What is the rate of reaction?

What are the different ways to measure the rate of reaction experimentally?
- Measuring change in electrical conductivity (if there is a change in the number of ions)
- Amount of mass lost (for when a gas is produced)
- Volume of gas produced with a gas syringe (whena. gas is produced)
- Change in pH (when there’s a change in H+)
- Titration (taking aliquots(small samples) )
- Colourimeter (Use calibration curve to know which %absorbance corresponds to a specific concentration)
How do you measure the rate of reaction with titrations?
Small samples (aliquots) are taken from the reaction mixture at specific time interval.
Before titration aliquotes are quenched to try and stop the reaction. This is done by dilution, cooling it down and even using a quenching agent.
Then titrations are carried out to find the concentration at certain times.
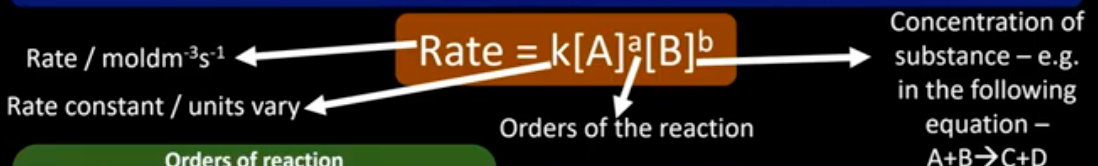
What is the rate equation?
Wha is the order of reaction?

What does it means if a reactant is zero order with respect to a reaction?

What does it means if a reactant is first order with respect to a reaction?

What does it means if a reactant is second order with respect to a reaction?

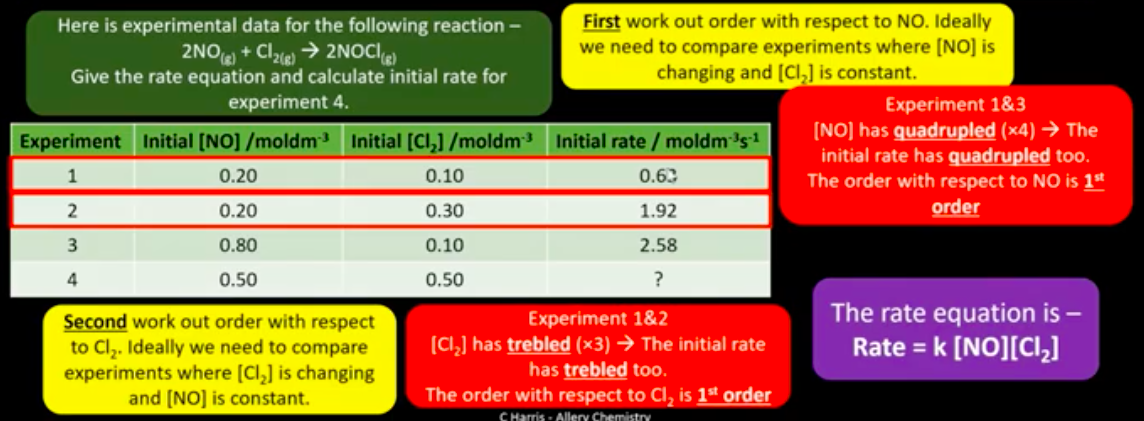
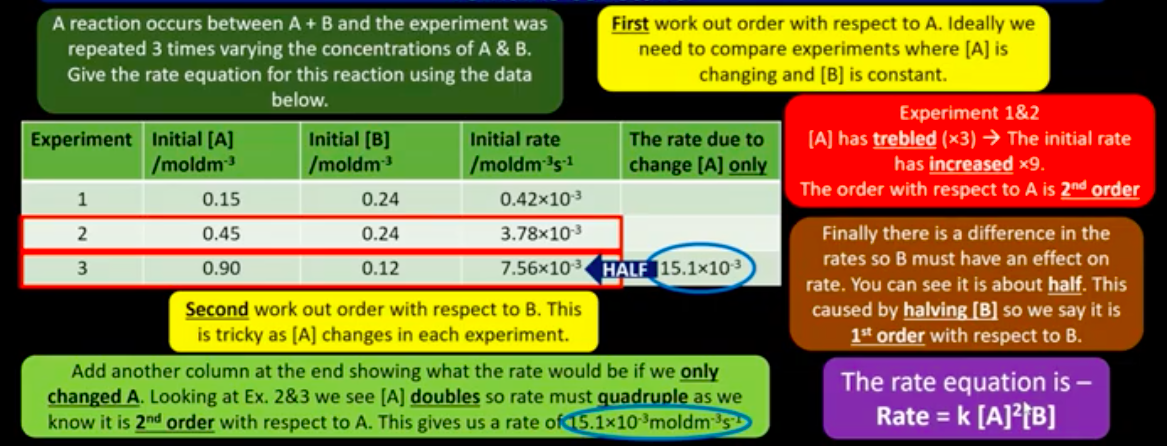
How is the order of a reactant determined?
From experimental data only.
Does K depend on temperature?
Yes. The higher the temperature, the bigger K is.




How is the rate equation determined experimentally for a reaction?



What 3 assumptions are made when finding the initial rate of a clock reaction?

How does the iodine clock reaction work?

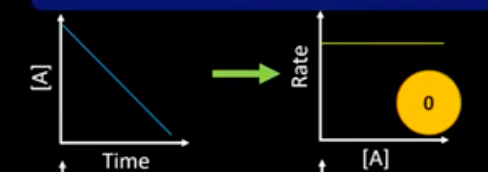
What are the concentration-time and rate-conc graph for a 0 order reactant?
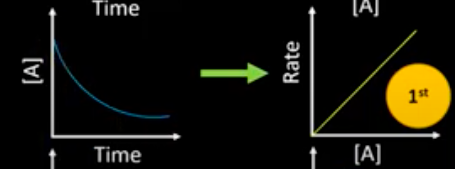
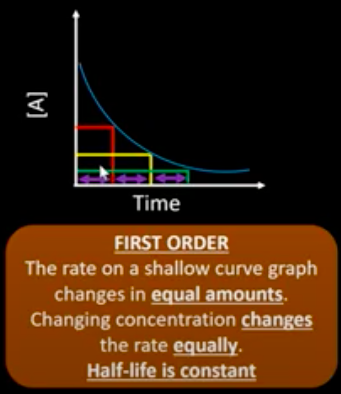
What are the concentration-time and rate-conc graph for a 1 order reactant?
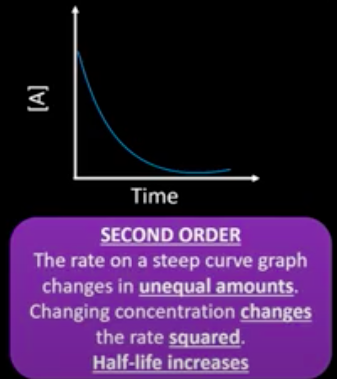
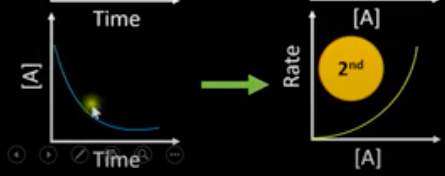
What are the concentration-time and rate-conc graph for a 2 order reactant?
What is the half life of a reactant?
The time it takes for half of the reactant to be used up.
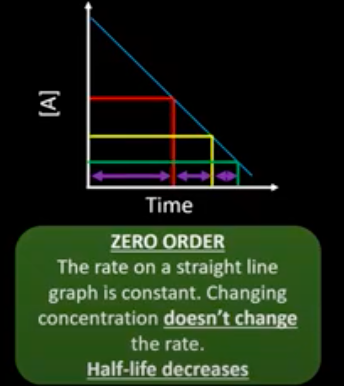
What happens to the half life of a zero order reactant?
What happens to the half life of a first order reactant?
What happens to the half life of a second order reactant?