What were Lenard’s Observations?
- The current is directly proportional to the light intensity.
- The current appears without delay.
- Photoelectrons are emitted only if the light frequency exceeds a threshold frequency.
- The value of the threshold frequency depends on the type of metal from which the cathode is made.
- If the potential difference is more than about 1V positive the current does not change as V is increased. If V is made negative them the current decreases until V = stopping potential.
- The value of the stopping potential is the same for both weak and intense light.
How does a voltage across the plates effect the current?
- If V=0 : The photoelectrons leave the cathode in all directions. Only a few reach the anode.
- If V>0 : A positive anode attracts all the photoelectrons to the anode.
- If V<0 : A negative anode repels the electons, only the fastest make it to the anode.
What were Einstein’s postulates?
- Light of frequency f consists of discrete quanta, each of energy hf
- Light can only be emitted or absorbed in these discrete quanta.
- When absorbed by metal, each light quantum delivers its whole energy to one electron
What is the proof for the de Broglie Wavelength?
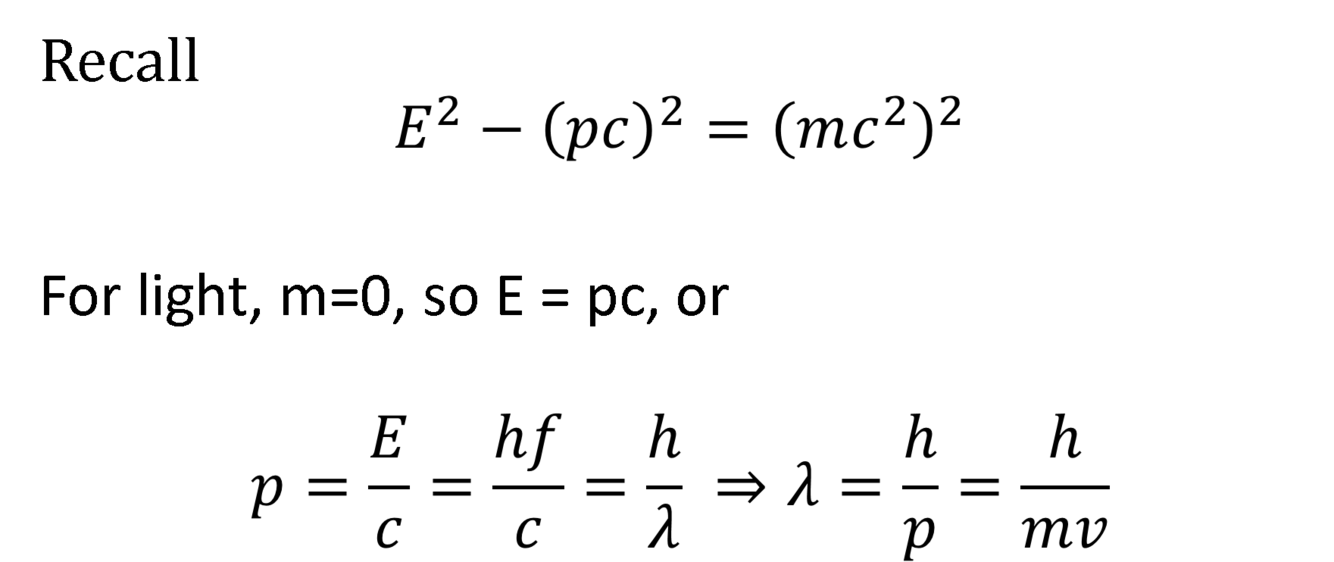
What equation describes the diffraction pattern of electrons through a pair of slits?
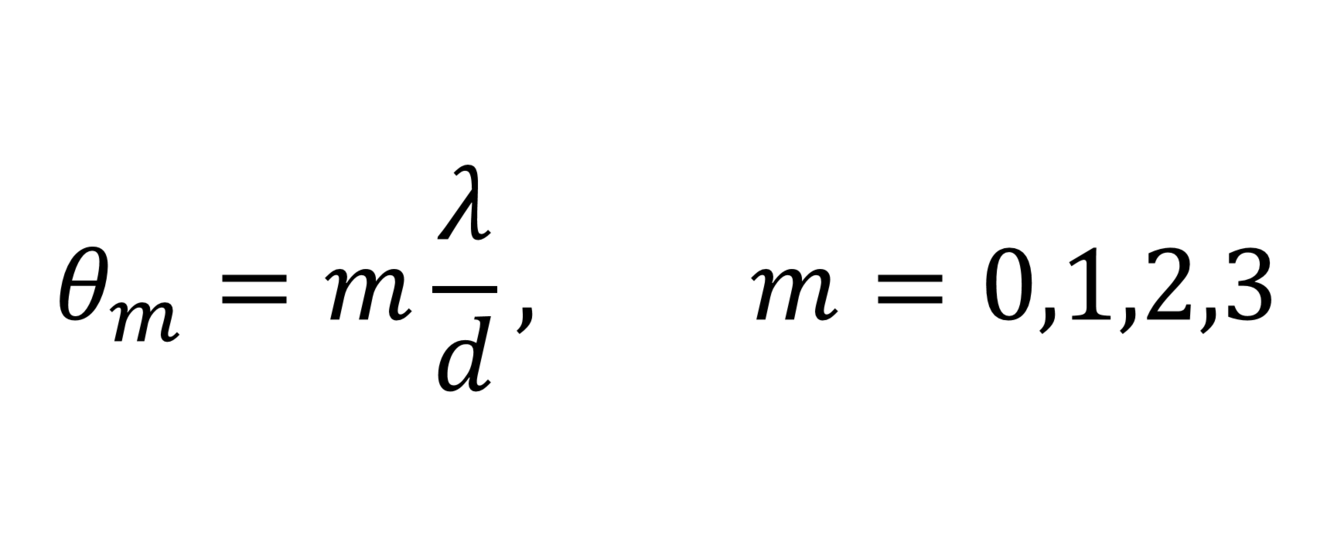
Show how energy is quantized when modelled as a particle in a box.
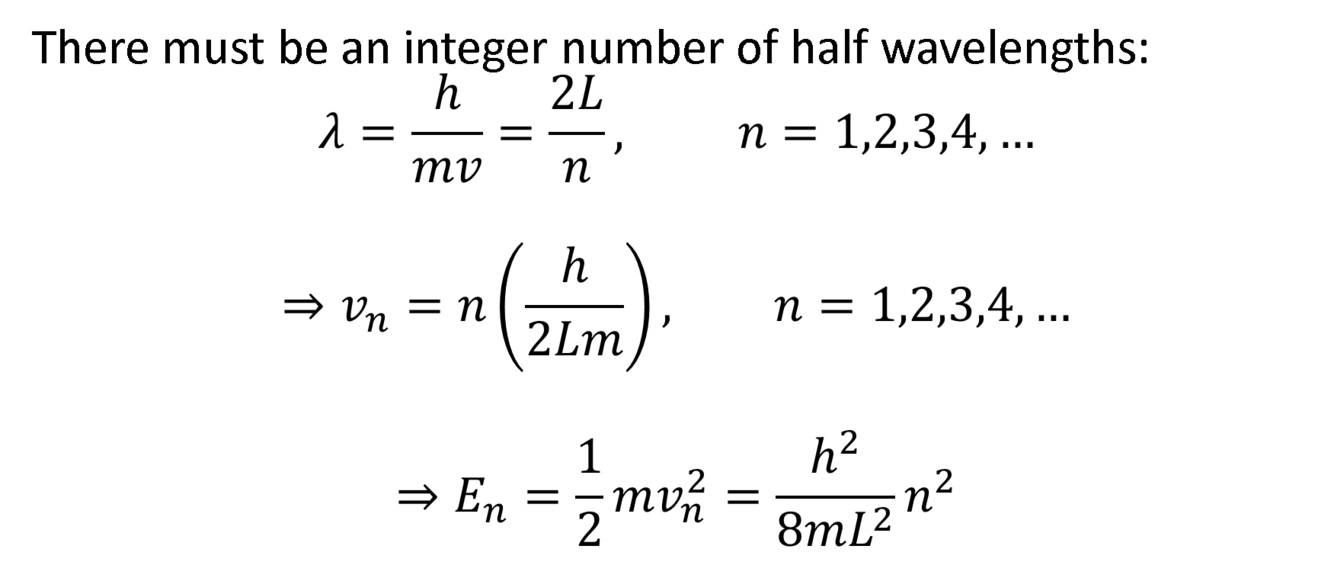
What causes absorbtion spectra?
The quantization of electrons means that electrons can only be at certain discrete energy levels.
This means only certain energies of photons can be absorbed.
What causes emission spectra?
Energy is emitted as light when an electron changes energy level, this can only happen at discrete energies.
Why do absorption spectras have less lines than emission spectras?
Usually, atoms exist in the lowest energy state, so that absorption only occurs from the lowest energy ground state whereas there are many possible energy states before emission.
Describe Bohr’s model of an atom.
- Negative electrons orbit a small positive nucleus
- Electrons can only occupy certain orbits, which can be labelled with quantum numbers, n.
- Each orbit has its own energy, E_n.
- Atoms jump from one state to another by absorbing or emitting a photon with an energy E = Ef-Ei
- Atoms can also change state through inelastic collisions with particles, or collisional excitation
- Left alone, an atom will decay to the lowest energy ground state.
How do you find the Bohr velocities?

How do you use the bohr velocity and the centripetal force to solve for the bohr radius and the bohr velocity?
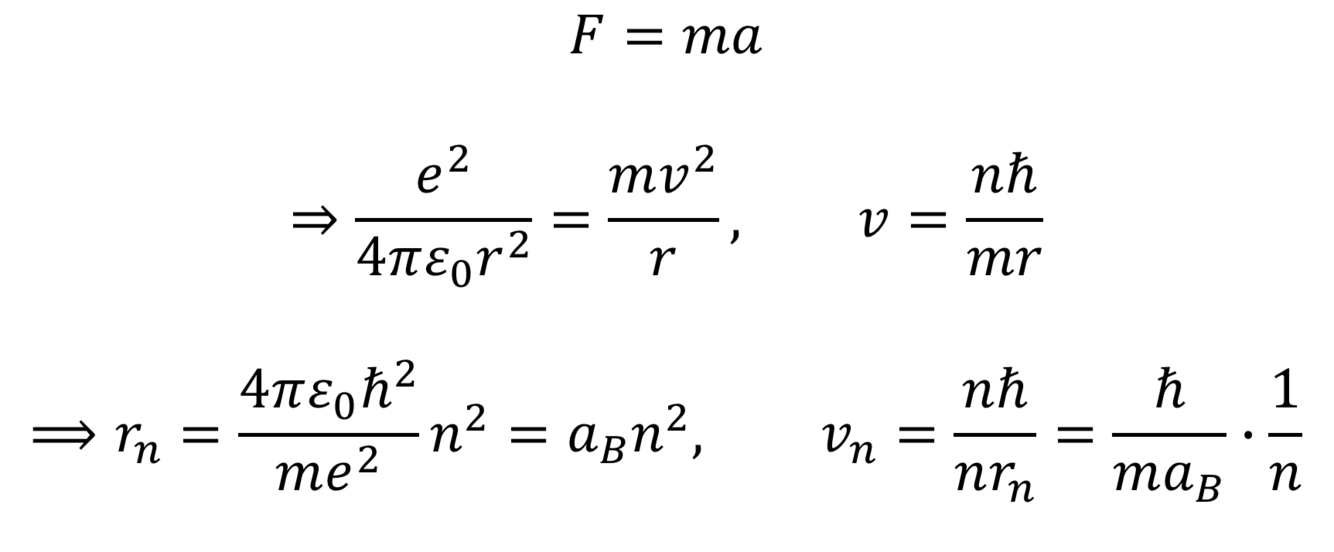
How do you find the Bohr energies of an electron?
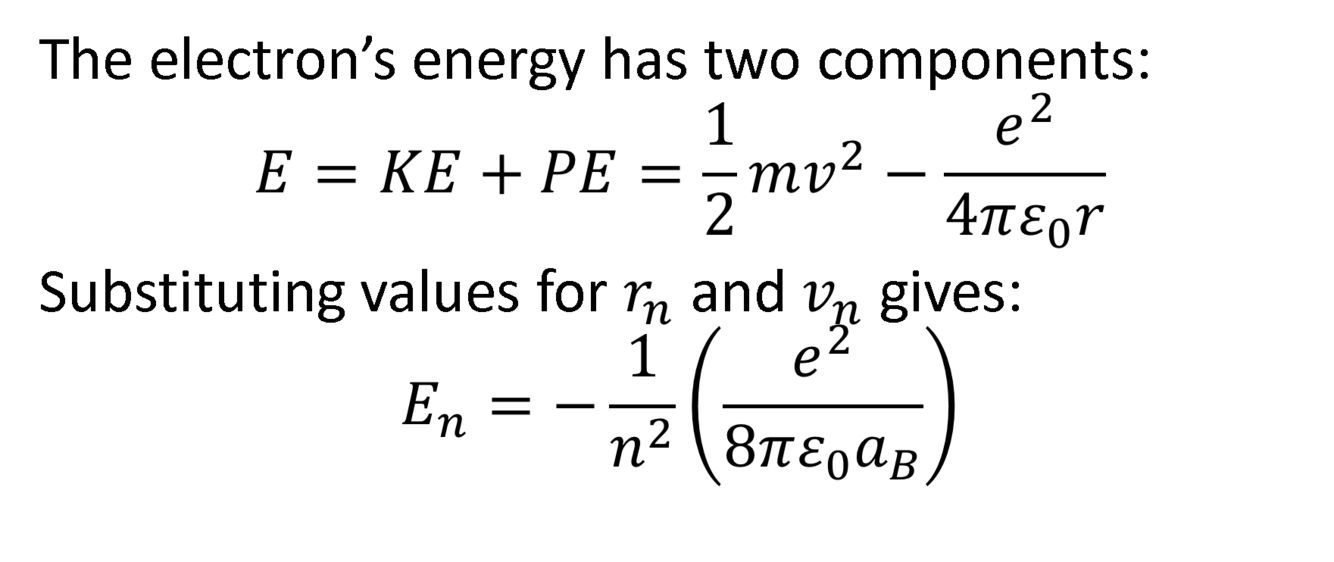
What are the limitations of the Bohr model?
Too difficult to solve when there’s more than one electron.
Explain or disprove the statement using the classical interpretation:
“The current is directly proportional to the light intensity”
Increased amount of heating will increase the flow of current.
Explain or disprove the statement using the classical interpretation:
The current appears immediately when light turns on
Doesn’t explain.
Thermal emission takes some time to heat up the electrons to sufficient energy.
Explain or disprove the statement using the classical interpretation:
“Current only flows if the light frequency exceeds a threshold frequency”
Can’t explain.
A low frequency but high intensity light could provide enough thermal energy to the electron to escape.
Explain or disprove the statement using the classical interpretation:
“The value of the threshold frequency depends on the type of metal from which the cathode is made”
Threshold frequency cannot be explained.
Explain or disprove the statement using the classical interpretation:
If the potential difference is positivem the current does not change as V is increased. If V is made negative, the current decreases until, at V= the stopping potential the current reaches zero.
This would just operate as normal attracting or repelling escaped electrons.
Explain or disprove the statement using the classical interpretation:
The value of the stopping potential is the same for both weak and intense light.
Can’t explain.
More intense light would increase the electrons kinetic energy thus increasing the stopping potential.
Explain or disprove the statement using Einstein’s interpretation.
“The current is directly proportional to the light intensity”
A more intense light means more quanta of the same energy. These quanta eject a larger number of photoelectrons and cause a larger current.
Explain or disprove the statement using Einstein’s interpretation.
“The current appears immediately when light turns on”
Each light quantum transfers its energy to just one electron, that electron immediately has enough energy to escape.
Explain or disprove the statement using Einstein’s interpretation.
“Current only flows if the light frequency exceeds a threshold frequency.”
An electron that has just absorbed a quantum of light energy has energy hf.
The work function E_0 is the minimum energy required to free an electron.
f0=E0/h
Explain or disprove the statement using Einstein’s interpretation.
“The value of the threshold frequency depends on the type of metal from which the cathode is made.”
All metals are different.


