X-rays are a form of what kind of radiation?
Electromagnetic radiation

The flow of energy from electromagnetic radiation is created by what? What is it represented by?
Simultaneously varying electrical and magnetic fields Represented as a sine wave

What are the properties of electromagnetic radiation? (4)
- no mass
- no charge
- Always travels at the ‘speed of light’ (3x108 ms-1 = 671 million mph)
- Can travel in a vaccum
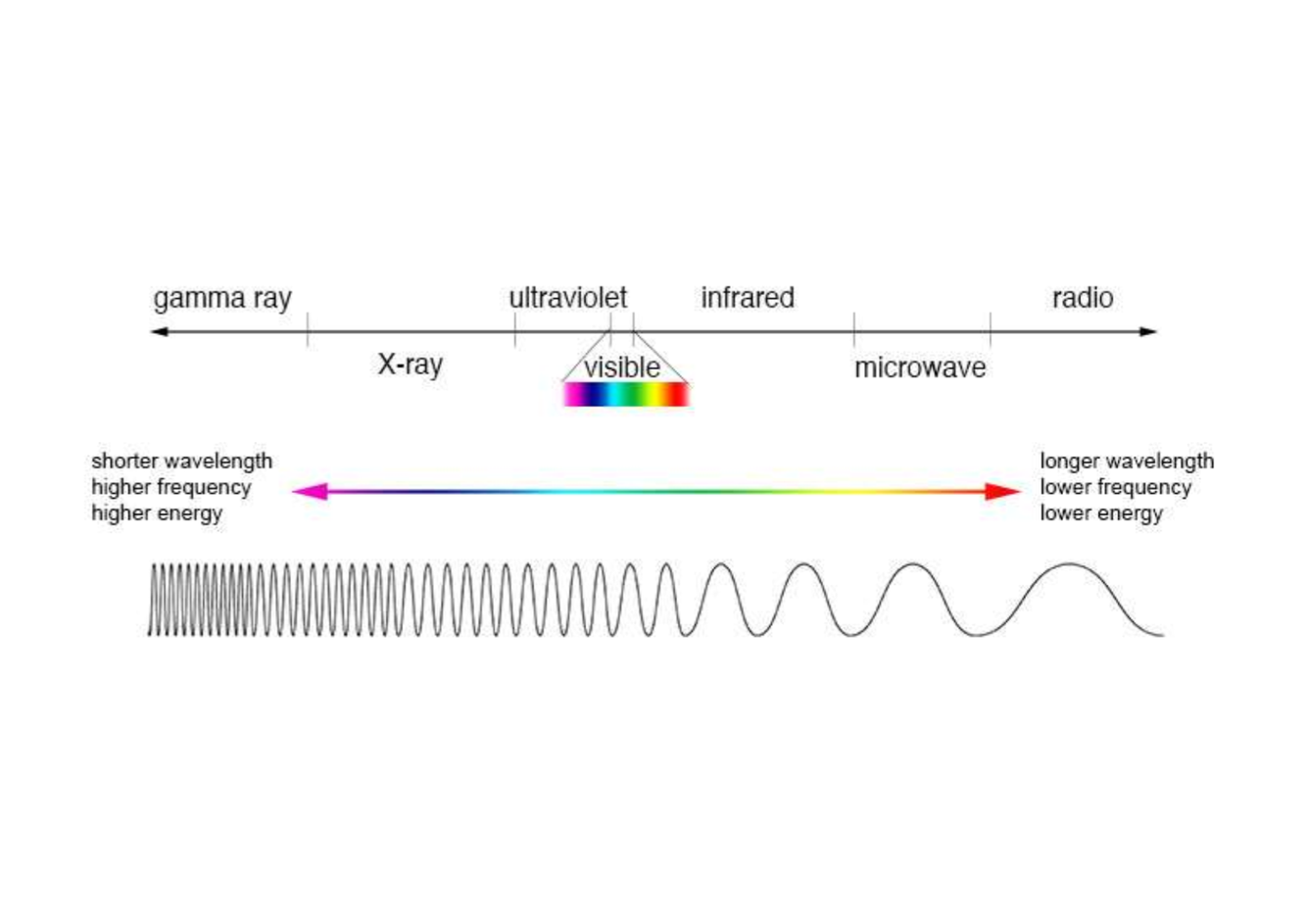
Describe the EM spectrum (3 main points).
- consists of all the diff types of EM radiation
- Each type has diff properties depening on its energy/wavelength/frequency
- 7 main groups
What are the 7 EM groups?
- gamma ray
- X-ray
- Ultraviolet
- Visible light
- Infrared
- Microwave
- Radio
Describe gamma rays in terms of wavelength, frequency and energy.
Shorter wavelength, higher frequency and higher energy
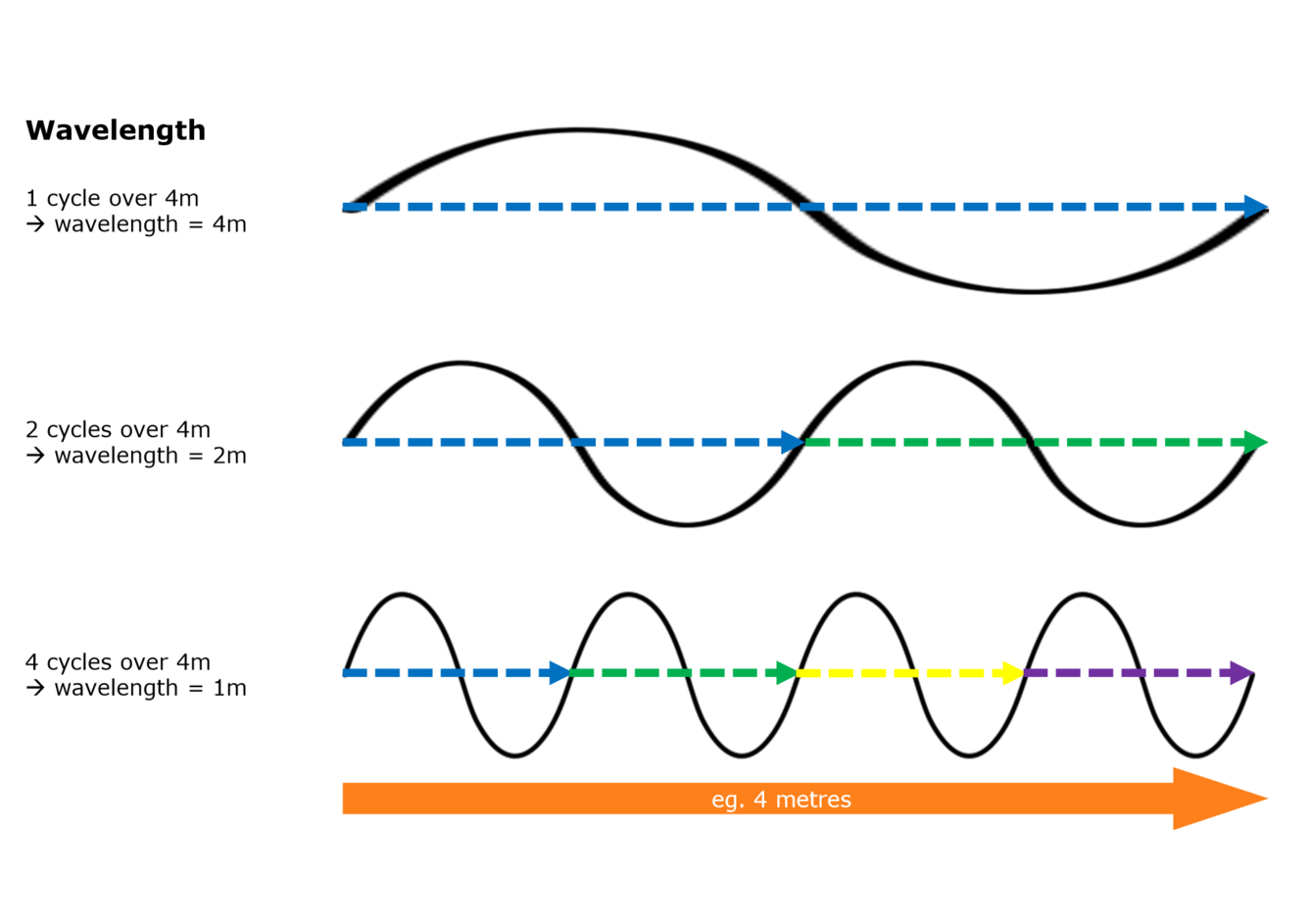
What is the wavelength?
How ‘big’ one cycle is
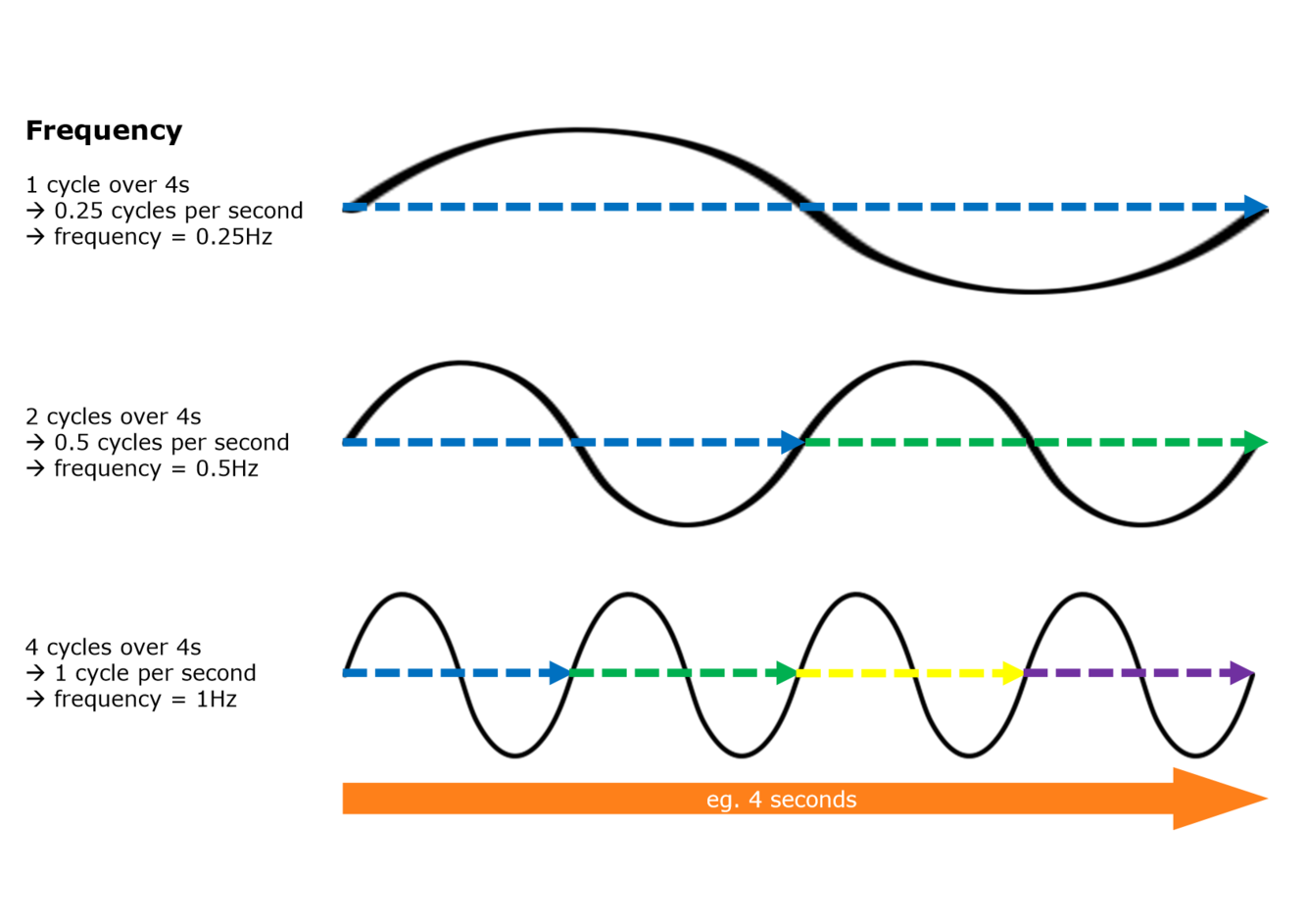
What is the frequency of a wave? What is it measured in?
How many times the wave’s shape repeats per unit time
Hertz (Hz) [one hertz = one cycle per sec]
Speed = frequecny x wavelength
BUT EM radiation speed is constant (3x108ms-1)
What does this mean for the relationship between frequency and wavelength in EM radiation?
If frequency increases then wavelength must decrease (and vis versa)
photons and mocement of enery etc
What are the 2 types of X-rays you get?
- Hard x-rays
- Soft x-rays
Describe hard x-rays.
Higher energies and are able to penetrate human tissues
(mediacal imaging mainly uses these types of x-rays)
Descibe soft X-rays.
Lower energies and easily absorbed
NOTE: dont want soft x-rays if want radiographs as you need some x-rays to reach the receptors
What are the basic properties of X-rays? (4)
- form of EM radiation (no mass, no charge, very fast, can travel in a vaccum)
- undetectable to human senses (contrasts with visible light)
- Man-made
- Cause ionisation (displacement of electrons from atoms/molecules)
How are x-rays and gamma rays similar?
They are identical except that gamma rays ocur naturally and X-rays are man-made
Gamma rays also generally have higher energies
Describe the basic production of X-rays.
- Electrons fired at atoms at very high speed
- On collision, the kinetic energy of these electrons is converted to electromagnetic radiation (ideally X-rays) and heat is produced as a side-effect
- The X-ray phoons are aimed at a subject
What is the basic structure of an atom?
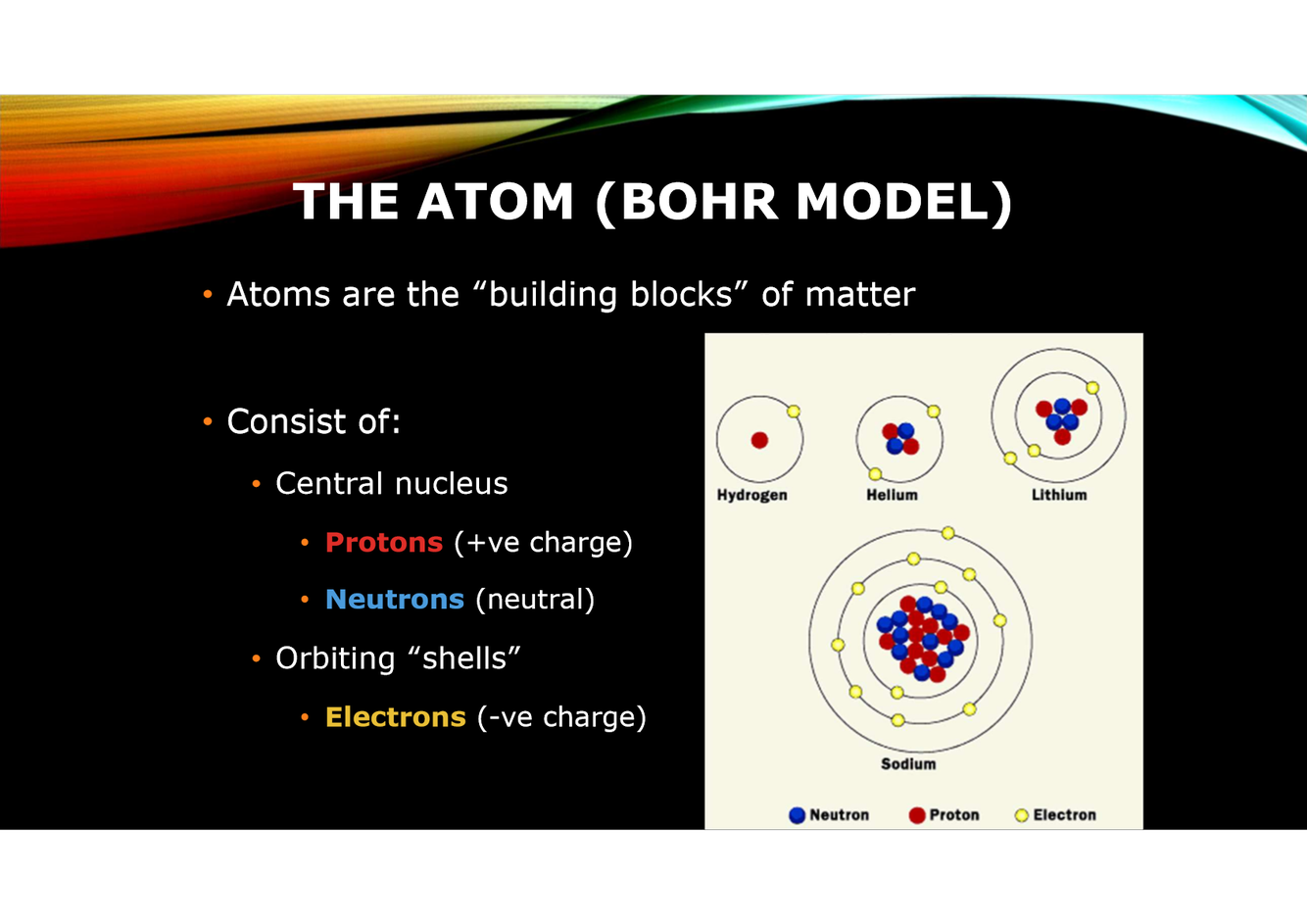
Describe neutrons, protons and electrons. (charge, mass, location)
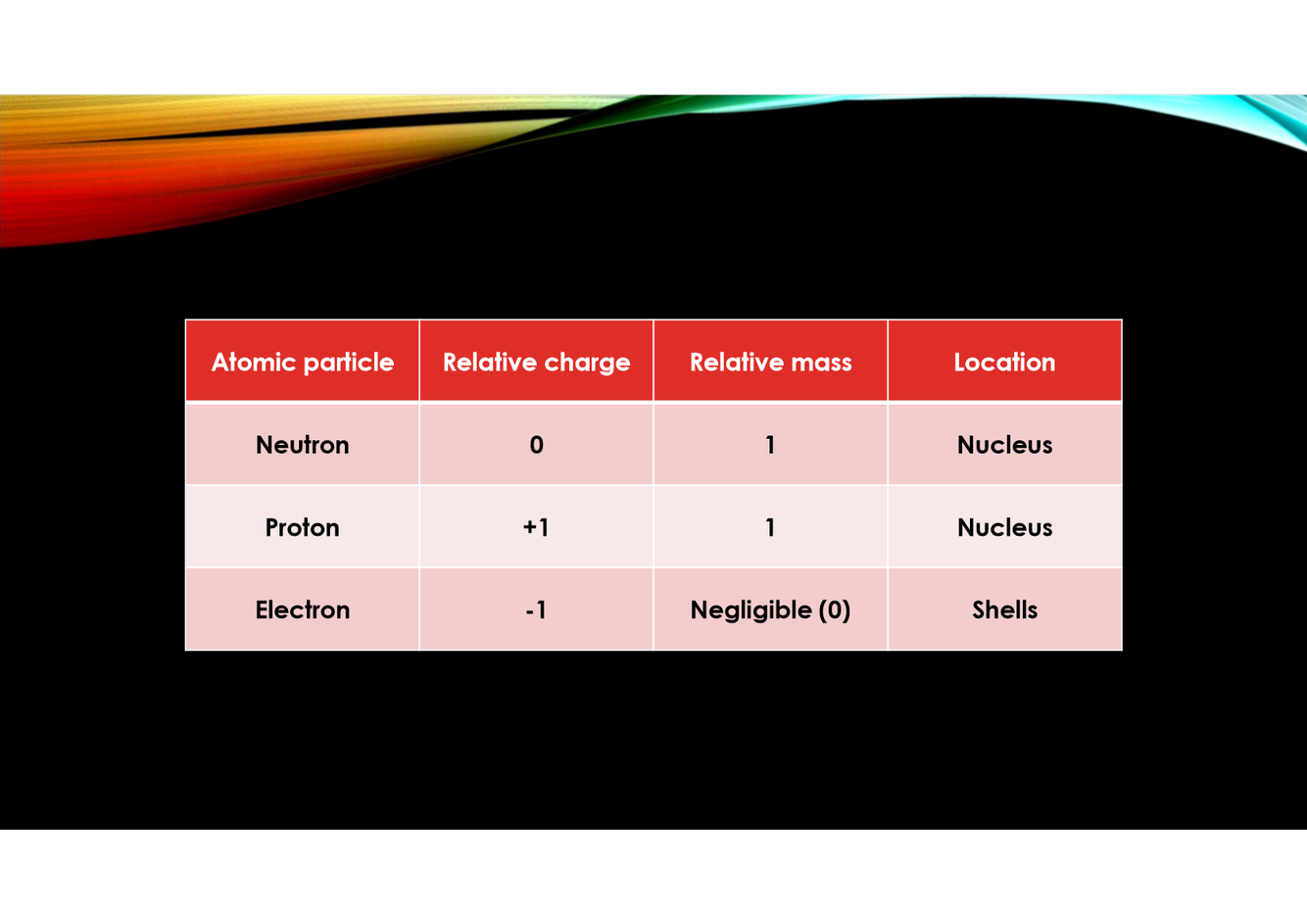
What is the overall charge of the nucleus?
Positive
What dictates the atomic number and mass number of an element?
AN = number of protons
Mass number = protons + neutrons
The number of electrons determines what in an atom?
Its chemical properties
What is the innermost electron shell?
K (then it foes L, M, N, O etc.)
How do you figure out the max no. of electrons a shell can hold?
max. no. = 2n2 (n=shell number)
e. g. L shell = 2x22 = 8 electrons
Max no.’s for first shell’s go (2, 8, 18, 32)
Orbiting electrons are held within their shells by what?
Electrostatic force
(-ve charge of electrons attracted to overall +ve charge of nucleus)












