In the following diagram, the conversion of X to Y is spontaneous, so:
- the forward reaction (X→Y) will always proceed more quickly than the reverse reaction (Y→X)
- the change in Gibbs free energy (ΔG) is negative, meaning that the conversion of X to Y always releases heat
- the concentration of Y will always rise until the rate of the reverse reaction (Y→X) equals the rate of the forward reaction (X→Y)
- at equilibrium, the concentration of Y will always be lower than X, because the Gibbs free energy of Y is lower
- the system will reach a standard state where the concentration of all species will equal 1 molar

C
the concentration of Y will always rise until the rate of the reverse reaction (Y→X) equals the rate of the forward reaction (X→Y)
A is wrong as the forward and reverse reaction rates are the same at equilibrium. B is wrong as ΔG can still be negative when heat is absorbed, provided that there is a large positive ΔS
In the drawings below, which pairs of atoms are involved in hydrogen bonding?
- (1,2)
- (1,2) & (3,4)
- (3,4) & (5,6)
- (1,2) & (5,6)
- all pairs are involved in hydrogen bonding

D

E is incorrect as 3&4 can’t form hydrogen bonds. This is due to the lack of polarisation in the C-H bond.
In the drawings below, which pairs of atoms are involved in hydrogen bonding?

The electronegativity values are: H=2.1; C=2.5; N=3.1; O=3.5
- (1,2)
- (1,2) & (3,4)
- (3,4) & (5,6)
- (1,2) & (5,6)
- all pairs are involved in hydrogen bonding
D

E is incorrect. When you look at the electronegativity values, the values between H and C are too close to create a partial positive/negative charge (polarisation)
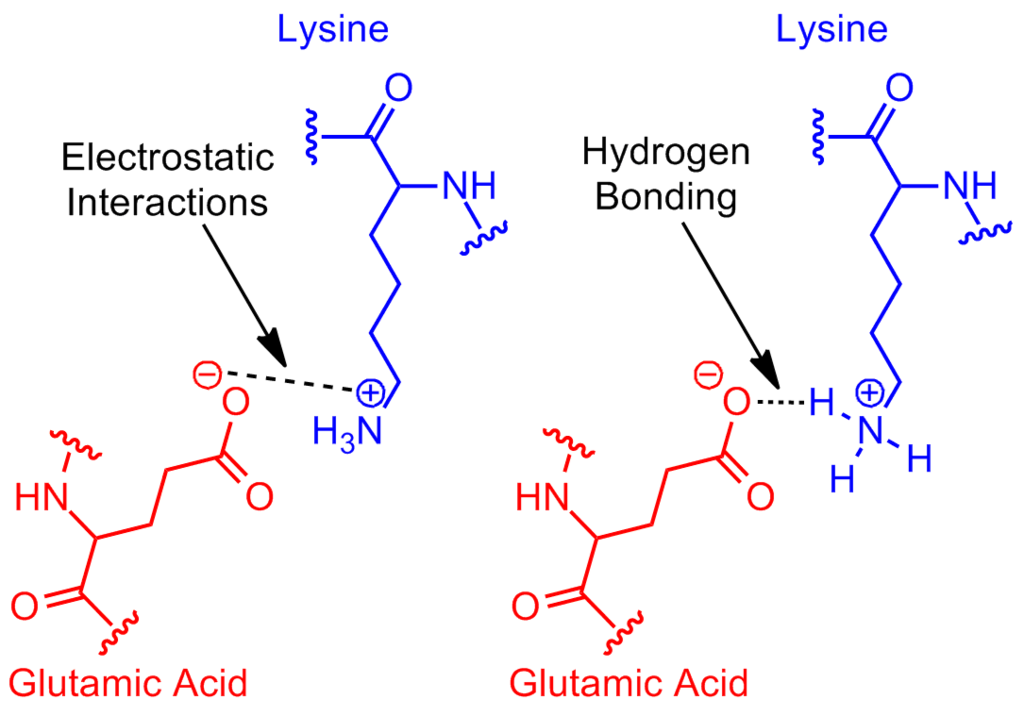
Define: salt bridge
- a salt bridge is a combination of two non-covalent interactions: hydrogen bonding and ionic bonding
- it is an electrostatic interaction between a positively charged group and a negatively charged group, mediated by a hydrogen bond
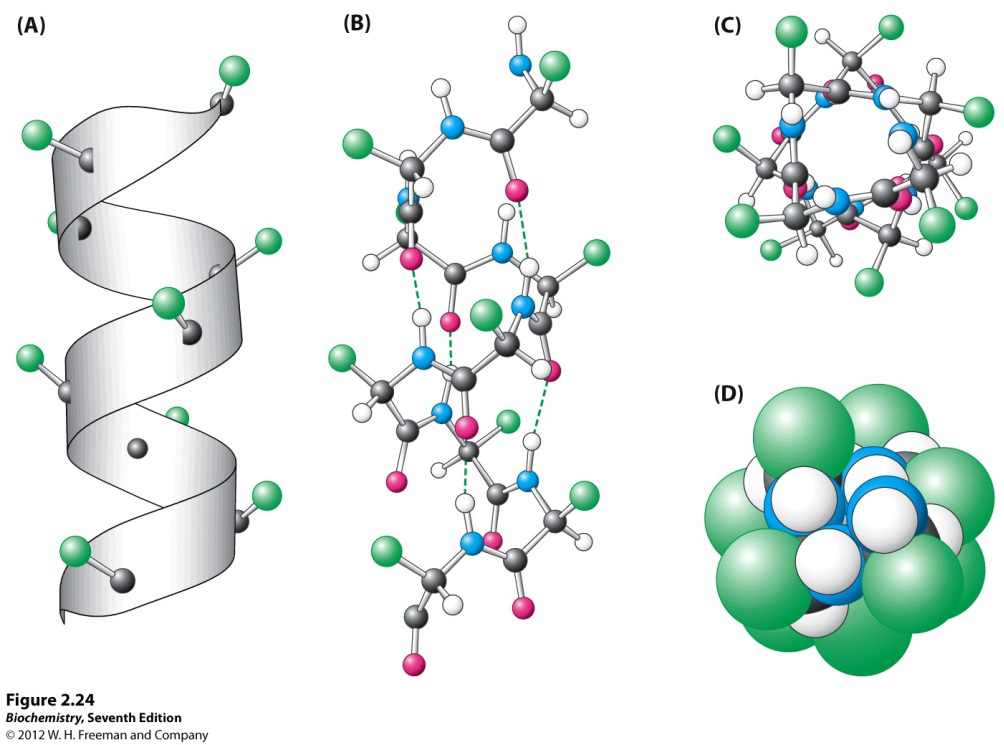
Which statement about alpha helices is true?
- the side chains project inwards filling the central axis
- the interior of an alpha-helix can be filled with water or other small molecules
- each alpha-helix is unique and has a different diameter
- the backbone atoms approach closely filling the interior of the helix
- alpha-helix formation is stabilised by salt-bridges between backbone NH and C=O groups
D
Side chains in an α-helix are projected outwards; there is NO space in the middle for small molecules as the backbone fills most of the interior; there are alpha-helices that are the same; salt-bridges = electrostatic interaction between a positively-charged group and a negatively charged group, mediated by a hydrogen bond
Which statement about alpha helices is true?
- the side chains project inwards filling the central axis
- the interior of an alpha-helix can be filled with water or other small molecules
- each alpha-helix is unique and has a different diameter
- the backbone atoms approach closely filling the interior of the helix
- alpha-helix formation is stabilised by salt-bridges between backbone NH and C=O groups
D
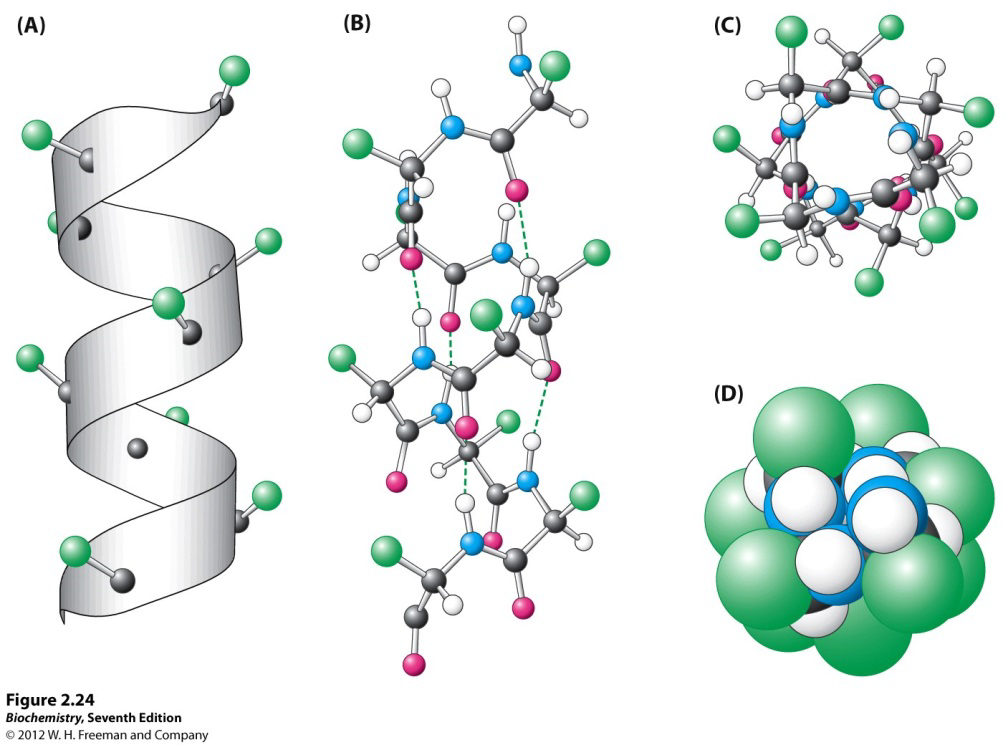
Side chains in an α-helix are projected outwards; there is NO space in the middle for small molecules as the backbone fills most of the interior; there are alpha-helices that are the same; salt-bridges = electrostatic interaction between a positively-charged group and a negatively charged group, mediated by a hydrogen bond
If the pKa of an amine group is 10.5, which statement is correct?
- at pH=5, the predominant charge state will be negative
- at pH=7, the predominant charge state will be positive
- at pH=10.5, the predominant charge state will be neutral
- at pH=14, the predominant charge state will be negative
- the predominant charge state of the group depends on base strength, not on pH
B
A and D are wrong because amines never have a negative charge; predominant charge states are reliant on base strength and pH; C isn’t true because, at pH=10.5, there is an equal balance of both species; B is correct because, as the pH is lowered, there are more protons around which will create more of the protonated state
If the pKa of an amine group is 10.5, which statement is correct?
- at pH=5, the predominant charge state will be negative
- at pH=7, the predominant charge state will be positive
- at pH=10.5, the predominant charge state will be neutral
- at pH=14, the predominant charge state will be negative
- the predominant charge state of the group depends on base strength, not on pH

B

A and D are wrong because amines never have a negative charge; predominant charge states are reliant on base strength and pH; C isn’t true because, at pH=10.5, there is an equal balance of both species; B is correct because, as the pH is lowered, there are more protons around which will create more of the protonated state
Define: pKa
pKa defines the pH where both species are equal in concentration at equilibrium
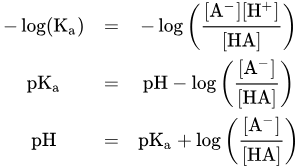
Heat is given off when ATP is hydrolysed according to the equation:
ATP + H2O ↔ ADP + Pi
Which of the following is responsible for the heat release?
- breaking the bonds in ATP and H2O releases heat
- forming bonds to make ADP and Pi releases heat
- both a. and b. release heat
- breaking bonds in H2O releases heat because they are stronger than bonds in ATP
- breaking bonds in ATP releases heat because it is an energy-rich molecule
B
making bonds release energy, breaking bonds require energy
Heat is given off when ATP is hydrolysed according to the equation:
ATP + H2O ↔ ADP + Pi
Which of the following is responsible for the heat release?
- breaking the bonds in ATP and H2O releases heat
- forming bonds to make ADP and Pi releases heat
- both a. and b. release heat
- breaking bonds in H2O releases heat because they are stronger than bonds in ATP
- breaking bonds in ATP releases heat because it is an energy-rich molecule

B

breaking bonds require energy, forming bonds release energy
Which statement about amino acid sequences is true as it applies to protein function? A change in the amino acid sequence of a folded, functional protein:
- will stop the protein from folding
- always changes the function of a protein
- results in a protein with a different level of function
- results in a protein with a lower level of function
- sometimes results in a protein with a different level of function
E
Need to consider:
- what is the change? (e.g. old⇒new amino acid)
- how drastic is the change? (how different are the amino acids?)
- where is the amino acid located? (on the surface, in the core, at the active site etc.)
Q1. The amino acid sequence of a 16-residue peptide is:
IFAILEDCHEMISTRY
- at pH 7 how many positively charged groups would be present?
- name those groups
- at pH 7 how many negatively charged groups would be present?
- name those groups
- at pH 7, what would the overall charge of this peptide be
- if the average mass of an amino acid is 110 Da, what would the mass of this peptide be under oxidising and reducing conditions, explain your answers
- 3
- N-terminus/ammonium; histidine/imidazole; arginine/guanidium/guanidino
- 4
- C-terminus/carboxylate/carboxylic acid; glutamic acid/glutamate/carboxylic acid; aspartic acid
- overall charge = (2x positive from N-terminus and arginine) + (4x negatives from C-terminus and 2 glutamates and an aspartate) = -2 (can be -1 if we consider histidine to carry a positive charge)
- Under reducing (separate cysteines) = 1.76 kDa; under oxidising (joint cystines) = 3.52 kDa
Q2. Draw a dipeptide composed of:
- a branched nonpolar amino acid and a basic amino acid. Indicate all atoms (including the number of hydrogen atoms attached to each heavy atom)
- give the full name, three letter code and single letter code for each amino acid
- show all ionisable groups in the state most highly populated at pH 7
- draw the peptide bond as trans, label the peptide bond
Show all atoms
insert drawing
Q3. Draw a simplified schematic diagram illustrating the backbone to backbone hydrogen bonding pattern in an alpha-helix
- indicate on your diagram whether the helix is right-handed or left-handed
- the position of N- and C- termini
- the sequence numbering relationship between a pair of hydrogen-bond residues
- show the location of sidechains with “R”
- indicate with an arrow and a numerical value the axial translation per turn
Q4. Draw a diagram illustrating the backbone to backbone hydrogen bonding pattern: between two antiparallel beta-strands
- show all the backbone atoms (including hydrogens) for three residues on each strand. Sidechains should be indicated with “R”
- indicate the hydrogen bonds with dashed lines
- show the appropriate stereochemistry for alpha carbon on the middle residue on each strand (i.e. correctly indicate which group is above/below the plane of the page)


