How do nucleophiles attack a carbon atom?
have a lone pair of electrons which they use to attack an electron-deficient carbon
What makes a good nucleophile?
best nucleophiles are the ones that are best at donating their lone pair
List the nucleophiles that react with acid chlorides and acid anhydrides in order of reactivity
- primary ammine
- ammonia
- alcohol
- water
What type of reaction is it the an acyl compound?
- addition-elimination
What do the nucleophiles loose in the process?
a hydrogen ion because all atoms are neutral
one way to look at this is that the hydorgen atoms of the nucleophile has been replaced by an acyl group
What is the product of the addition of ammonia?
an amide
WHat is the product of the addition of a primary amine?
N-substituted amide
What is the product of the addition of water and alcohol?
water - carboxylic acid
alcohol -OH group - ester
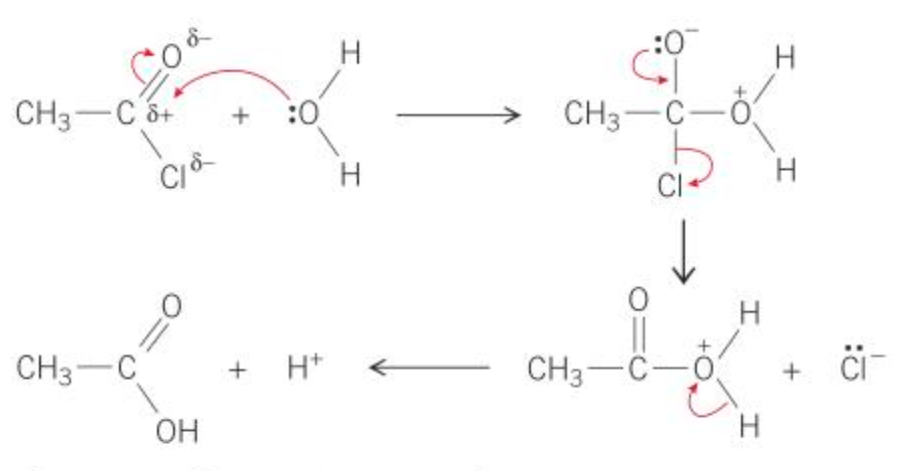
Draw the mechanism for this reaction for ethanoyl chloride and water
the mechanism of these reactions follow the same pattern
Why is ethanoic anhydride used over ethanoyl chloride?
- cheaper
- less corrosive
- does not react with water as readily
- safter, by-product is ethanoic acid rather than HCl
What are the uses of ethanoic anhydride?
- production of asprin
Why is asprin described as ‘if it were a new drug today it would be perscription only’?
has side effects
e.g. intestinal bleeding
WHat is asprin used for?
- moderate pain
- fever
- shown to also reduce risk of some cancers an d heart attacks


