What is AMI?
Acute Myocardial Infarction: macroscopic death of myocardium due to vascular insufficiency
What is VI?
Vascular Insufficiency: myocardial ischaemia i.e. balance between supply/flow (perfusion) and demand for oxygenated blood
What percentage of deaths are related to AMI?
80% of cardiac related mortality and 18% of all deaths in australia
What is the most common cause of death in industrialised countries?
AMI
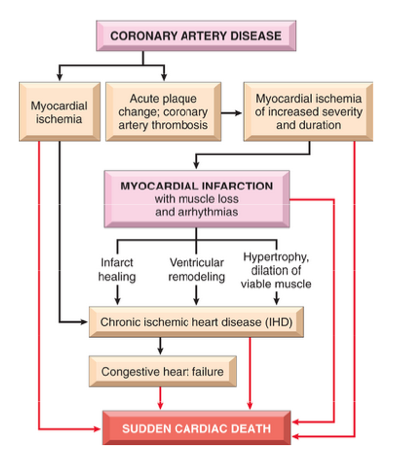
What are some of the various pathways in the progression of ischemic heart disease?
- atherosclerosis is one of the major causes
- hypertrophy/dilation of viable muscle is a common feature of someone’s heart with blockages in the coronary arteries
What is the major underlying cause of AMI?
Atherosclerosis is an underlying cause in 95% of cases. It is a disease caused by fibrofatty plaques in intima of large arteries.
Other causes include spasm, drugs and trauma
What are some risk factors associated with AMI?
- risk of AMI increases with age (10% <40 yrs, 45%<65 years)
- men at greater risk than women
- hypertension, diabetes, smoking, hypercholesterolaemia
- major lifestyle disease
- some genetic factors could predispose people
What sequence of events in the typical case of MI is considered most likely?
- The initial event is a sudden change in an atheromatous plaque, which may consist of intraplaque haemorrage, erosion or ulceration, or rupture or fissuring. This plaque is caused by a build up of cholesterol and may take decades to form.
- When exposed to subendothelial collagen and necrotic plaque contents, platelets adhere, become activated, release their granule contents, and aggregate to form microthrombi
- Vasospasm is stimulated by mediators released from platelets (blood clotting factors)
- Tissue factor activates the coagulation pathway, adding to the bulk of the thrombus (viscious cycle)
- Frequently within minutes, the thrombus evolves to completely occlude the lumen of the vessel (rapid)
On what do the presice location, size, and specific morphological features of an AMI depend?
- The location, severity, and rate of development of coronary obstructions due to atherosclerosis and thromboses
- The size of the vascular bed perfused by the obstructed vessels
- The duration of the occlusion
- The metabolic/oxygen needs of the myocardium at risk
- The extent of collateral blood vessels
- The presence, site, and severity of coronary arterial spasm
- Other factors, such as heart rate, cardiac rhythm, and blood oxygenation
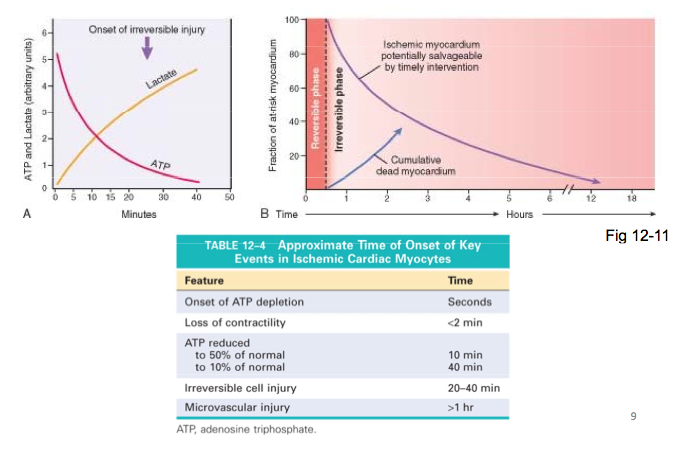
What are the approximate onset times of key events in ischemic cardiac myocytes?
What is atherosclerosis?
A chronic inflammatory and healing response of the arterial wall to endothelial injury. Lesion progression occurs through the interaction of modified lipoproteins, monocyte-derived macrophages, and T lymphocytes with the normal cellular constituents of the arterial wall.
What pathogenic events produce atherosclerosis according to the previously stated model?
- endothelial injury (damage to inner lining of arteries) causes, among other things, increased vascular permeability, leukocyte adhesion, and thrombosis. Only slight damage needed.
- accumulation of lipoproteins (mainly LDL and its oxidised forms) in the vessel wall, often slow, building up over time.
- monocyte adhesion to the endothelium, followed by migration into the intima and transformation into macrophages and foam cells. (responding to injured cell region and causing local inflammation.)
- platelet adhesion - normally only occurs when there is haemorrhage/cut etc
- factor release from activated platelets, macrophages, and vascular wall cells, inducing smooth muscle cell recruitment, either from the media or from circulating precursors (release a lot of insoluble factors that build up)
- smooth muscle cell proliferation and ECM production (layer inside artery begins to thicken, therefore become less flexible.)
- Lipid accumulation: both extracellular and within cells (macrophages and smooth muscle cells)
How does the distribution of myocardial ischemic necrosis correlate with the location and nature of decreased perfusion?
Transmural infarcts will occur with permanent occlusion of an arterial branch whereas non-transmural infarcts will occur with transient/partial obstruction.
Whatever area of heart the occluded vessel normally supplies is the area that will suffer the infarct e.g. left anterior descending branch > anterior wall of LV.
Small intramural vessel occulusions > microinfarcts.
Global hyptoension >> circumferential subendocardial infarct.
How do coronary arteries change over time?
Infant artery: smooth endothelial layer, smooth muscle layer, completely open blood vessel with easy flow.
Adult artery: as people age it’s natural that the heart muscle should thicken: hpb will thicken arteries quicker as they try to compensate. The wall of the artery becomes much thicker.
How is plaque stability an issue?
A stable plaque can still allow blood flow. However if this stability is altered through rupture, erosion, ulceration, haemorrhage the internal contents of the plaque will leak out, initiating an immune response and platelet activity that will essentially cause a thrombus.
What is/are the natural history, morphological features, main pathogenic events and clinical complications of atherosclerosis?
A normal artery may develop a fatty streak which can develop into a fibrofatty plaque at lesion-prone areas, accelerated by such risk factors as endothelial dysfunction, monocyte adhesion/emigration, smooth muscle cell migration to intima, smooth muscle cell proliferation, ECM elaboration and Lipid accumulation. This fibrofatty plaque can become an advanced/vulnerable plaque after such events as cell death/degeneration, inflammation, plaque growth, remodeling of plauq and wall ECM, organisation of thrombus or calcification.
From here it transforms into the clinical stage (usually middle age to elderly).
- Mural thrombosis, embolisation and wall weakening can lead to aneurysm and rupture
- Plaque rupture, plaque erosion, plaque hemorrhage, mural thrombosis and emobolisation can lead to occlusion by the thrombus
- Progressive plaque growth can lead to critical stenosis (the abnormal narrowing of a passage in the body.)
What are the major components of a well-developed intimal atheromatous plaque?
- There is an evolution of cells involved as well as general factors.
- General factors: cholesterol (LDL)
- Recruitment of smooth muscle cell precursors (abnormally, by release of factors)
- These then infiltrate and embed themselves and begin to grow
- Proliferation of smooth muscle cells
- migration of smooth muscle cells to where they wouldn’t normally be
- Recruitment of monocytes: differentiate into macrophages which try to eat up the abnormal stuff but the inflammatory response becomes so severe that the macrophages themselves seem to contribute negatively
Describe the structure of an artery (basics)
- Lumen
- Endothelium
- Intima
- Internal elastic membrane
- Media
This structure is completely lost in the area with the damaged plaque
Describe the progression of myocardial necrosis after coronary artery occlusion.
Necrosis begins in a small zone of the myocardium beneath the endocardial surface in the center of the ischemic zone. The area that depends on the occluded vessel for perfusion is the “at risk” myocardium.
Irreversible injury first occurs in the sub-endocardial zone and extends/progresses transmurally.
Location and size depend on the location/severity of the plaque, duration of occlusion, collateral circulation and the needs of the myocardium.
Describe the pathogenisis of AMI at the cellular level.
- Apoptosis - occurs rapidly post-ischaemia (apoptosis inhibitors reduce infarct size)
- Reperfusion - in <20 min myocytes survive infarct event
- No reperfusion - necrosis complete in 6-12 hr
How do you recognise a heart attack in a biopsy of a tissue?
- a loss of regular heart muscle striation
- inclusion of a lot of monocytes: beginnings of an acute inflammatory response
Describe the evolution of an infarct site in a surviving patient.
- Day 1: some normal tissue with adjacently damaged tissue, loss of regular heart muscle strands, infiltration of white blood cells (in particular neutrophils)
- Day 3-4: mass migration of neutrophils disrupting regular structure
- Day 7-10: massive amount of necrosis, heart muscle cellular tissue lost almost completely, still lots of WBCs
- Day ~21: Tentative repair of muscle tissue, lots of collagen deposition from fribroplast cells (creating scar tissue… not good)
- Day >60: Scar tissue remains in the heart: not recoverable
What can be done to help a patient? Can the infarct be modified?
- Restoration of coronary blood flow
- Thrombolysis: streptokinase or tissue plasminogen activator activates fibronolytic enzyme systems that dissolve thrombus
- Balloon angioplasty (percutaneous transluminal coronary angioplasty PTCA)
- Coronary arterial bypass graft (CABG)
What are the inflammatory markers of AMI?
At the onset of myocardial infarction the plasma membrane of necrotic myocytes becomes leaky. Some molecules leak out of the cell into circulation. These molecules can be used as biomarkers for diagnosis of myocardial infarction.
Some of these proteins include troponin I, C, or T, and creatine phosophokinase, MB fraction (CK-MB).


