Why do only very few drugs contain furan?
Furan (5 membered “O” heterocycle) undergoes CYP mediated metabolism into BDA
BDA can react with variety of endogenous molecules including 2′-deoxycytidine, 2′-deoxyadenosine, and 2′-deoxyguanosine in DNA - potential genotoxicity of drugs containing furan
Lipinski’s Rules
- MW > 500
- logP >5
- > 5 H-bond donors (expressed as the sum of OH and NH groups)
- > 10 H-bond acceptors (expressed as the sum of N and O atoms. Note: It is a very simplified rule. This approach to counting does not always predict the actual number of h-bond acceptors correctly)
- Antibiotics, antifungals, vitamins, and cardiac glycosides are the exception because they often have active transporters to carry them across membranes.
It is highly likely (>90%) that compounds with two or more of these characteristics will have poor absorption properties.
DESIRED TPSA to be able to cross CELL MEMBRANE
TPSA < 140 Angstroms
LESS THAN 140
DESIRED TPSA to be able to cross BBB
TPSA < 90 Angstroms
LESS THAN 90
Huckel’s Rule
4n + 2pi
= 2,6,10,14,18 pi electrons
to be AROMATIC
Heteroatoms in order of Polarity
N is most POLAR
N > O > S
Heteroatoms in order of Lipophilicity
S > O > N
S is most LIPOPHILIC
Increasing logP of the heterocycle that contains the atom
Ex.
Thiophene (S) logP = 1.79 > Pyrrole (N) logP = 0.75
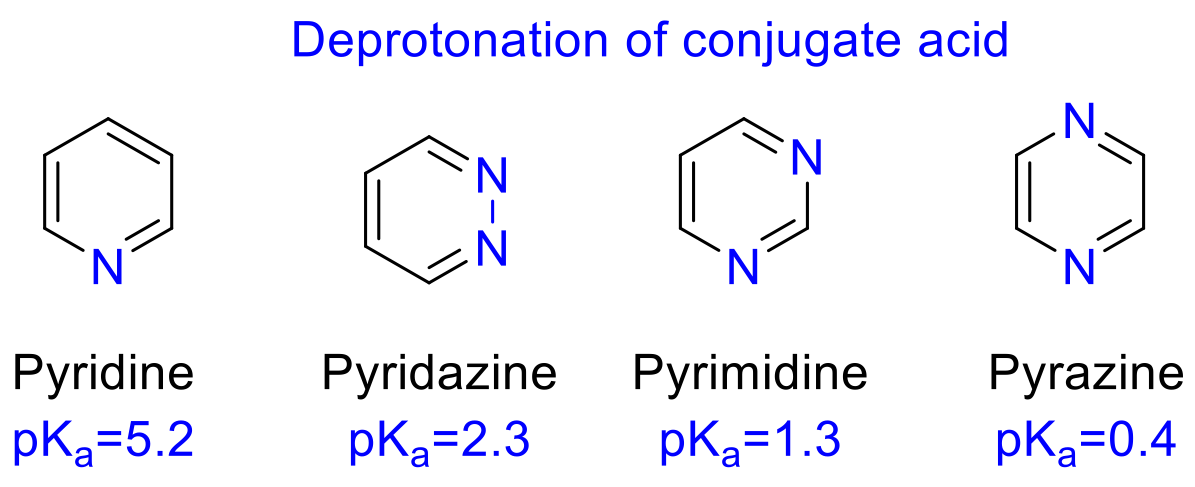
pKa trend for basic aromatic heterocycles
More Nitrogens = Less Basic ( lower pKa )
logP
logP = octanol / H2O
Drugs & Bioactive molecules - logP= 1-5
logP=5 >>> 105 parts octanol : 1 part H2O
Saturated 5-mem heterocycles
Structural Properties
Puckered Conformation
NOT FLAT
ex. pyrrolidine
5-membered AROMATIC heterocycles
Structural properties
ALL FLAT
ex. Pyrrole (pka 1-5, weak base)
O/S hetrocycles not considered Acid or base
Unsaturated 5-membered heterocycles
Structural Properties
ALL FLAT
Beta-Lactams MOA
- β-Lactam antibiotics inhibit DD-transpeptidase
- β-Lactam Covalently Bonds to the ester on DDTP
- Irreversible reaction
Factors Affecting Beta Lactam REACTIVITY
- MORE pyramidal > MORE REACTIVE
- free pair of electrons is conjugated w/ pi electron of C=O bond
- MORE orbitals of N are sp3 hybridized > MORE reactive
- reduces partial double bond character
- WEAKER the C-N bond > More reactive
- LOWER partial double bond character > More Reactive
Effect of Reactivity of Beta Lactam Antibiotics
Very Reactive - FAST antibiotic effect, ~fast degradation
Moderately reactive - Extended effect, ~slow degradation
Low Reactivity - may NOT inhibit DDTP
instead used for inhibition of Beta-lactamase (cleaves B-lactam)
Responsible for ANTIBIOTIC RESISTANCE
3-mem heterocycle DRUGS
All Reactive due to Ring Strain (rxn w/ nu-)
Steric hinderence = unusually stable
Aziridine Rings –> DNA alkylating agents
VERY REACTIVE, not very selective can hurt your DNA
O-containing 3Rings
NOT ALL are reactive, have to be accessible
S-containing 3rings = No FDA approved drugs
4-mem heterocycle DRUGS
O-containing 4rings = Not all Reactive
Oxetane = NOT REACTIVE
Lactone = REACTIVE
S-containing 4rings = No FDA approved drugs
3/4-membered heterocycles
Structural Properties
- Very RIGID
- The 3- and 4-membered saturated heterocycles ARE NOT FLAT as they are made from sp3 hybridized atoms. This may improve dissolution and solubility.
- 3-membered saturated heterocycles have only one conformer
- 4-membered saturated heterocycles have two “envelop” conformers
3/4-membered heterocycles
effect on DRUG PROPERTIES
Off Target Reactivity > Adverse Rxns & Shortening duration of Action
ex. Alkylating agents > tissue dmg
Beta Lactams > form haptens > Immunogenic response
Chemical Instability > Storage/Formulation/Routes of Administration
Electronic Properties of 6mem Heterocycles
- Aromatic/unsaturated 6mem heterocycles = ALL FLAT
- 6mem Aromatic N-heterocycles = weakly basic
- pKa ~-1 - 6
- 6mem S-heterocycles do not have acidic or basic properties
Factors that help diffusion through a cell membrane
To INCREASE diffusion / slipperiness (k):
HIGHER = logP or logD
HIGHER = HSA
less = H-bond acceptors & donors
lower = MW
lower = SASA / TPSA
SASA
SASA = surface area of a molecule accessible to a solvent (WATER)
INVERSELY RELATED TO PERMEABILITY
Decreasing SASA > MORE PERMEABLE (less water solubility)
The Higher the density of the packing (pi-pi stacking / sp2 vdw interactions)
MORE energy required to disrupt interactions between molecules to achieve dissolution & solubility
MORE difficult for other molecules to get IN BETWEEN
TPSA (Topological polar surface area)
TPSA = surface area of all the polar parts of a molecule (mainly N,O)
INVERSELY RELATED TO PERMEABILITY
DECREASING TPSA > INCREASES SOLUBILITY





