1
Q
you look new around here, let me show you the ropes
A
The powerpoint slides will be prefaced with E Rt R C O M N
E -> word Equation
Rt -> Reaction type
R -> Reagents
C -> Conditions
O -> Observations
M -> Mechanism
N -> Notes
2
Q
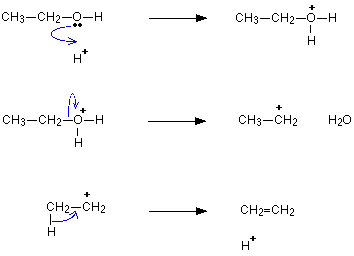
Give the E Rt R C O M N of the dehydration of alcohols
A
E: alcohol -> alkene + water
Rt: Elimination
R: Concentrated H2SO4 or Al2O3
C: Heat under reflux at 180°C
O: -
M:Elimination
3
Q
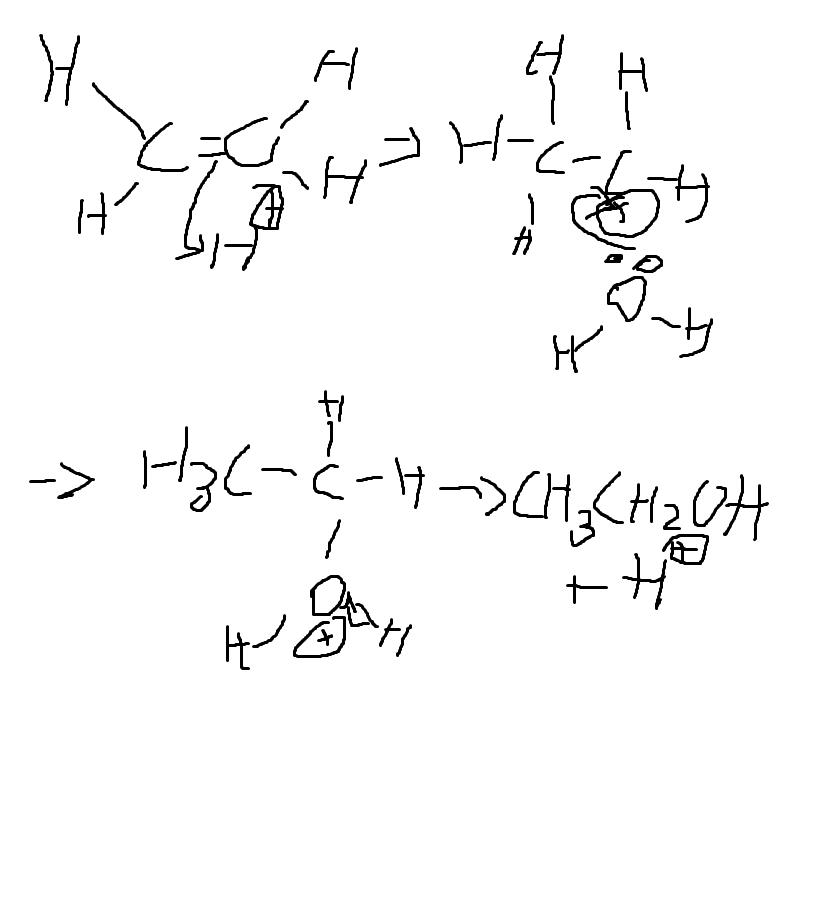
Give the E Rt R C O M of the hydration of alkenes
A
E: water + alkene -> alcohol
Rt: addition/hydration
R: steam, concentrated H3PO4 catalyst
C: concentrated H3PO4 catalyst, 60atm, 300°
O:-
M: Electrophilic addition
4
Q
Give the C of the substitution of NH3 onto a haloalkane
A
- Concentrated NH3
- Heat in a sealed tube
- High pressure


