What are cofactors?
Differentiate.
needed by some enzymes in order to function
- prosthetic groups: organic mol. covalently bound to an enzyme → non-dissociable
- coenzymes: organic mol. non-covalently bound to an enzyme → dissociable
- metall ions: bound to sec. interactions
What is the native conformation of enzymes?
maximal catalytic activity at distinct ph and temp.
- pH: optimum is NEVER at IP
- T: ↑ T → ↑ Ekin → ↑ activity, but if T too high, denaturation
How do enzymes change reaction kinetics?
change reaction mechanism
BUT: catalyze in both direction
- decreased Ea
- unchanged ΔG
- unchanged EQ-state, but decreased time to reach EQ
- unchanged thermodynamics (half life, reaction order, etc.)
REMEMBER diagram
What is enzyme activity?
Formula + unit.
amount of substrate converted to product per unit time
EA = -Δ[S]/Δt = Δ[P]/Δt
measured in [Catal] = [mol/sec], [μmol/min]
What is specific enzyme activity?
Unit.
amount of product formed in a given amount of time per mg of total protein
→ enzyme activity per per mg of total protein
in [kat/mg]
Define turnover number.
Formula + unit.
max. amount of products that is formed by a single catalytic site per second
kcat = np / nE * t
in [mol(P) / mol(E)*sec]
What is an important medical application of enzyme activity measurements?
measurement of non-functional plasma enzymes
⇒ marker enzymes for blood plasma indicate died tissue cells → information abt tissue problems
ex: after heart attack
- creatine kinase CPK
- lactate dehydrogenase LD
- α-hydroxbutyrate dehydrogenase HBDH
Explain the 2 different enzyme models.
enzyme-substrate complex ES stabilized by sec., ionic, covalent bonds
lock and key model:
S + E have specific shape → fit exactly into one another
induced fit model:
E changes slightly its shape as S binds
Distinguish btw different enzyme mechanisms.
- catalysis by proximity: enzyme brings together substrates in proper orientiation, incr. the prob of succesfull collisions
- acid-base catalysis: enzyme transfers H+/OH- from one substrate to another
- covalent catalysis: enzyme forms covalent bonds substrates, providing an energetically more favorable reaction partner
What are proteases?
What is the most important group among proteases?
List some examples.
cuts proteins by hydrolysis of peptide bonds
Ser-proteases = Ser in active center
- trypsin, chymotrypsin
- elastase
- thrombin + other blood coag. factors
What is DIPF?
Why is it pretty cool?
diisopropylphosphofluoridate
- *irreversible inhibitor of Ser-enzymes**
- (ex: chymotrypsin + acetylcholinesterase)*
→ used as biochemical weapon
What are zymogens?
proenzymes = inactive enzyme precursors
What are trypsinogen and chymotrypsinogen?
Explain their proteolytic activation pathway.
trypsinogen + chymotrypsinogen stored as zymogens to protect pancreas from autodigestion
- activation of trypsinogen → trypsin
- activates chymotrypsinogen → π-chymotrypsin
- cut each other into shorter fragments → α-chymotrypsin = active peptidase
- trypsin inactivated by inhibitor → chymotrypsinogen activation ceases
What is the catalytic triad in case of chymotrypsin?
What type of catalysis is it?
proton shuttle in active center of Ser-proteases
here: 102Asp 57His 195Ser
→ covalent catalysis
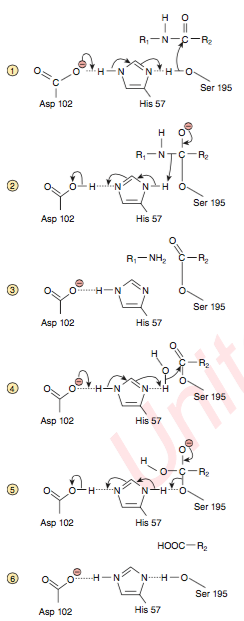
Explain the reaction mechanism of chymotrypsin.
catalysis of peptide bond
- S binds to 195Ser, shifts -OH H+ to 102Asp
- nucleophilic attack of Ser O on C of peptide bond → acyl-enzyme intermediate
- H+ shuttled to peptide → H2N-R pinches off (= 1st product) when replaced by H20
- H20 withdraws H+ to 102Asp
- OH- attacks acyl-enzyme intermediate → H+ shuttled back to 195Ser
- original conformation restored → COOH-R pinches off (= 2nd product)
Explain the specificity of the Ser-proteases chymotrypsin, trypsin and elastase.
depends on R-side chain
- chymotrypsin: big, non-polar pocket
→ only aromatic rings: Phe, Tyr, Trp - trypsin: negatively charged pocket
→ only pos. charged: Arg, Lys - elastase: small pocket
→ only small -R: Gly, Ala, Ser
REMEMBER: chY, tYr. And just know the rest
What is the vi of enzyme kinetics?
Which conditions must be fulfilled in order to measure it correctly?
initial reaction rate
rate (approx. linear) where highest amount of products is formed by enzyme
conditions for the measurement:
- rel. short time
- molar excess of S over E
- reverse reaction negligible
What is the Michaelis-Menten equation?
Consequences?
describes enzyme kinetics
→ the lower KM, the greater vi
consequences:
- low substrate conc: vi = vmax*[S]/KM
- substrate conc = Km: vi = vmax/2
- high substrate conc: vi approaches vmax
→ all E in ES complex
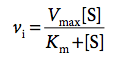
What does the Michaelis constant describe?
- low KM = enzyme has high affinity for substrate
- high KM = enzyme has low affinity for substrate
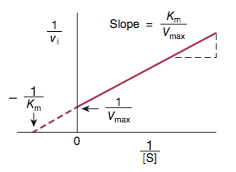
What is the Lineweaver-Burk plot?
Another name.
double reciprocal plot
instead of Vi vs [S], 1/Vi vs 1/[s]
- y-intercept = 1/Vmax
- x-intercept = -1/Km
- slope = KM/Vmax
⇒ used to determine KM, Vmax
What are isoenzymes?
Characteristics?
enzymes that differ in AA sequence but catalyze the same reaction
- coded by diff. genes
- may have diff. kinetic parameters (v, KM)
- same substrate specificity, BUT may also catalyze other substrates
- activated under diff. circumstances
Compare the Km of btw glucokinase and hexokinase.
- hexokinase: always active to supply O2 dependent tissues w/ glucose (↓ KM → ↑ affinity)
- glucokinase: only active when hyperglycaemia, e.g. after meal (↑ KM → ↓ affinity)
What is lactate dehydrogenase?
tetrameric enzyme consisting of 2 monomer types:
H (heart) and M (muscle) → 5 LDH isozymes:
- HHHH predominates in heart tissue
- HHHM, HHMM, HMMM,
- MMMM in the liver
→ LDH levels rise in blood plasma, injured tissue type can recognized by isozyme pattern (marker enzyme)
Differentiate btw mechanisms of irreversible inhibitors like poisons.
Examples.
- action on active center: covalent bond on act. center (e.g. DIPF)
- nonspecific inhibitors: denaturation (e.g. heavy metals, heat)






