What type of bond is a peptide bond?
- A specialized form of an amide bond that is formed between the COO- group of one amino acid and the NH3+ group fo antoher amino acid
- Peptide bonds are formed as a result of a condensation (or dehydration) reaction because it results in the removal of water
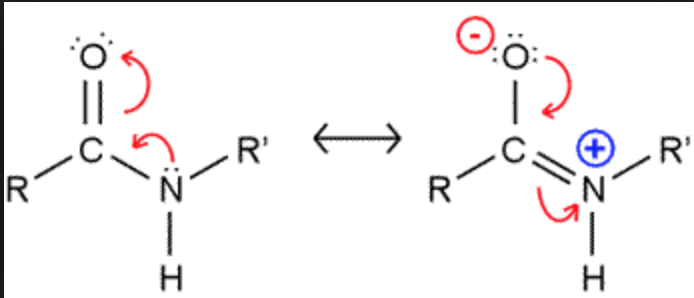
Why is rotation of the protein backbone around its C-N amide bond restricted?
- Because amide groups have delocalizable pi electrons in the cabonyl and in the lone pair on the nitrogen, they can exhibit resonance; thus, the C-N bond in the amide has partial double bond character
What are conjugated proteins?
- Derive part of their function from covalently attached molecules called prosthetic groups
- Proteins with a lipid, carbohydrate, and nucleic acid prosthetic group are called lipoproteins, glycoproteins and nucleoproteins, respectively
What effect do enzymes have on a reaction?
- Lower the activation energy
- Increase the rate of the reaction
- Do not alter the equilibrium constant
- Are not changed or consumed in the reaction
- Are pH and temperature sensitive, with optimal activity at specific pH ranges and temperatures
- Do not affect the overall ΔG of the reaction
- Are specific for a particular reaction or class of reactions
What do transferases do?
- Catalyze the movement of a functional group of one molecule to another
Example: an aminotransferase can convert aspartate and α-ketoglutarate to glutamate and oxaloacetate by moving the amino group from aspartate to α-ketoglutarate
What do hydrolases do?
- Catalyze the breaking of a compound into two molecules, using the additon of water
What do lyases do?
- catalyze the cleavage of a single moleulce into two products
- They do not require water
- Because most enzymes can also catalyze the reverse of their specific reactions, the synthesis of two molecules into a single molecule may also be catalyzed by a lyase (these are synthases)
What do isomerases do?
- Catalyze the rearragement of bonds within a molecule
- Catalyze reactions between stereoisomers as well as constitutional isomers
What do ligases do?
- Catalyze addition or synthesis reactions, generally between large similar molecules and often require ATP
What is a cofactor?
- Generally inorganic molecules or metal ions and are often ingested as dietary minerals
What is a coenzyme?
- Small organic groups
What is an apoenzyme?
- Enzymes without their cofactors
What is a holoenzyme?
- Enzymes with their cofactor
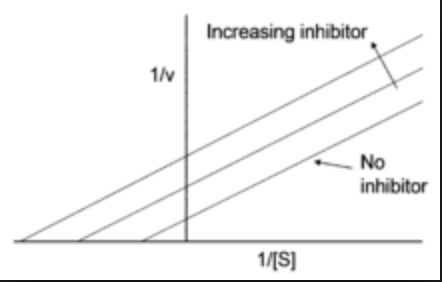
What is the effect of a competitive inhibitor on Vmax and Km?
Vmax: unchanged; if enough substrate is added, it will displace the inhibtor
Km: increased; the substrate concentration has be higher to react half of the maximum velocity in the presence of the inhibitor
What is the effect of a non-competitive inhibitor on Vmax and Km?
Vmax: decreased, becuse there is less enzyme available to react
Km: same; because any enzymes that are still active maintain the same affinity for their substrate
What is the effect of uncompetitive inhibition on Vmax and Km?
Vmax: decreased
Km: decreased
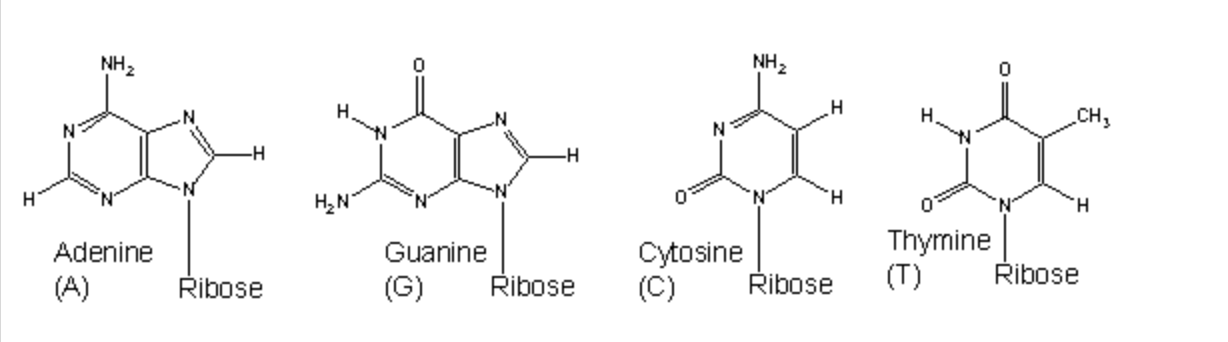
Draw out the 4 nucleotides
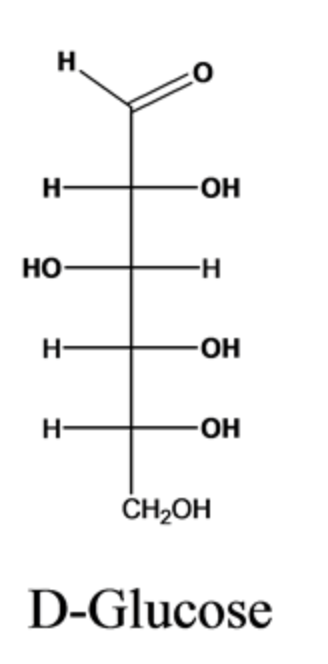
Draw out the Fischer projection for D- glucose
Draw the Fischer projection for D-galactose
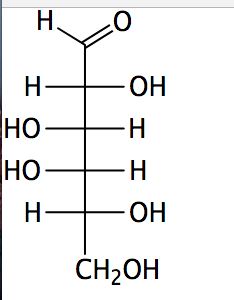
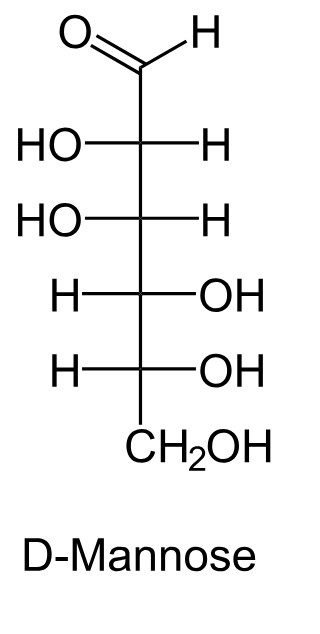
Draw the Fischer projection for D-mannose
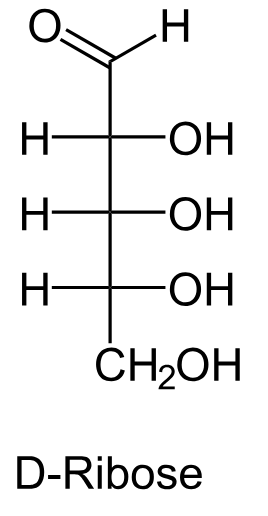
Draw the Fischer projection for D-ribose
What is the primary thing that cause secondary structures of proteins?
Secondary structures are primarily the result of hydrogen bonding between nearby amino acids
What is the role of proline in secondary structures?
- Proline will introduce a kink in the peptide chain when it is found in the middle of an α helix or β pleated sheets
- However, proline is often found in the turns between the chains of β pleated sheets and is often found as the residuce at the start of an alpha helix
What factors determine a proteins tertiary structure?
- Mostly determined by hydrophilic and hydrophobic interactions between R groups of amino acids
- Hydrophobic residues prefer to be on the interior or proteins, which reduces their proximity to water and hydrophilic residues prefer to be on the outside of the protein
- Also determined by disulfide bonds, the form when two cysteine molecules become oxidized to form cystine
- This creates loops in the protein chain































