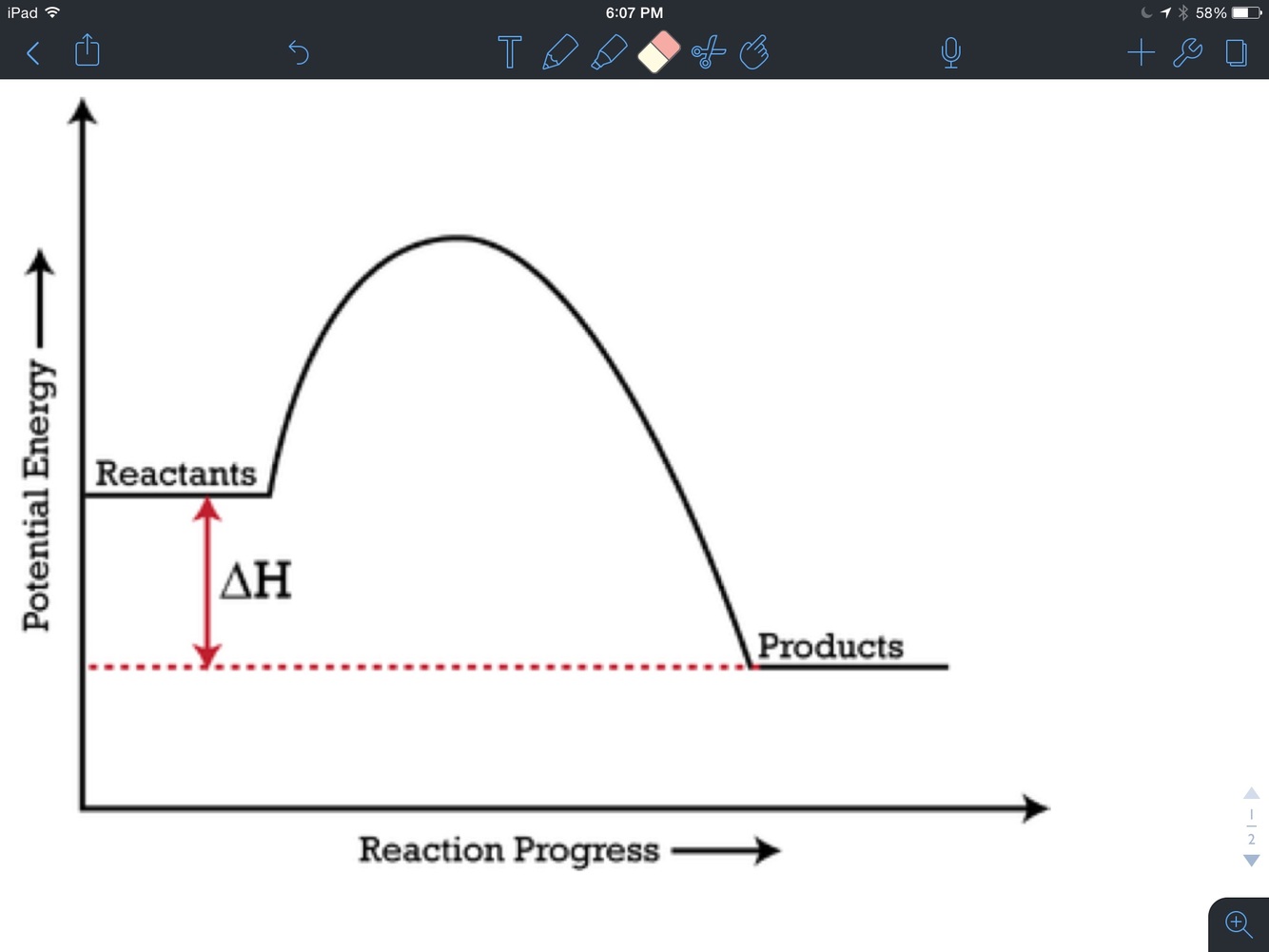
Exothermic

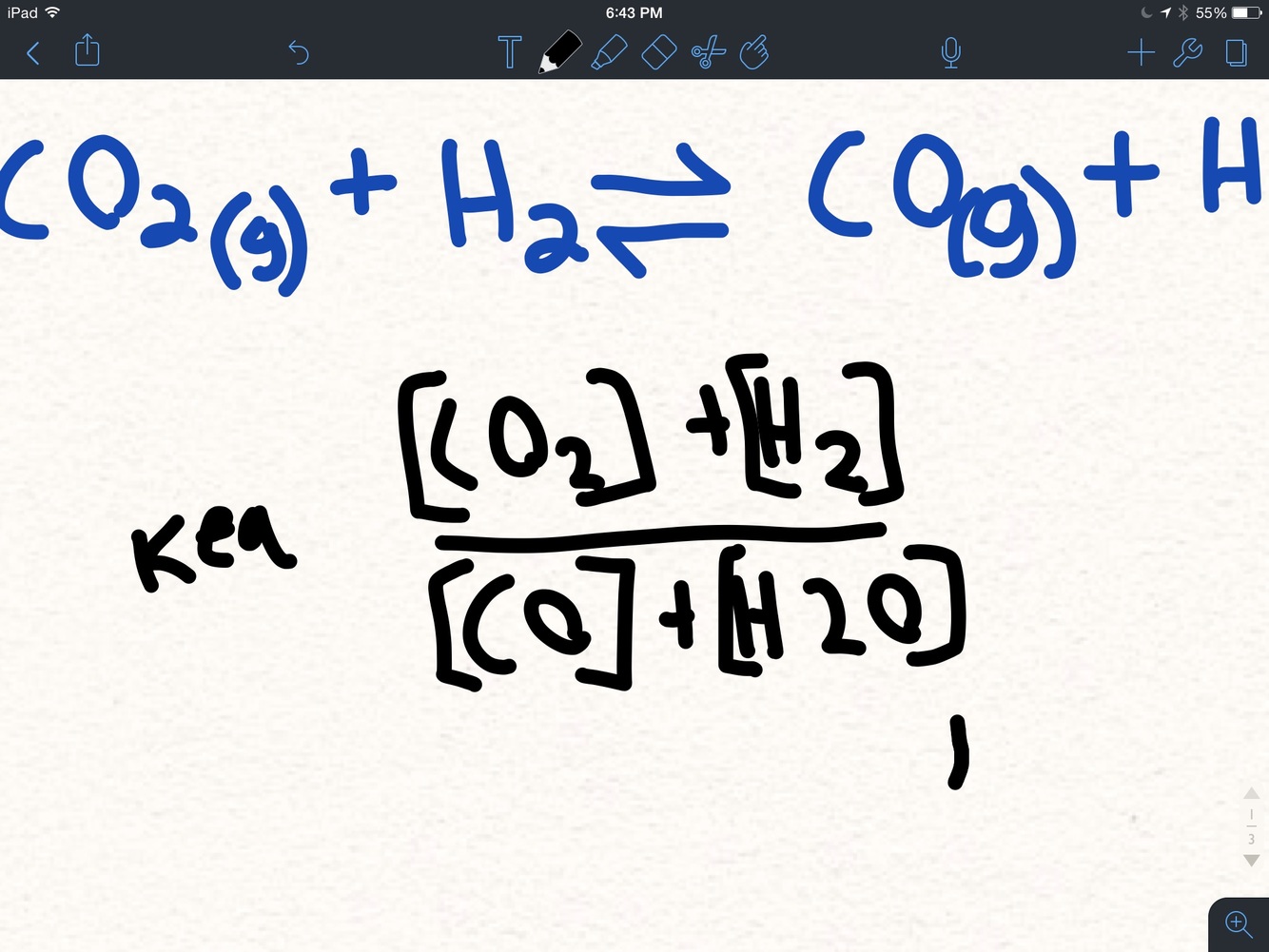
Steps for finding the equilibrium consistant
- Fill in the formula with the elements
2 cross out all solids and liquids., and add coefficients
- Calculate


