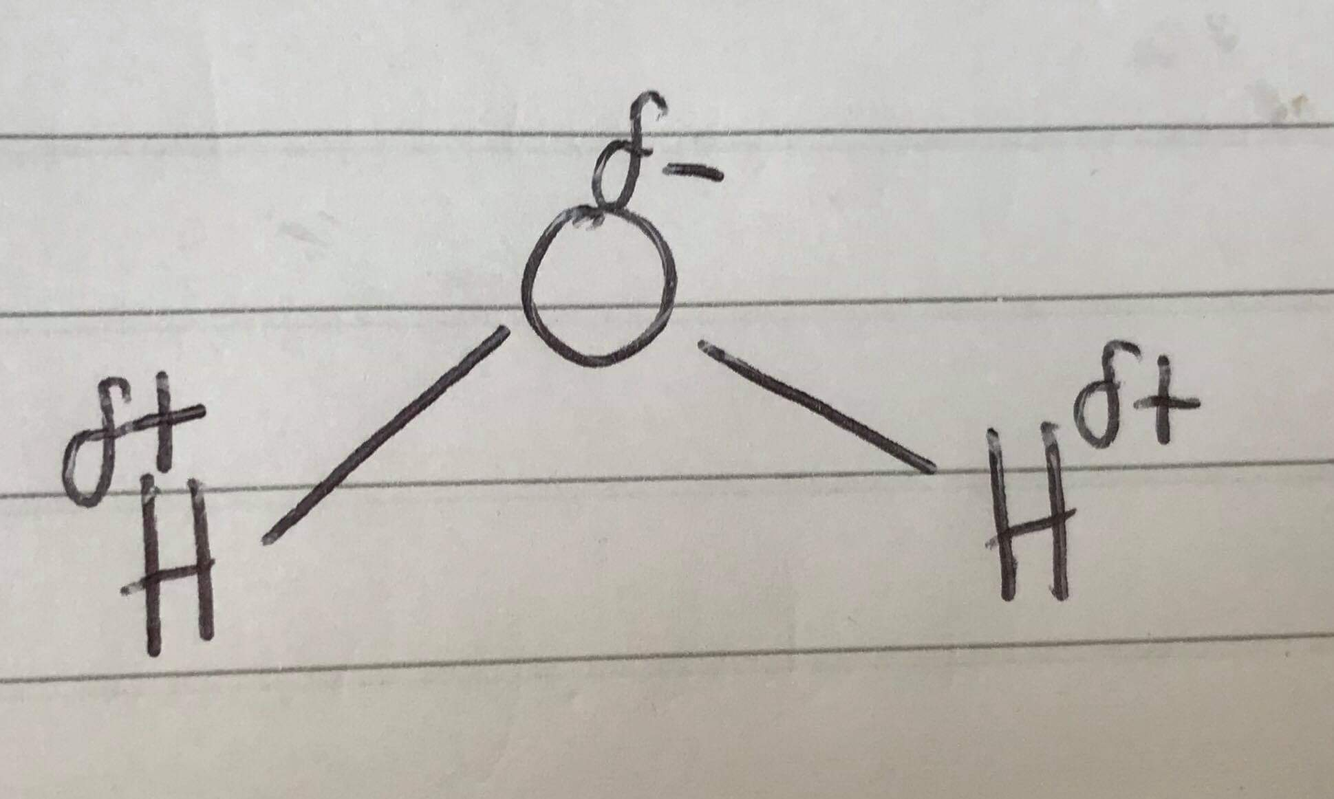
Draw a water molecule and label the charges on the atoms
Delta positive charge on H
Delta negative charge on O
What bonds are formed between the hydrogen and oxygen of different water molecules?
Hydrogen bonds
What does it mean for a molecule to be polar?
It means the molecule has an unequal spread of electrons so has partial charges within the molecule
How are the electrons shared within a water molecule? Why?
Unequally, electrons spend more time around oxygen because:
- Oxygen is highly electronegative element, more electronegative than hydrogen
- Therefore the oxygen atom attracts electrons towards it
How is a hydrogen bond drawn?
A dotted line
Between which atoms does a Hydrogen bond form?
Hydrogen and Oxygen of different water molecules
What is the difference between adhesion and cohesion?
Adhesion is when water ‘sticks’ to another surface whereas cohesion is when water is attracted to other water molecules.
What is meant by a high specific heat capacity of water?
A lot of energy is require to increase the temperature of water
Why does water have a high specific heat capacity?
- the hydrogen bonds between the molecules must be overcome to increase the temperature of water
- this requires energy
Why does ice float on liquid water?
- When water freezes a crystal lattice structure is formed —so the molecules are held further apart
- this decreases density
Explain what is meant by the phrase ‘like dissolves like’
- polar substances dissolve other polar substances
- non-polar substances dissolve non-polar substances
Name 6 physical properties of water
- High specific heat capacity
- High latent heat of evaporation
- Ice floats on liquid water
- Surface tension
- Can act as a solvent for polar molecules
- Good transport medium
How does a high latent heat of evaporation of water help organisms to survive?
A lot of energy is required to change water from liquid to gas therefore it is very effective in cooling surfaces by evaporation. This helps animals in hot environments (E.g dogs panting)
How does the ability of ice to float on liquid water enable organisms to survive? (3 ways)
1-Provides LAND animals a means of sheltering and catching food even in freezing temperatures
2-Floating ice insulates water floating underneath, assisting aquatic animals survival in low temperatures as they do not freeze
3-Ice insulates water so water remains liquid and organisms can still swim and catch prey
How does water as a liquid assist the survival of organisms? (5 ways)
- Water is polar so is a good solvent for polar molecules
- As good solvent water can transport vital substances around organisms (e.g. glucose in the blood)
- Can dilute toxic substances within an organism
- Cohesion gives water surface tension which provides invertebrates with a habitat
- High specific heat capacity means water provides aquatic organisms with a thermostable habitat, ensuring enzymes work optimally
Suggest how water acting as a solvent for ions (such as nitrates) can contribute to a stable environment for aquatic organisms
- Plants are able to absorb the dissolved nitrates and grow well
- Plants provide food source for other organisms
- Plants photosynthesise and produce oxygen which is needed by other organisms for survival
- Nitrates are used to build amino acids in plants
Define Hydrophilic
Readily attracted to water
Define Hydrophobic
Repelled by water
Why do hydrogen bonds form between hydrogen and oxygen atoms of different water molecules?
Hydrogen has a partial positive charge and Oxygen has a partial negative charge (due to the electronegativity of oxygen) so they attract and form a hydrogen bond.


