What is molarity?
M = moles solute / Liters solution
- One of the five concentration units
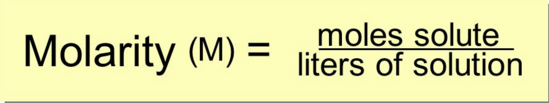
What is mass percent?
% = grams solute / grams solution * 100
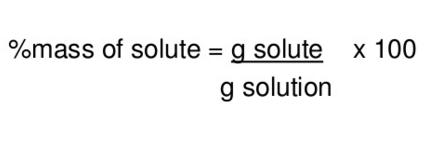
What is mole fraction?
χ = moles solute / moles solution
- All the mole fractions should add up to 1

What is molality?
m = moles solute / kg solvent
- Independent of Temperature, unlike molarity
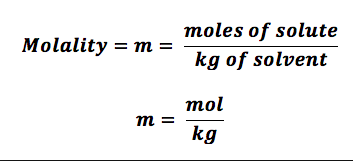
What is parts per million?
ppm = grams solute / grams solvent * 1,0000
or ppm = mg solute / kg solution
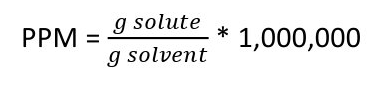
What is benzene?
C6H6
Example of a molecular solid, held together by dispersion forces (nonpolar)
In what four ways can solids be classified?
- Ionic
- Molecular
- Ionic and molecular solids are held together by IMF
- Metallic
- electrons exist in molecular orbitals that are delocalized over many atoms, producing an electronic band structure
- Network
- networks or chains of atoms or molecules held together by covalent bonds
- diamond, graphite
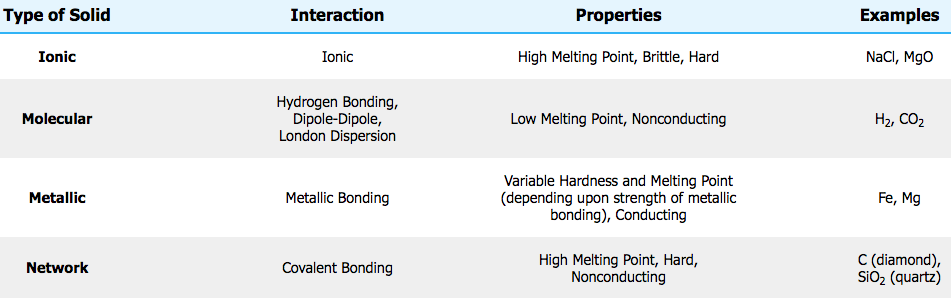
What is the effect of Temperature on semiconductor conductivity?
- Direct: conductivity increases with temperature because more electrons are able to move up to the conduction band
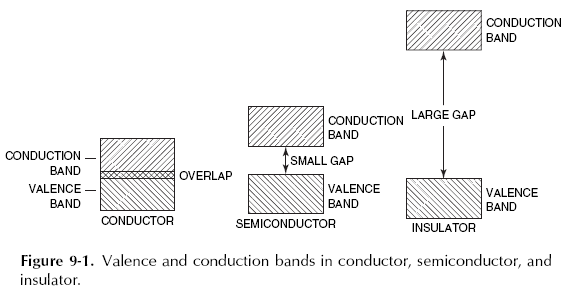
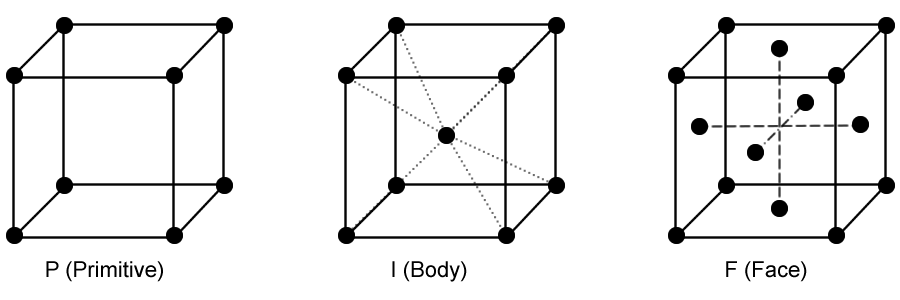
Face-centered cubic cell: how many atoms, and how can you calculate the atomic radius?
- 4 atoms
- hypotenuse = 4r
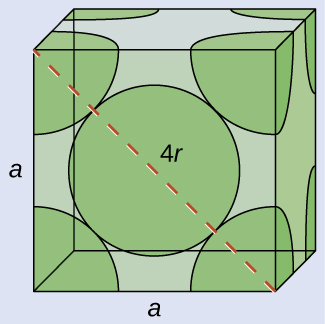
Body centered cubic cell: how many atoms are there,
and how can you calculate atomic radius (given density)?
- 2 atoms
- 4r = edge√ 3
Primitive cubic cell: how many atoms,
and how can you calculate the atomic radius given the density?
- 1 atom
- 2r = edge
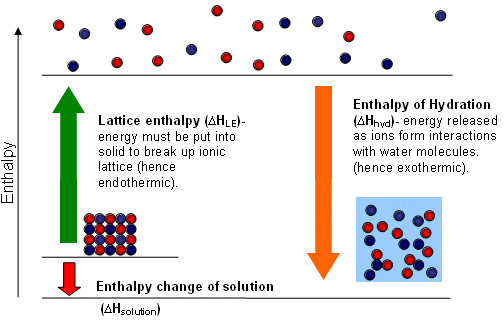
How can you calculate overall enthalpy of solution?
- ΔsolutionH = ΔhydrationH - ΔlatticeH
- Hydration enthalpy is the energy released when bonds are formed between the ions and water molecules (negative)
- Lattice enthalpy is the energy released when gaseous ions bind to form an ionic solid (negative)
- (Subtract this because you have to break the lattice)
How do solute-solvent interactions determine solubility?
- The stronger the attractions between solute and solvent molecules, the greater the solubility
- Like dissolves like
- Substances with similar intermolecular attractive forces tend to be soluble in one another.
- Nonpolar substances are more likely to be soluble in nonpolar solvents
- Ionic and polar solutes are more likely to be soluble in polar solvents
- Network solids are not soluble in either polar or nonpolar solvents because of the strong bonding forces within the solid
- Substances with similar intermolecular attractive forces tend to be soluble in one another.
How does pressure affect solubility of a gas?
- The solubility of a gas in any solvent is increased as the pressure of the gas over the solvent increases
- (Whereas the solubilities of solids and liquids are not affected by pressure.)
- Henry’s law

What is Henry’s Law?
- The solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the liquid
- Sg = KH * Pg
- Where KH is Henry’s constant
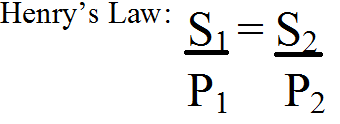
What is Raoult’s Law?
What do positive and negative deviations mean?
- Vapor pressure depression: VP lowers as you increase the mole fraction of a solute
- Multiply pure pressure by the mole fraction of solute
- Psoln = XsolventPosolvent
- Ptotal = XAPoA + XBPoB …
- Multiply pure pressure by the mole fraction of solute
- Only works for ideal mixtures, where there are no IMFs between the particles
- If there is a positive deviation from Raoult’s law (higher vapor pressure), that’s because molecules are breaking away more easily than they do in the pure liquids. The IMFs are less than they are in the pure liquids.
- If there is a negative deviation from Raoult’s law, new stronger forces must exist in the mixture than in the original liquids.

How does temperature affect solubility for solids vs. gases?
- The solubility of most solid solutes in water increases as the temperature of the solution increases
- Whereas the solubility of gases in water decreases with increasing temperature
- Because more gas escapes from solution
What is the van’t Hoff factor?
- i= the number of particles a formula breaks up into
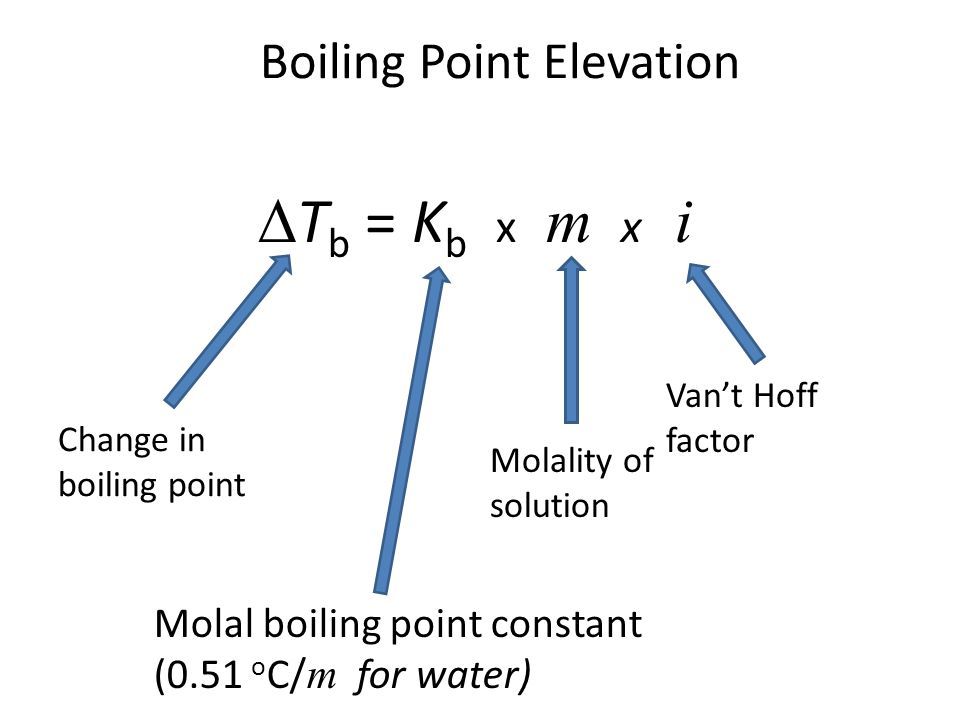
What is Boiling Point elevation?

- The addition of a nonvolatile solute causes the boiling point of the solvent to increase (and the freezing point of the solvent to decrease)
- Calculate the change with FIKm
- molality!
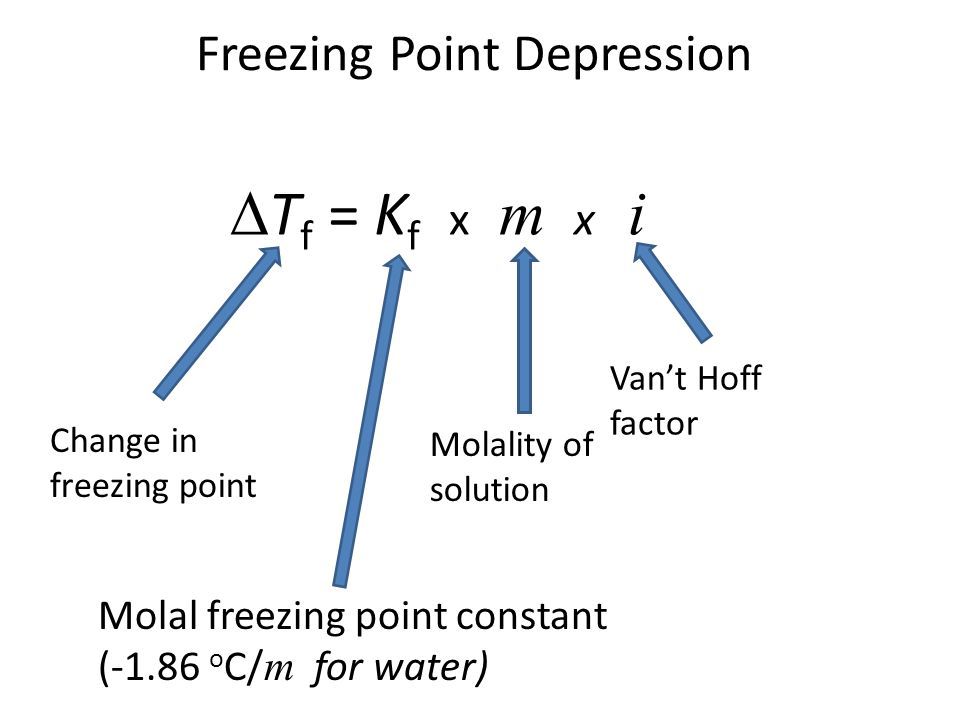
What is Freezing Point depression?
- The addition of a nonvolatile solute causes the freezing point of the solvent to decrease
- Calculate change with BIKm
- molality!
What is Osmotic Pressure elevation?
- The osmotic pressure of a solution is the pressure needed to stop the flow of solvent across a semipermeable membrane
- Increases with concentration of the solute
- Calculate osmotic pressure with PIMRT
- MOLARITY!

With the addition of a solute,
what’s elevated and what’s depressed?
- Boiling Point and Osmotic Pressure are elevated
- Freezing Point and Vapor Pressure are depressed
What are colligative properties?
- Colligative properties of solutions are properties that depend upon the concentration of solute molecules or ions, but not upon the identity of the solute
- Vapor pressure depression, boiling point elevation, freezing point depression, and osmotic pressure elevation
What are the 3 ways a reaction rate can be expressed?
- Initial rate of reaction
- Average rate of reaction
- Instantaneous rate of reaction



