Substrate Concentration
- The number of molecules of reactants (substrate concentration) determines the rate of a reaction.
- Increasing their number boosts the likelihood of interaction with the enzyme.
Substrate Binding Site
- The substrate binding site is a specific site on an enzyme where the reactant attaches, and the product is released from the neighboring active site.
- Binding of the substrate creates an enzyme-substrate complex.
- When all enzyme molecules contain bound substrate, the enzyme is saturated so that the rate of the reaction becomes independent of the substrate concentration.
Vmax
- When all enzyme molecules contain bound substrate, the enzyme is saturated so that the rate of the reaction becomes independent of the substrate concentration.
- Under these conditions the maximal velocity (Vmax) of the reaction is achieved. Vmax is in part a function of inherent enzyme properties (i.e., some enzymes catalyze their reactions faster than others – greater efficiency).
Catalytic Constant (Kcat)
•The rate at which an enzyme can catalyze a single reaction is called the catalytic constant (Kcat), which is the number of substrate molecules converted to product per second per enzyme molecule.
Relationship betwen Substrate Use and Product Formation
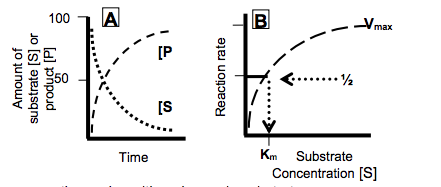
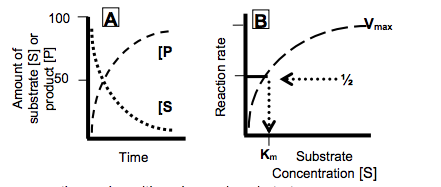
- As substrate is consumed, the product appears initially at a linear rate but more slowly as substrate disappears.
- Vmax also depends on the total concentration of enzyme.
-While the reaction rate at Vmax is independent of substrate concentration, the Vmax depends on the number of enzyme molecules. For instance, doubling the number of molecules doubles the Vmax value.
•The change in the rate of a simple enzyme reaction varies with a change in substrate concentration.
Substarte Affinity
•The substrate affinity reflects how well the substrate site binds the reactant (substrate).
-This affinity is expressed as the Michaelis constant, Km, which is the concentration of substrate that causes half of the enzyme to have substrate bound and the velocity (rate) of the reaction to be ½Vmax.
•The stronger the interaction between the substrate and its binding site on the enzyme the lower the concentration needed to have substrate occupying half of the enzyme molecules and thereby to achieve ½Vmax.
-A lower Km reflects a stronger affinity of the enzyme for its substrate.
- Some enzymatic defects cause this affinity of the enzyme for the substrate, or even for the cofactor, to be weakened.
- For that defective enzyme, the concentration of substrate (or cofactor) needed to achieve the same rate as for the unaffected enzyme, is increased.
-Such a situation leads to a measured Km that is greater than normal and the clinical symptoms resulting from that reduced reaction rate.
- The reduced rate leads to a lower amount of product, under usual conditions, and in most instances an increase in the concentration of substrate because of its decreased conversion to product.
- Many medications act as competitive inhibitors by competing with the natural substrate for the substrate-binding site. Each medication has a Ki value, which like Km indicates how strongly the medication interacts with the enzyme.
-Ideally medications should have a Ki much lower than the Km so that only small amounts of medication are needed to reduce the rate of the reaction.
Structurak Features of Collagen
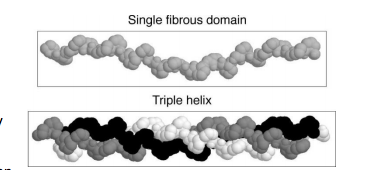
- Collagens, predominantly synthesized by fibroblasts, share a common structure with a distinguishing unique triple helix, stabilized by unusual cross-links.
- The triple helix consists of three pro-collagen-alpha-chains very tightly wound around each other.
- Each of these alpha-chains is itself helical because the alphachain has a glycine residue at every third position.
-Thus, the sequence of collagen chains is [Gly–X–Y]N.
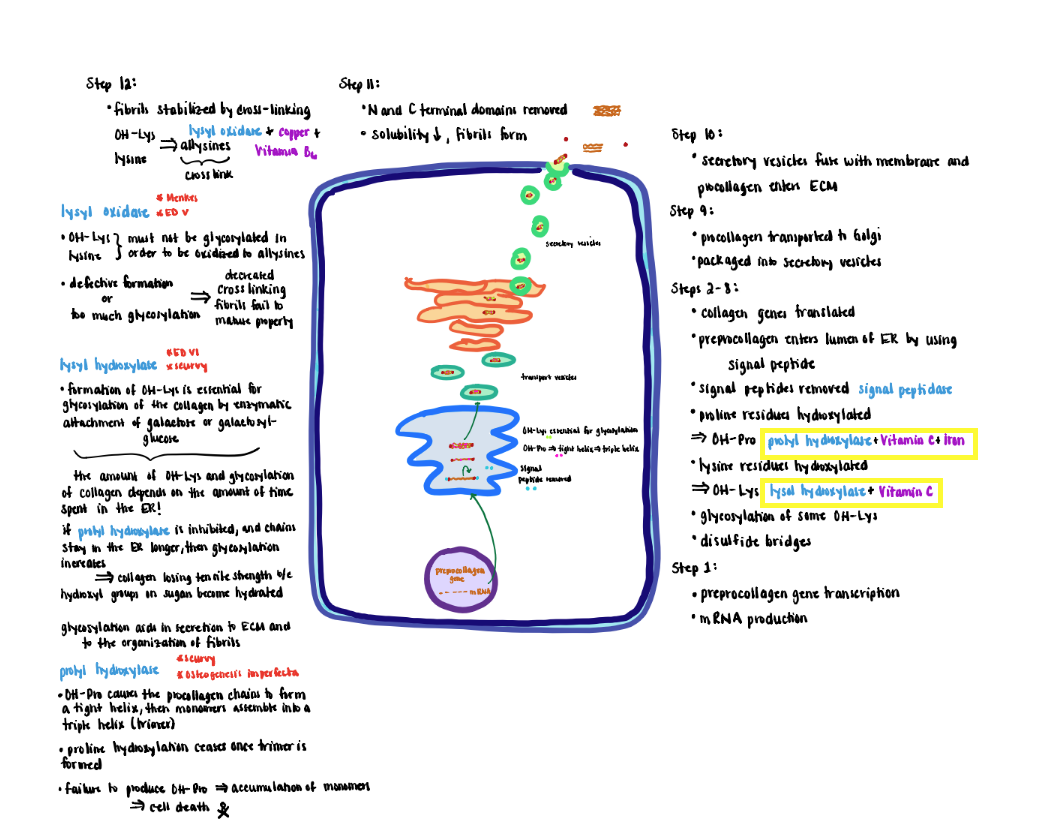
Collagen Processing - Step 1 (Step1-2)
1) Collagen genes transcribed in the nucleus produce mRNA, which is translated by ribosomes on the rough endoplasmic reticulum (ER).
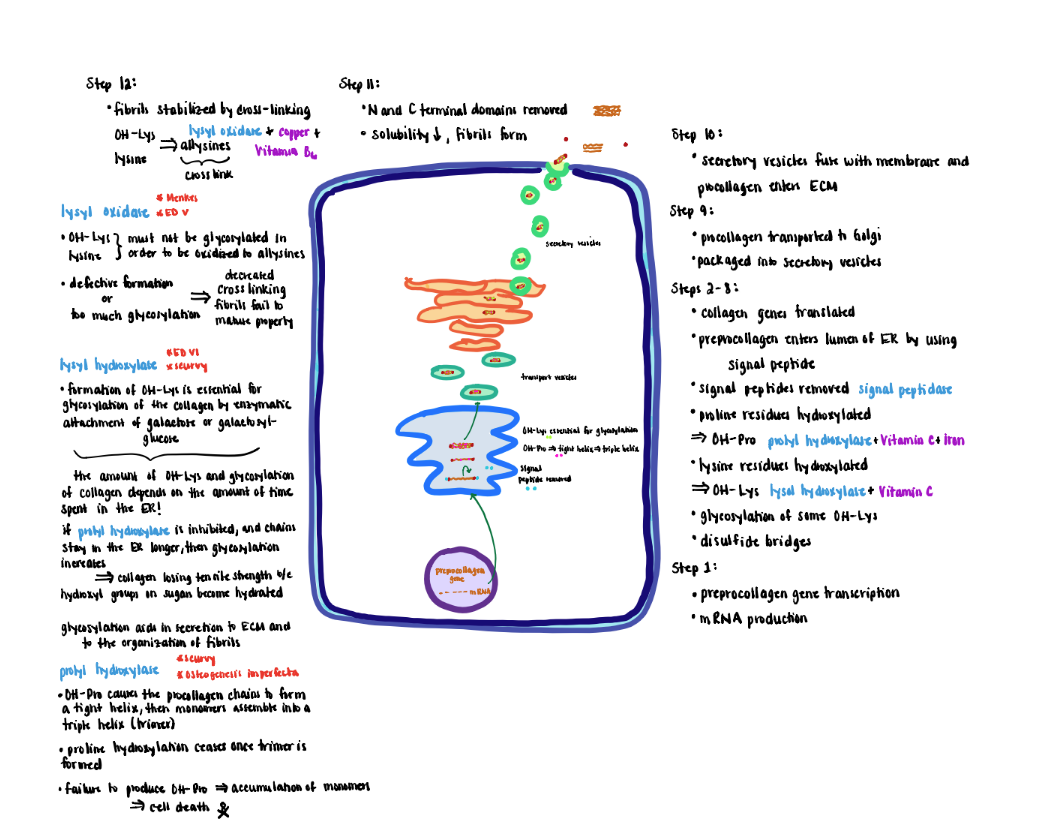
Collagen Processing Step 2 (Step 3)
2) The prepro-alpha-chain moves into the lumen of the ER where the signal peptide (‘pre’ portion) is cleaved by a signal peptidase to form the pro-alpha chains.
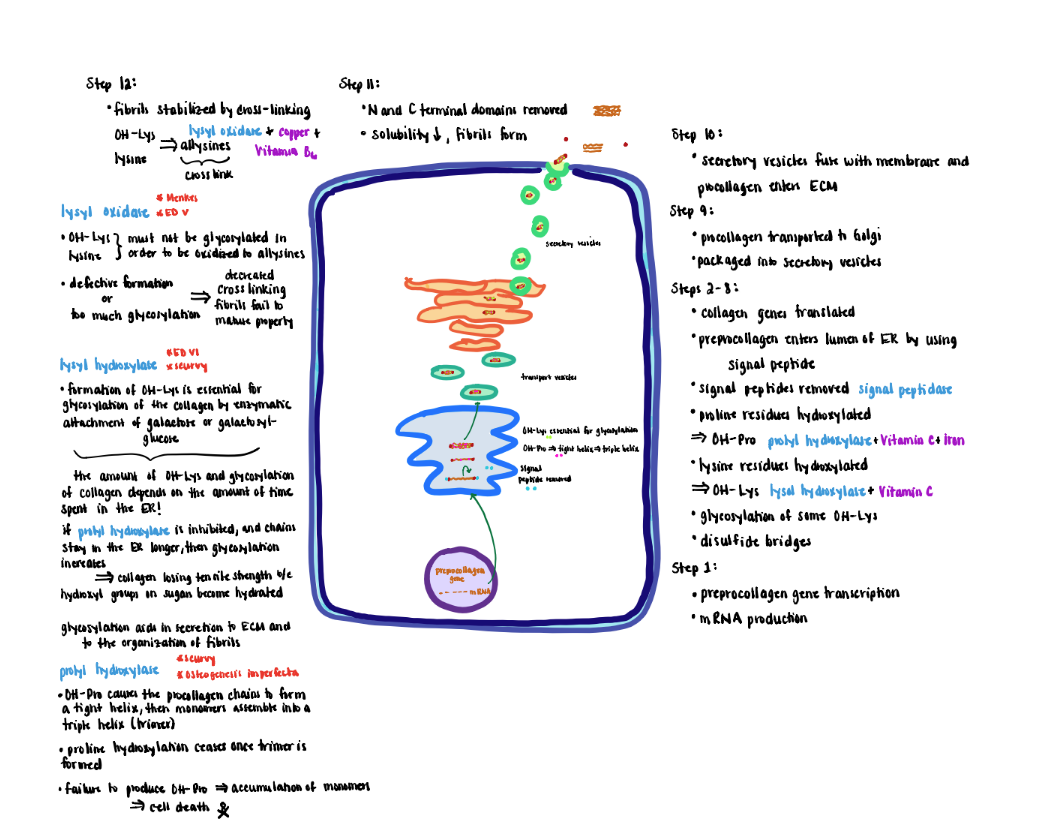
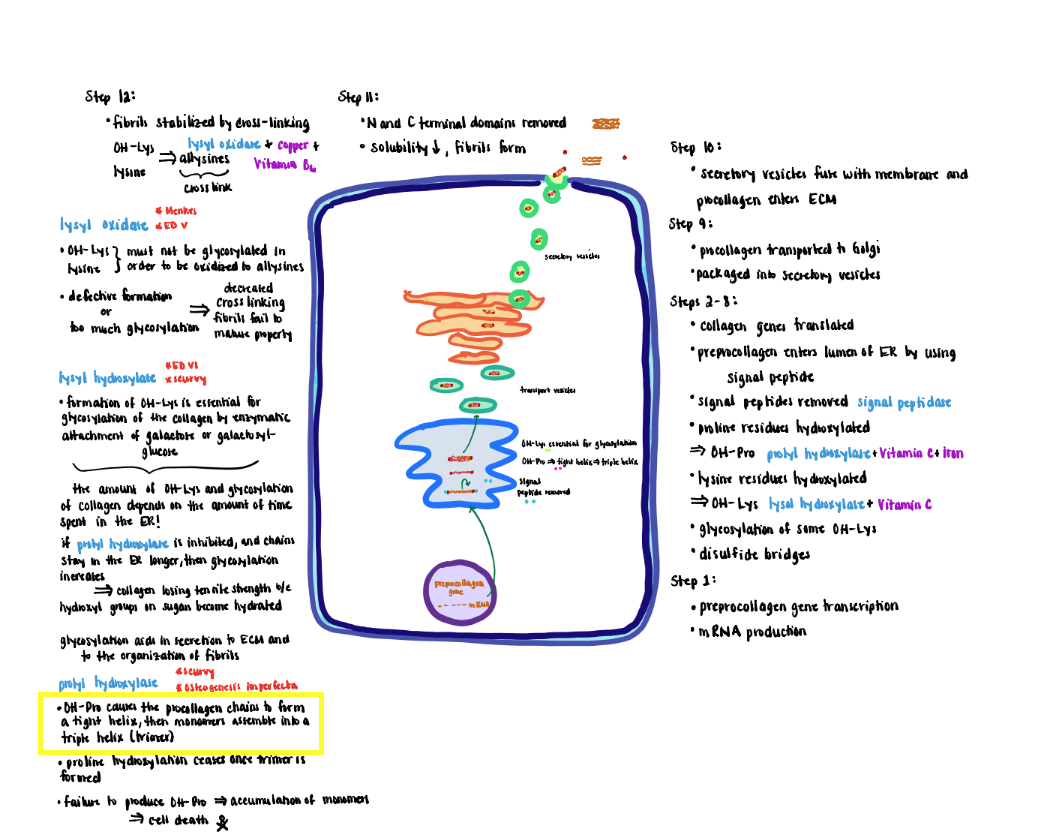
Collagen Processing Step 3 (Step 5)
3) Proline residues are hydroxylated to form hydroxyproline (OH-Pro) by prolyl hydroxylase, which contains iron and requires ascorbic acid (vitamin C) as a cofactor to maintain the iron in its Fe+2 (ferrous; reduced) state.
- Proline hydroxylation causes the proalpha-chain to become a tight helix that is required for the subsequent assembly of monomers into a triple helix (trimer).
- Prolyl hydroxylase only recognizes monomer chains. Therefore, proline hydroxylation ceases once the trimer forms.
- Failure to produce OH-Pro prevents the monomers from assembling into mature trimers causing the alphachain monomers to accumulate in the ER eventually leading to cell death.
- Formation of OH-Pro is tightly coupled to triple helix formation that inhibits further activity of the prolyl hydroxylase. Hence OH-Pro plays an important role in the initial assembly process.
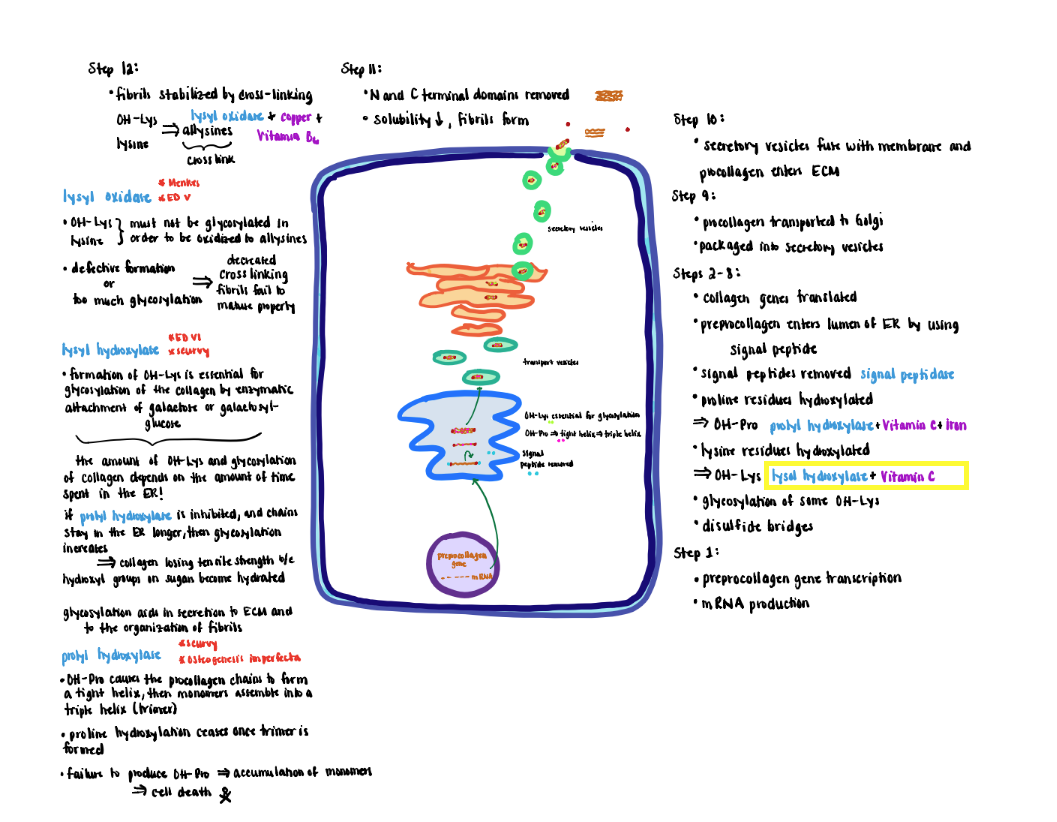
Collagen Processing Step 4 (Steps 6-8)
4) Lysine residues are hydroxylated to form hydroxylysine (OH-Lys) by lysyl hydroxylase that also requires ascorbic acid and iron.
- Formation of OH-Lys is essential for the glycosylation of the collagen by enzymatic attachment of sugars (galactose or galactosylglucose).
- The OH-Lys and sugar content of collagen is determined by how long the procollagen alpha-chains remain in the lumen of the ER before they are assembled into a triple helix.
- If prolyl hydroxylase is inhibited causing the chains to remain longer in the ER, the extent of glycosylation increases. Increased glycosylation results in collagen losing tensile strength because the hydroxyl groups on the sugars become hydrated.
- Potential roles for glycosylation include aiding in the secretion to the extracellular matrix and organization of the fibrils.
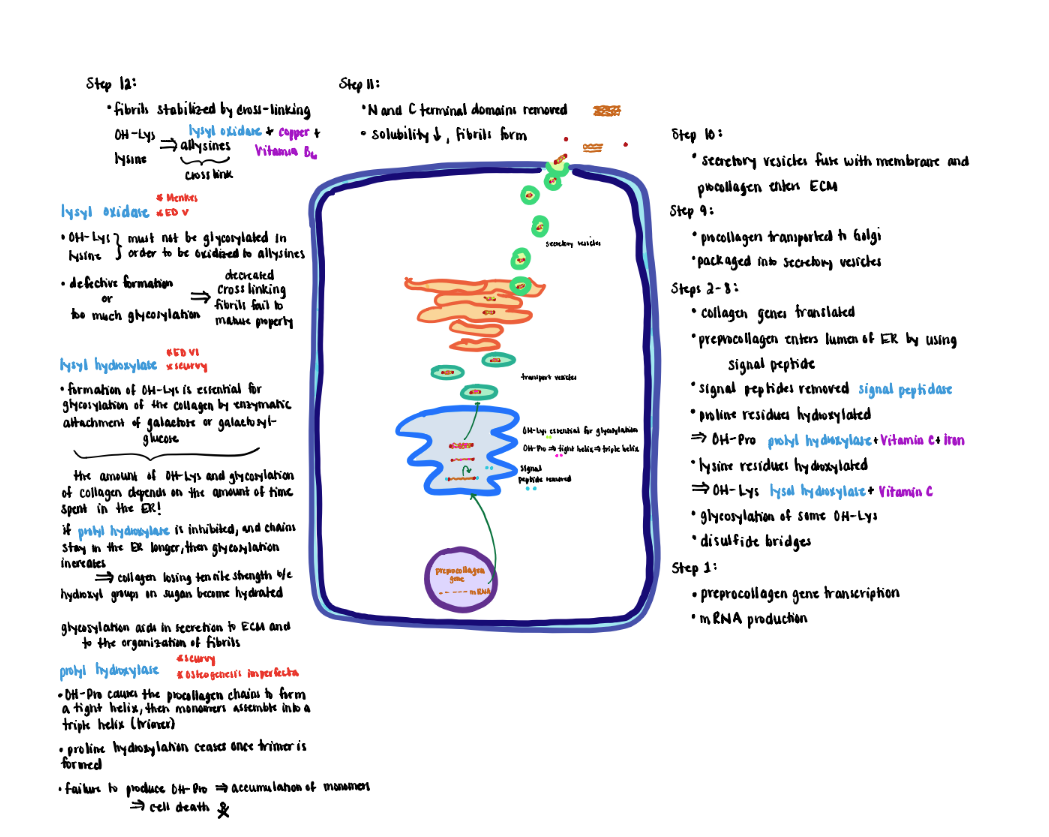
Collagen Processing Step 5 (Step 8)
5) Efficient formation of the triple helix requires that the three alpha-chains be properly aligned. Proper alignment is achieved by oxidation of cysteine residues (free SH groups) in the C-terminal non-helical segments to disulfide bridges (-S-S-) that hold the chains in position for the intertwining of the three chains.
Collagen Processing Step 6 - 7 (Step 9-10)
6) Next pro-collagen is transported from the ER to the Golgi via transfer vesicles.
7) The Golgi packages the pro-collagen triple helix into secretory vesicles that fuse with the membrane and secrete their contents to the extracellular matrix for further processing.
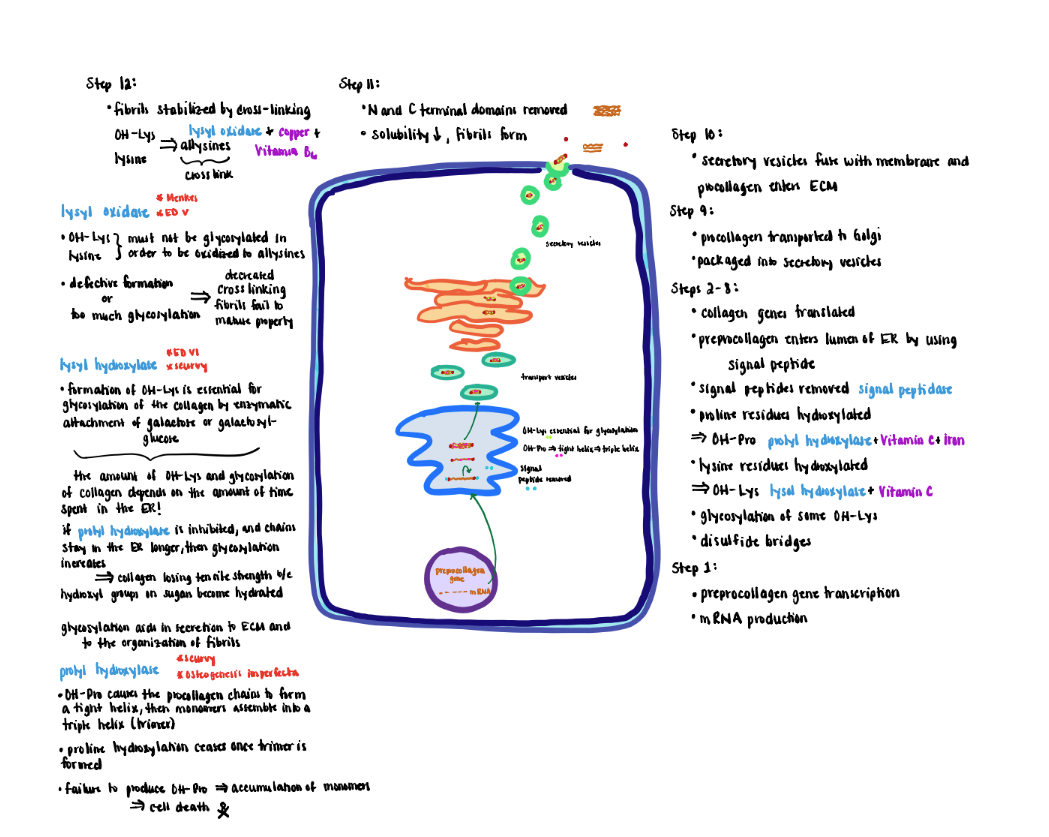
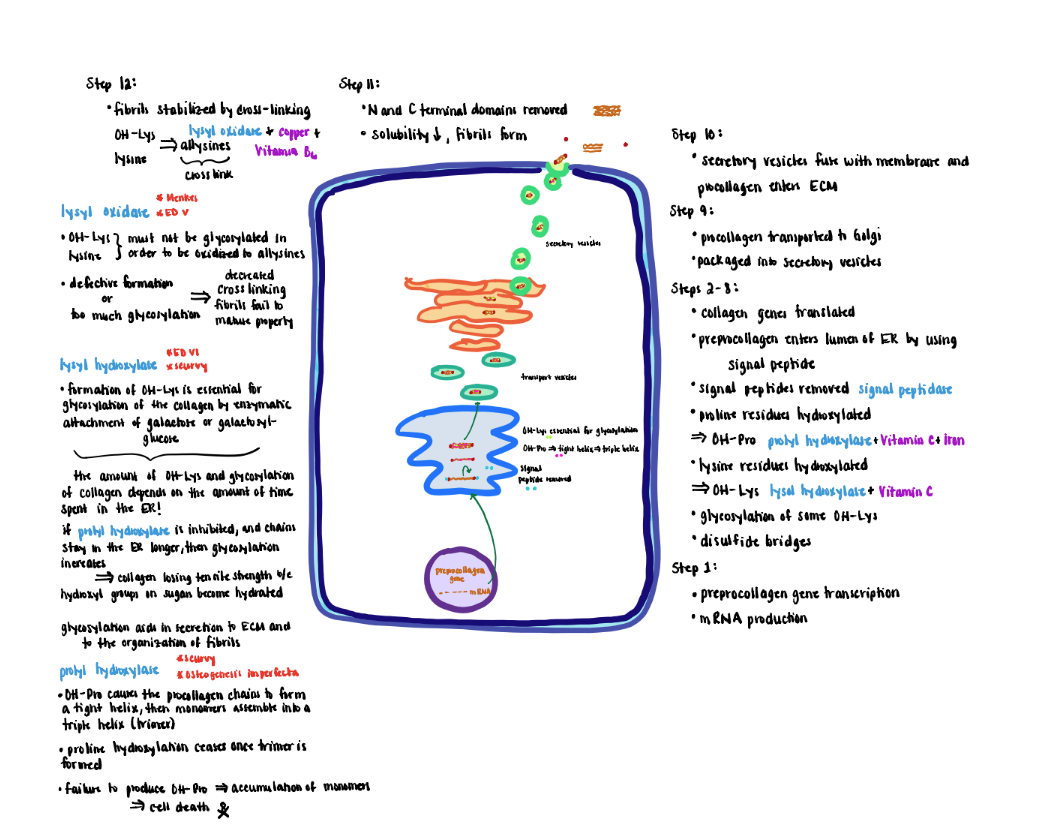
Collagen Processing-Extracellular (Steps 11-12)
Extracellular Processing:
1) After secretion, collagen is further modified by removal of the globular cysteine-rich Nand C- terminal domains. This modification decreases the solubility 1000-fold, causing the collagen units to form spontaneously into fibrils.
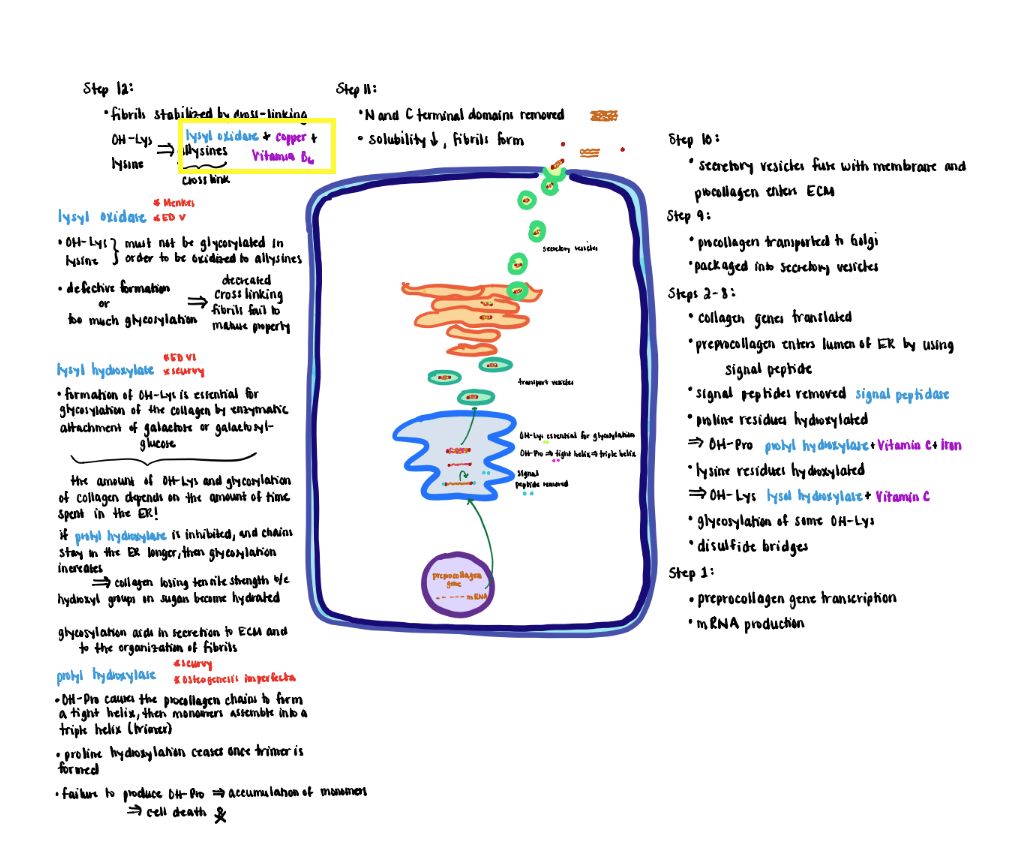
2) Newly formed fibrils are covalently stabilized through crosslinking potentiated by the action of lysyl oxidase.
- The primary substrate for lysyl oxidase is OH-lys and as well as lysine residues although to a lesser extent. In either case the OH-lys or lysine must lack glycosylation.
- Oxidation converts the residue sidechains from amines to active aldehydes known as allysines.
- Lysyl oxidase (do not confuse with lysyl hydroxylase) requires copper and vitamin B6 as cofactors.
- Reaction of allysine with free amino groups on other amino acid residues (e.g, unmodified lysine, hydroxylysine or allysine) eventually forms stable covalent crosslinks that are vital to the structure of mature collagen fibers.
- Defective formation of OH-Lys diminishes markedly the extent of cross-link formation so that fibers fail to mature properly
Diseases Affecting Lysyl and Prolyl Hydroxylase
- Scurvy
- Ehlers-Danlos Syndrome Type VI
- Ehlers-Danlos Syndrome Type V
- Menkes Disease
- Osteogenesis Imperfecta
- Marfan Syndrome
- Alport Syndrome
Scurvy
- Scurvy is associated with a deficiency of ascorbic acid (vitamin C), which serves as a cofactor for the activity of both the prolyl and lysyl hydroxylases and reduces hydroxylation of both proline and lysine.
- Consequently, collagen trimer formation and cross-linking are abnormal leading to weakened blood vessels and the characteristic bleeding gums.
Ehlers-Danlos Type VI
- Ehlers-Danlos Syndrome (EDS) type VI is caused by a defect of the lysyl hydroxylase enzyme.
- Major symptoms include hyperextensible joints and eye defects.
- The product of this reaction, hydroxylysine, is a substrate for the lysyl oxidase reaction. Therefore, patients with EDS type VI will show some symptoms apparent in patients with a defect of lysyl oxidase.
Ehlers-Danlos Type V
- Lysyl oxidase is defective in patients with EDS type V. Though hydroxylysine can be formed, it cannot be oxidized.
- Major symptoms of this disease include thin skin and cardiac valves, with frequent hernias.
Menkes Disease
•an X-linked recessive disorder, results in defects of a protein that actively transports copper across cell membranes.
- Thus these patients exhibit poor distribution of copper to the body’s cells.
- Copper is a cofactor for numerous enzymes, including lysyl oxidase.
- Reduced copper leads to reduced activity of lysyl oxidase and, consequently, reduced collagen crosslinking and stability.
- Symptoms include sparse, kinky hair, growth retardation, deterioration of the nervous system, and hypotonia.
Osteogenesis Imperfecta
•Osteogenesis imperfecta (OI), also known as Brittle bone disease, Fragilitas ossium, and Vrolik disease, is a consequence of mutations primarily in the genes that encode for the proalpha-1(I) [COL1A1] and pro-alpha-2(I) [COL1A2] chains.
-These defects affect type 1 collagen, the most abundant in the body and found in skin, tendon, arterial walls, and bone.
- The most common form is autosomal dominant with more than 100 mutations identified in these genes causing disease that range from mild to lethal.
- Even if only one abnormal chain is incorporated into the collagen trimer, normal folding is prevented.
-Since the cell views this misfolded trimer as abnormal, all the chains are degraded by cell proteolytic processes.
•Manifestations include fractures, blue sclerae due to translucent connective tissue over choroidal veins, tooth abnormalities, and hearing loss.
Marfans Syndrome
- Elastic fibers in connective tissues provide resilience and elasticity. The protein elastin constitutes the majority of elastic fibers.
- During fiber formation, elastin binds to other structural extracellular matrix proteins, including fibrillins.
-Fibrillin proteins assemble in microfibrils that form an outer mantle on mature elastic fibers.
- Marfan syndrome is an autosomal dominant connective tissue disorder that results from mutations in the gene that encodes for fibrillin-1.
- Reduced functional fibrillin-1 results in decreased formation of microfibrils leading to diminished tissue elasticity and stability.
- Marfan syndrome symptoms include vision problems due to dislocated lens and heart problems due to defects of the aorta.
Alport Syndrome
- Defect in type IV collagen: a primary component of basement membrane/basal lamina of the kidney X linked inheritance pattern
- glomerulonephritis, hearing loss, and eye abnormalities


