1
Q
- Which of the following is trigonal planar?
A. boron trifluoride, BF3
B. methyl anion, CH3-
C. methane, CH4
D. ammonia, NH3
A
A. boron trifluoride, BF3
2
Q
- Which of the following molecules is not linear?
A. H2O
B. CO2
C. HCºCH
D. Cl2
A
A. H2O
3
Q
- What is the approximate value of the H-C-H bond angles in methane, CH4?
A. 90
B. 109
C. 120
D. 180
A
B. 109
4
Q
- What is the approximate C-C-C bond angle in propene, CH3CH=CH2?
A. 90
B. 109
C. 120
D. 180
A
C. 120
5
Q
- What is the approximate C-C-C bond angle in propyne, HCºCCH3?
A. 90
B. 109
C. 120
D. 180
A
D. 180
6
Q
- What is the approximate H-C-O bond angle in formaldehyde, H2C=O?
A. 90
B. 109
C. 120
D. 180
A
C. 120
7
Q
- Which of the following elements has the highest electronegativity?
A. N
B. C
C. O
D. S
A
C. O
8
Q
- Which of the following elements has the highest electronegativity?
A. C
B. P
C. Si
D. Cl
A
D. Cl
9
Q
- Which of the following bonds is the most polar?
A. F-F
B. H-F
C. C-H
D. C-Si
A
B. H-F
10
Q
- Which of the following bonds is the most polar?
A. O-H
B. C-H
C. C-C
D. H-H
A
A. O-H
11
Q
- Which of the following is a polar covalent bond?
A. Na-F
B. C-H
C. C-O
D. Cl-Cl
A
C. C-O
12
Q
- Which of the following is a polar covalent bond?
A. Na-Cl
B. C-Cl
C. C-H
D. Cl-Cl
A
B. C-Cl
13
Q
- Which of the following is an ionic bond?
A. Br-Br
B. C-Cl
C. C-S
D. Na-O
A
D. Na-O
14
Q
- Which of the following is an ionic bond?
A. F-F
B. C-H
C. Li-O
D. C-N
A
C. Li-O
15
Q
- Which of the following best represents the shape of the 2s atomic orbital of carbon?
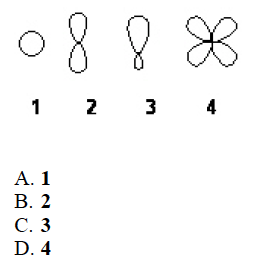
A
A. 1
16
Q
- Which of the following best represents the shape of a 2p atomic orbital of carbon?
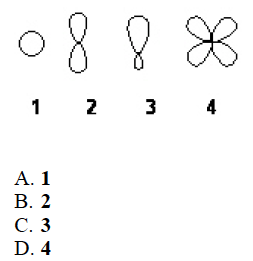
A
B. 2
17
Q
- Which of the following best represents an sp2 hybridized atomic orbital of carbon which overlaps with the 1s atomic orbital of hydrogen to form a C-H s bonding molecular orbital in ethene, H2C=CH2
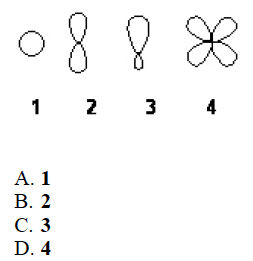
A
C. 3
18
Q
- Which of the following best represents an sp3 hybridized atomic orbital containing the lone pair of electrons of ammonia, NH3?
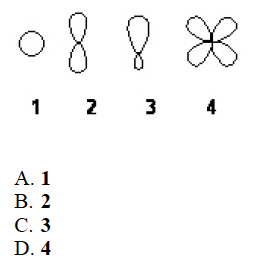
A
C. 3
19
Q
- Which atomic orbitals overlap to form the C=O bond of acetone, (CH3)2C=O?
A. C 2sp3 + O 2sp2
B. C 2sp2 + O 2p
C. C 2sp2 + O 2sp2
D. C 2sp3 + O 2sp
A
C. C 2sp2 + O 2sp2
20
Q
- Which atomic orbitals overlap to form the C-O bond of dimethyl ether, (CH3)2O?
A. C 2sp3 + O 2sp2
B. C 2sp2 + O 2p
C. C 2sp2 + O 2sp2
D. C 2sp3 + O 2sp3
A
D. C 2sp3 + O 2sp3
21
Q
- Which of the following statements is not true regarding resonance structures?
A. All resonance structures must have the same number of electrons
B. Each atom in all of the resonance structures must have a complete shell of valence electrons
C. All resonance structures must have the same arrangement of atoms
D. All resonance structures must be valid Lewis structures
A
B. Each atom in all of the resonance structures must have a complete shell of valence electrons
22
Q
- Which of the following statements is not true regarding resonance structures?
A. Each resonance structure is in rapid equilibrium with all of the other structures
B. The resonance structures may have different energies
C. All resonance structures must have the same arrangement of atoms
D. All resonance structures must have the same number of electrons
A
A. Each resonance structure is in rapid equilibrium with all of the other structures
23
Q
- Which of the following statements is not true about the carbonate anion, CO32-?
A. All of the oxygen atoms bear the same amount of charge
B. All of the carbon-oxygen bonds are the same length
C. The carbon atom bears the negative charge
D. It is basic
A
C. The carbon atom bears the negative charge
24
Q
- Which of the following statements is not true about the acetate anion, CH3CO2-?
A. The oxygen atoms bear the same amount of charge
B. The two carbon-oxygen bonds are the same length
C. The carbon atom bears the negative charge
D. It is basic
A
C. The carbon atom bears the negative charge
25
54. How many electrons are there in the valence shell of the carbon atom of a methyl cation, CH3+?
A. 4
B. 5
C. 6
D. 7
C. 6
26
55. How many electrons are there in the valence shell of the carbon atom of the methyl anion, CH3-?
A. 2
B. 4
C. 6
D. 8
D. 8
27
56. How many electrons are there in the valence shell of the oxygen atom of water?
A. 2
B. 4
C. 6
D. 8
D. 8
28
57. How many electrons are there in the valence shell of the nitrogen atom of ammonia?
A. 4
B. 5
C. 6
D. 8
D. 8
29
58. What is the approximate value of the H-C-H bond angles in a methyl cation, CH3+?
A. 90
B. 109
C. 120
D. 180
C. 120
30
59. What is the approximate value of the H-C-H bond angles in a methyl anion, CH3-?
A. 90
B. 109
C. 120
D. 180
B. 109
31
60. Which atomic orbitals overlap to form the carbon-hydrogen s bonding molecular orbitals of ethane, CH3CH3?
A. C2p + H1s
B. C2sp + H1s
C. C2sp2 + H1s
D. C2sp3 + H1s
D. C2sp3 + H1s
32
61. Which atomic orbitals overlap to form the carbon-hydrogen s bonding molecular orbitals of ethene, H2C=CH2?
A. C2p + H1s
B. C2sp + H1s
C. C2sp2 + H1s
D. C2sp3 + H1s
C. C2sp2 + H1s
33
62. Which atomic orbitals overlap to form the carbon-carbon s and p bonding molecular orbitals of ethene, H2C=CH2?
A. C2sp3 + C2sp3, and C2p + C2p
B. C2sp2 + C2sp2, and C2sp2 + C2sp2
C. C2sp2 + C2sp2, and C2p + C2p
D. C2sp3 + C2sp3, and C2sp2 + C2sp2
C. C2sp2 + C2sp2, and C2p + C2p
34
63. Which atomic orbitals overlap to form the carbon-hydrogen s bonding molecular orbitals of ethyne, HCºCH?
A. C2p + H1s
B. C2sp + H1s
C. C2sp2 + H1s
D. C2sp3 + H1s
B. C2sp + H1s
35
64. Which atomic orbitals overlap to form the carbon-carbon s molecular bonding orbital of ethyne, HCºCH?
A. C2p + C2p
B. C2sp + C2sp
C. C2sp2 + C2sp2
D. C2sp3 + C2sp3
B. C2sp + C2sp
36
73. Which of the following shows curved arrows that correctly accounts for the differences between the two structures?

C. 3
37
74. Which of the following statements is not true?
A. The sp3C-H bond of an alkane is weaker than the spC-H bond of an alkyne.
B. The carbon-carbon triple bond of an alkyne is shorter than the carbon-carbon bond of alkenes.
C. The carbon-carbon triple bond of an alkene is exactly three times as strong as a carbon-carbon single bond of an alkane.
D. The sp3C-H bond of an alkane is longer than the spC-H bond of an alkyne.
C. The carbon-carbon triple bond of an alkene is exactly three times as strong as a carbon-carbon single bond of an alkane.
38
75. Which of the following is/are tetrahedral?

B. only 1 and 3
39
1. Which of the following is the definition of a pair of enantiomers?
A. A pair of structures that are superposable mirror images of one another
B. A pair of stereoisomers that are non-superposable mirror images of one another
C. A pair of stereoisomers that are not mirror images of one another
D. A pair of stereoisomers that have equal specific rotations
B. A pair of stereoisomers that are non-superposable mirror images of one another
40
2. Which of the following is the definition of a pair of diastereomers?
A. A pair of structures that are superposable mirror images of one another
B. A pair of stereoisomers that are non-superposable mirror images of one another
C. A pair of stereoisomers that are not mirror images of one another
D. A pair of stereoisomers that have equal specific rotations
C. A pair of stereoisomers that are not mirror images of one another
41
3. Which of the following is the definition of chirality?
A. The non-superposability of an object on its mirror image
B. The superposability of an object on its mirror image
C. A molecule that has a carbon atom with four different substituents
D. A molecule with a mirror image
A. The non-superposability of an object on its mirror image
42
4. Which of the following is the definition of a meso compound?
A. A molecule with stereocenter centers which is chiral
B. A molecule with stereocenter centers which is not chiral
C. A diastereomer with no stereocenter centers
D. A chiral compound with more than one stereocenter center
B. A molecule with stereocenter centers which is not chiral
43
5. Which of the following statements is not true regarding pairs of enantiomers?
A. They have identical melting points
B. They have identical boiling points.
C. They rotate plane polarized light in opposite directions
D. They react at identical rates with chiral reagents
D. They react at identical rates with chiral reagents
44
6. Which of the following statements is true?
A. All mirror images are enantiomers
B. All molecules that have stereocenter centers are chiral
C. Isomers that are not superposable on their mirror images are enantiomers
D. Superposable structural isomers are enantiomers
C. Isomers that are not superposable on their mirror images are enantiomers
45
7. Which of the following structures is different from the other three?

D. 4
46
8. Which of the following structures is different from the other three?

C. 3
47
9. Which of the following compounds is/are chiral?

A. only 1
48
10. Which of the following compounds is/are chiral?

A. only 1
49
11. Which of the following have the S configuration?

C. only 1 and 2
50
12. Which of the following have the R configuration?

D. 1, 2 and 3
51
13. What is the configuration of the two chiral centers in the following molecule?

B. 3R,5S
52


