1
Q
Rate of reaction
A
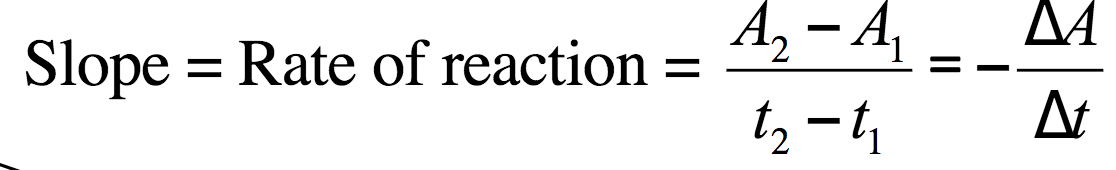
2
Q
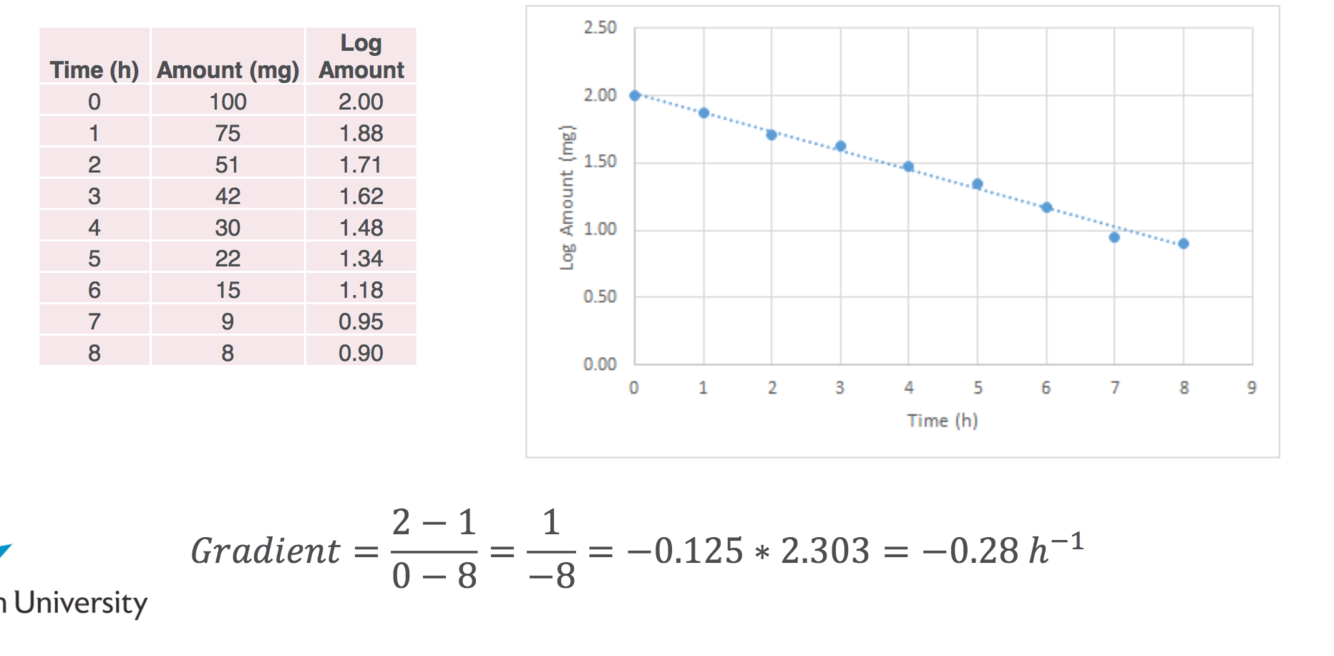
FIRST ORDER REACTIONS- WORKING OUT SLOPE OR RATE OF REACTION
A
3
Q
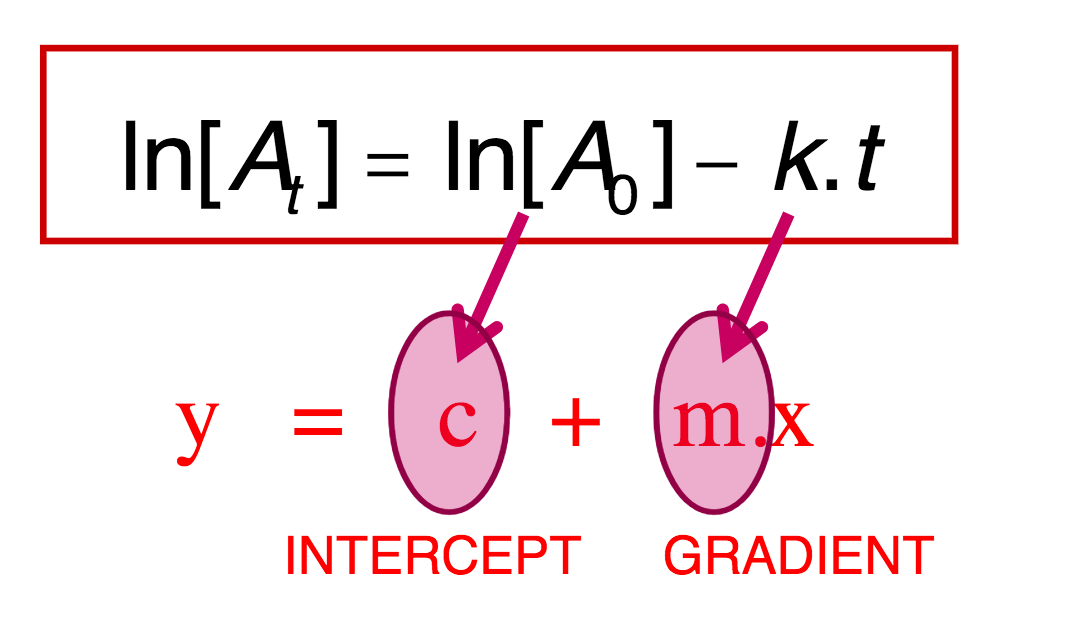
FIRST ORDER REACTIONS- WHEN USING Ln(At)- to work out rate of reaction (slope -k) - integrated rate law
A
4
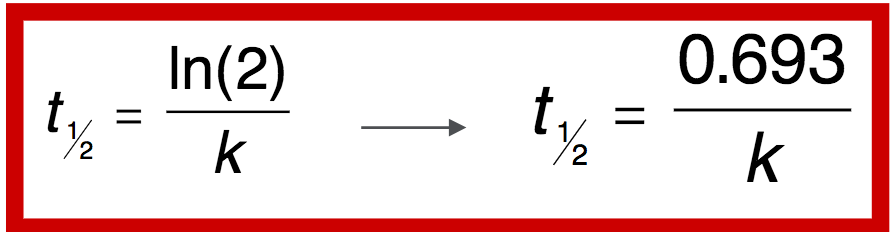
Q
Half life
A
5
Q
Shelf life- t90%
A

6
Q
Zero order- rate equation
A

7
Q
Half-life 0 order
A
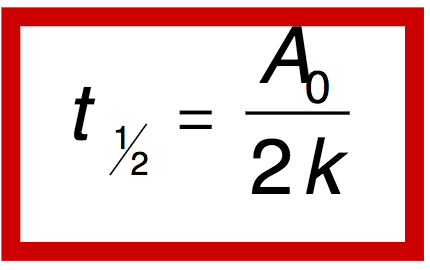
8
Q
0 order- 2nd half life
A
9
Q
0 order- shelf life- t90%
A
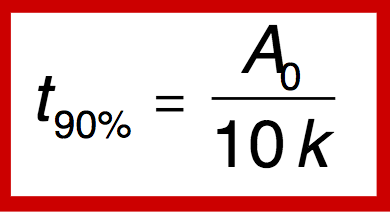
10
Q
First order- arrhenious equation- linear form (stability and temperature)
A

11
Q
Temperature- arrhenious equation
A

12
Q
A
13
Q
Temperature arrhenious equation
A
14
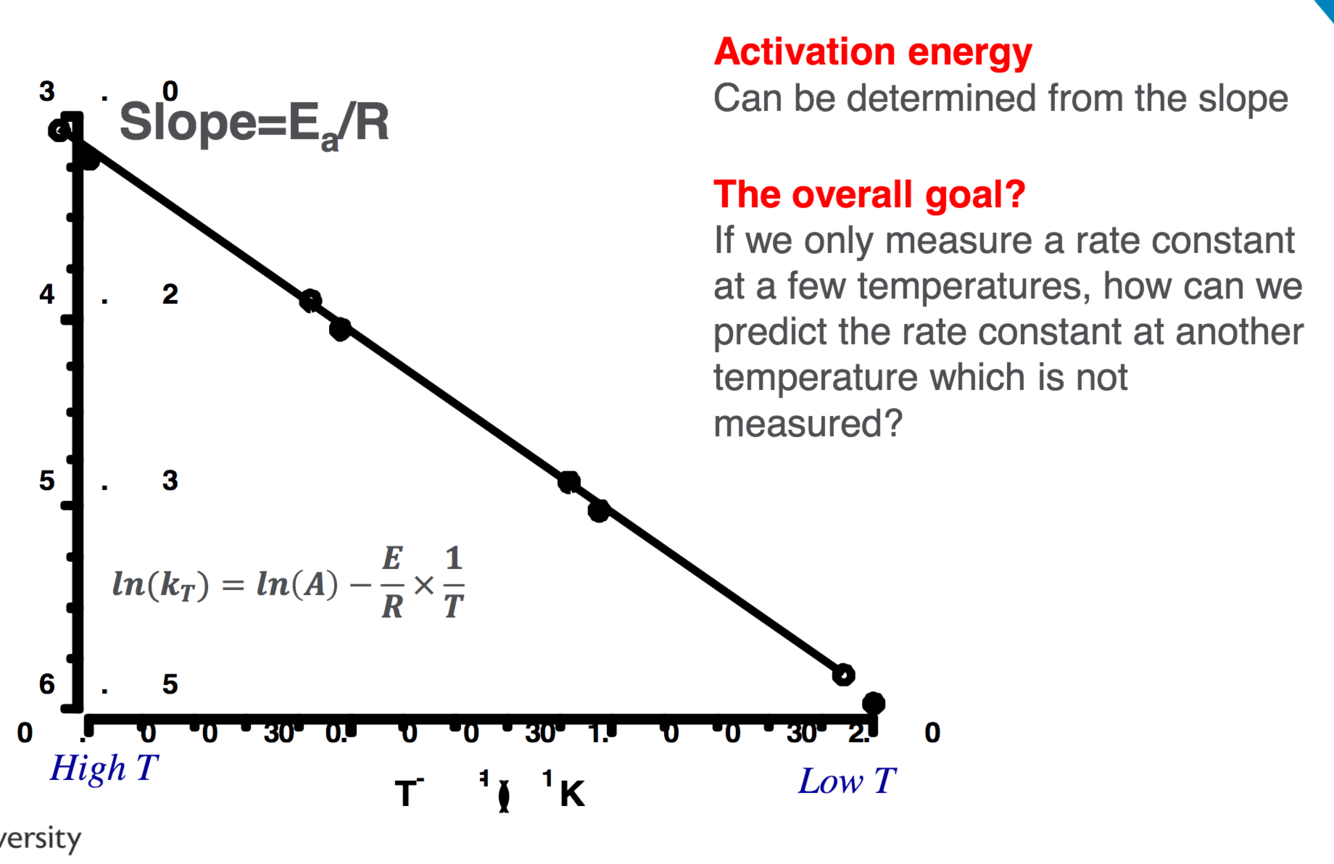
Q
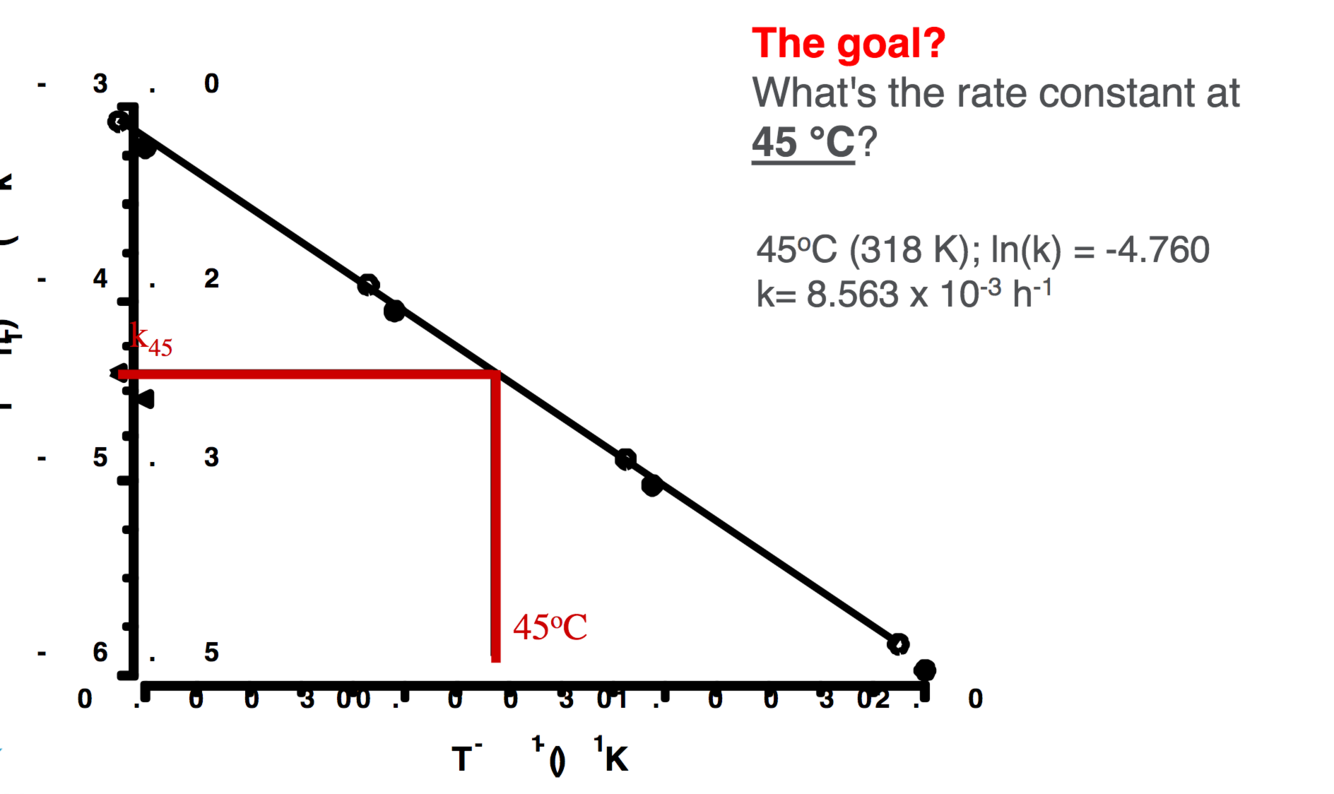
Temperature arrhenious equations- graph how to work out k at certain temperatures
A