emission spectrum
- expose a container of gas at low pressure to a strong electric field
- light emitted from gas
- light analysed by passing it through a prism or diffraction grating
- definition: set of possible wavelengths that can be emitted by a gas
discrete energy
energy can have one of a specific set of values
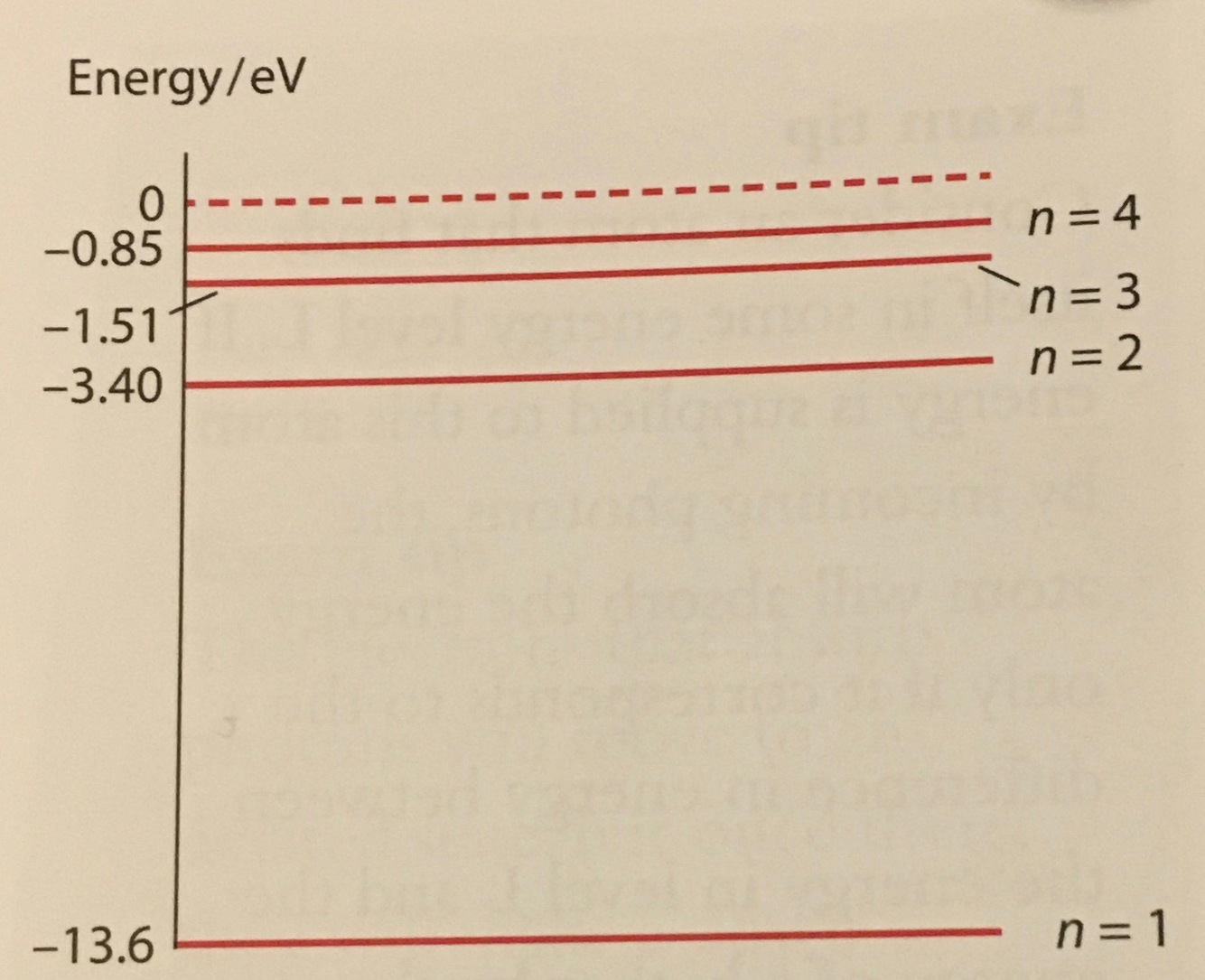
energy level diagram
each horizontal value represents a possible energy of the atom
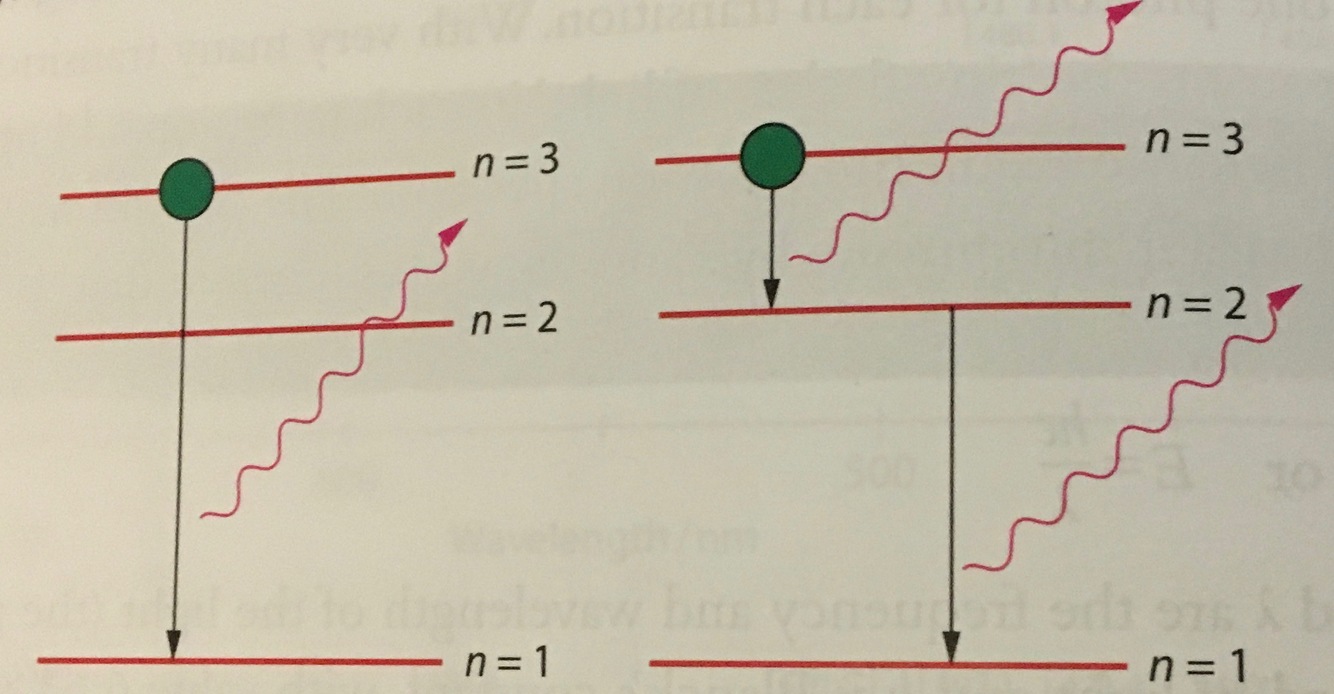
how to explain the emission spectrum?
- atom can make a transition from a state of higher energy to a state of lower energy by emitting a photon
- the energy of the emitted photon is the difference in energy between the two levels
ground state
lowest energy state
excited state
- if energy is supplied to the atom, the electron may move to a higher energy level by absorbing the right amount of energy exactly to move up
- electron immediately makes transition down to lower energy (relaxation)
hydrogen energy level diagrams for all possible transitions from n=3
whether the electron will make a direct or indirect transition is just probability
how is the absorption spectrum produced?
- beam of white light through gas
- majority of atoms in ground state
- electrons may absorb photons in beam and become excited
- only happens if photon that is absorbed has the exact right energy that corresponds to difference in levels
- light that is transmitted through gas will be missing photons - corresponds to dark lines
why are the photons in an absorption spectrum missing?
photons emitted in all directions, not necessarily along the direction of the observer
nucleon
proton or neutron
nuclide
nucleus with specific number of protons and neutrons
isotopes
nuclei that have the same number of protons but a different number of neutrons
unstable nucleus
nucleus that randomly and spontaneously emits particles that carry energy away from nucleus
radioactivity
emission of particles and energy from a nucleus
alpha decay
alpha particle is emitted from the nucleus and the decaying nucleus turns into a different nucleus
beta minus decay
neutron in the decaying nucleus turns into a proton, emitting an electron and an anti-neutrino
alpha particle
helium nucleus
beta minus particle
electron
beta plus decay
nucleus emits positron and neutrino
gamma decay
nucleus emits a gamma ray
gamma particle
photon of high-frequency electromagnetic radiation
penetrative power of alpha, beta minus, gamma particles
- alpha is least penetrating
- beta minus has less charge and travels faster so interacts less with environment so more penetrative
- gamma has no ionising power bc no charge so most penetrative
ionising power of alpha, beta minus and gamma
- alpha has a lot of momentum and double charge so a lot of interaction
- beta minus has less momentum and less charge so less interaction
- gamma is not very ionising - depends on intensity
decay series
set of decays that takes place until a given nucleus ends up as a stable nucleus





