Chemical Bonding
- attractive forces that hold atoms together in compounds
- Three types of chemical bonding
- Ionic Bonding
- Covalent Bonding
- Metallic Bonding
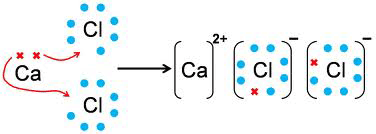
Ionic Bonds
- Also known as Electrovalent bond
- Chemical bond formed by the electrostatic attraction between positive and negative ions.
- Bonds result from transfer of valence shell electrons from one atom to another.
- Atoms that lose electrons, becomes cations
- Atoms that gain electrons, becomse anion
- purpose is to acquire a noble gas configuration.
- Favored when
- low ionization energy + large negative electron affinity
- metal element + non-metal element.
Covalent Bond
two atoms share valence electrons, which are attracted to the positively charged cores of both atoms.
Metallic Bond
The metal is held together by the strong forces of attraction between the positive nuclei and the delocalized electrons.
Octet Rule
- Most elements follow the octet rule in which the purpose of chemical bonding is to aquire a noble gas electron configuration.
- Exceptions:
- H, Li, Be follow the duet rule, they aquire the He electron configuration
- Octet rule is less applicable to the transitional elements.
Lewis Electron Dot Symbols
- a symbol in which the electrons in the valence shell of an atom or ion are represented by dots placed around the letter symbol of the element.
- Lewis developed it for covalent bonds, but can use for ionic bonds.
Lattice Energy
- the change in energy that occurs when an ionic solid is seperated into isolated ions in the gas phase.
- the formation of 1 mol of an ionic solid from its seperate ions
- measure of the strength of the crystal’s ionic bonds
Born-Haber Cycle
- Application of Hess’s law to ionic solids.
- Although the reaction happens occur all at once, it is easier to obtain energy calculations stepwise.
- Hypothetical multistep process (5 steps)
- Sublimation of sodium
- Dissociation of chlorine
- Ionization of sodium
- Formation of chloride ions
- Formation of NaCl(s) from ions
What is the energy involved in ionic bonding?
- strong forces arising from electrostatic attraction
- Coulomb’s law is used to measure the energy involved in the interaction of electric charges.
- Formula: E = (kQ1Q2)/r
- where: r = distance
- k = constant: 8.99 x 109 J•m/C2
- Q1 = charge on one element
- Q2 = charge on second element
What are the properties of Ionic Substances?
- solids with high melting points, the higher the charge the higher the melting point.
- molten compounds and aqueous solutions conduct electricity well because they contain mobile charged particles.
- are soluble in polar solvents. i.e. water.
Electron configuration for an anion.
- add electrons to the valence shell of the neutral nonmetal atom without adding protons or neutrons to the nucleus.
- number of electrons gain, is usually enough to complete the valence shell of the atom
Electron configuration for a cation
- a metal atom loses one or more electrons in forming a cation.
- the “p” valence electrons (if they are any) are lost first and then “s” valence electrons in some cases, “d” electrons of the next two outermost shell follow.
Electron configuration for Transitional metal ions
- can form several cations of different charges
- first electrons lost are in the ‘s’ then one or more in the (n-1)d electrons. most have the +2 charge because of loss of ‘s’ electrons.
- colors of the transitional element ions are due to the ‘d’ electrons.
Ionic Radii
- measure of the size of the spherical region around the nucleus of an ion within which elecrons are most likely to be found.
- Radii can be obtained by crystal structure studies or x-ray diffraction.
- Cations are smaller than the atoms from which they are formed because of the loss of valence shell
- Anions are larger than the atoms from which they are formed because of the gain of electrons.
- Generally decrease in radii across the period and increase down the group.
Isoelectronic
- refers to different species having the same number and configuration of electrons. e.g. Ne, Na+ and Mg2+ have the same configuration of electrons.
- for a series of isoelectronic species with the same electron configuration, the greater the charge, the smaller the species.
Lewis Electron Dot Formula
Using dots to represent valence electrons
Bonding Pair
Shared pair of electrons, can be represented by a dash.
Lone Pair
Non-bonding pair, not shared, stays in one atom
Lewis Formulas
- Purpose of sharing is to aquire a complete octet or noble gas configuration.
- Non-metals of the second period, except boron, form a number of covalent bond equal to eight minus the group number
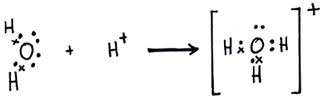
Coordinate Covalent Bonds
- a bond formed when both electrons of the bond are donated by one atom
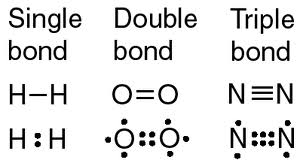
Multiple Bonds
- Single-bond:
- a covalent bond in which a single pair of electrons is shared by two atoms
- Double-Bond:
- covalent bond in which two pairs of electrons is shared by two atoms
- Triple-Bond
- covalent bond in which three pairs of electrons is shared by two atoms
Compare ionic, polar and non-polar bonding.
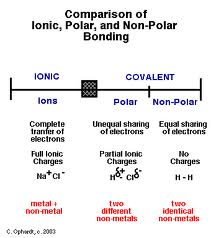
Polar covalent bond
- electrons are drawn closer to one of the atoms, the one that is more electronegative. Electrons are not shared equally.
- Unsymmetrical distribution of electrons leads to a partial negative charge on one end and a partial positive at the other end. the whole molecule is still neutral
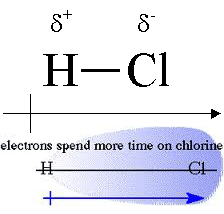
Electronegativity
- Measure of the ability of an atom in a molecule to attact the shared bonding electrons to itself in a covalent bond. it is related to ionization energy and electron affinity.
- Robert Mullik scale
- X = ((ionization energy)-(Electron affinity))/2
- Linus Pauling’s scale
- fluorine is the most electronegative with a value of 4
- Electonegativity increases across the period, electronegativity decreases down a group.




