What is the equation for energy change?
(first law of thermodynamics)
- ΔE = q + w
- E (or U) stands for internal energy (total energy, kinetic + potential)
- First law of thermo: Internal energy can be transferred as heat or work, but not created or destroyed.
- Work is organized energy. Heat is chaotic energy.
- Within work, there is PV work (expansion of a gas), and non-PV work (electrical, free energy).
- Work is organized energy. Heat is chaotic energy.
- When volume is constant: ΔE = qv
- Heat at constant volume. (Bomb calorimetry gives this.)
What is enthalpy and its equations?
- Just like energy change, but for reactions under constant pressure
- ΔH= qp
- heat at constant pressure. (Solution calorimetry gives this.)
- To calculate the difference between energy and enthalpy:
- ΔH= ΔE + PΔV
- can also be written as ΔH= ΔE + ΔngasRT
What is a spontaneous process, and where does it take us?
- A reaction that occurs by itself, and continues without outside assistance
- It takes us to equilibrium, Q=K
- Ex: adding or removing a reactant or product, or changing volume or temperature is spontaneous. Le Chatlier’s principle.
- It takes us to the minimum free energy, ΔG=0
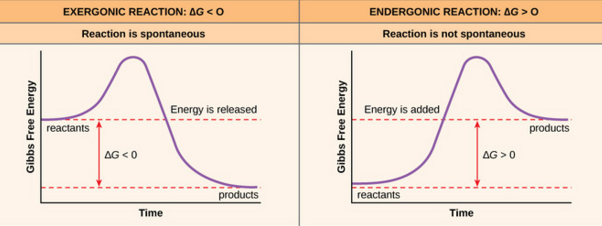
- Most exothermic reactions occur spontaneously
- Most reactions that increase entropy will occur spontaneously
- But either the enthalpic or entropic term can take over, so if possible calculate ΔG = ΔH−TΔS
What drives nonspontaneous reactions?
- Spontaneous reactions, some source of energy
- Ex: photosynthesis needs sunlight
What is entropy (S) and equations for it?
- A state function for the randomness/dispersal of energy, a measurement of disorder in a system
- positive ΔS = increase in entropy of a system
- ΔS = Sfinal – Sinitial
- Two ways to think about entropy:
- Statistically, the more ways energy can be distributed in a system, the higher the entropy
- S = k * lnW
- where k = Boltzman’s constant, and W = the number of microstates, the ways of distributing energy, Ω. Plug in.
- S = k * lnW
- How much heat is transferred. (Heat is chaotic energy whereas work is disorganized energy)
- ΔS = qrev/T or ΔS = ΔH/T
- Statistically, the more ways energy can be distributed in a system, the higher the entropy
- Sidenote: We can get an absolute value for S, not just its change (unlike ΔE and ΔH)
- Common units for S: J/mol*K
What factors influence the magnitude of entropy, and how?
ΔS = Sfinal – Sinitial
- Volume: For gases, entropy increases with volume.
- When Δngas is positive, so is the entropy change
- Temperature: Entropy increases with temperature
- Heat: Entropy increases with heat.
- At one temperature, the amount of heat transferred in a reversible process is your change in entropy. (Applies to phase change.)
- Physical state: the greater freedom of molecular movement, the higher the entropy
- # Particles: Entropy increases with the number of particles in a system, all else being equal
- Molecular complexity: Entropy increases with complexity
- Especially without doube bonds
- Ex: beta carotene, a long molecule, has more ways to distribute energy by wiggling around
- Forming mixtures
What three factors can influence spontaneity?
ΔG = ΔH - TΔS
- Enthalpy change
- Generally, enthalpy has the greater influence on spontaneity. (ΔH is in KJ/mol, ΔS in J/Kmol.)
- Entropy change
- Entropy affects spontaneity. Entropy of the UNIVERSE predicts spontaneity.
- Temperature
- The free energy of the system changes with temperature
What is entropy change for the universe?
(2nd law of thermodynamics)
ΔSouniverse = ΔSosystem + ΔSosurroundings
- A spontaneous change increases the entropy of the universe
- A process is considered to be spontaneous under standard conditions if ΔSouniverse > 0
- Calculate entropy change of universe:
- ΔSosurroundings = -ΔHosystem/T ADD TO
- ΔSosystem
What is the Gibbs free energy function?
ΔG = ΔH - TΔS
Good Honey Tastes Sweet
- Combines enthalpy and entropy into a state function
- ΔG = Gfinal - Ginitial
- At constant temperature and pressure, a change can only be spontaneous if ΔG is negative
- G, the free energy, is the amount of electrical work a system can do
- Can be measured under any set of conditions. If the data are collected under standard-state conditions, the result is the standard-state free energy of reaction (ΔGo).
What is the difference between exergonic and endergonic?
- Exergonic: free energy decrease ΔG<0
- Endergonic: free energy increase ΔG>0
(extra vocab from book)
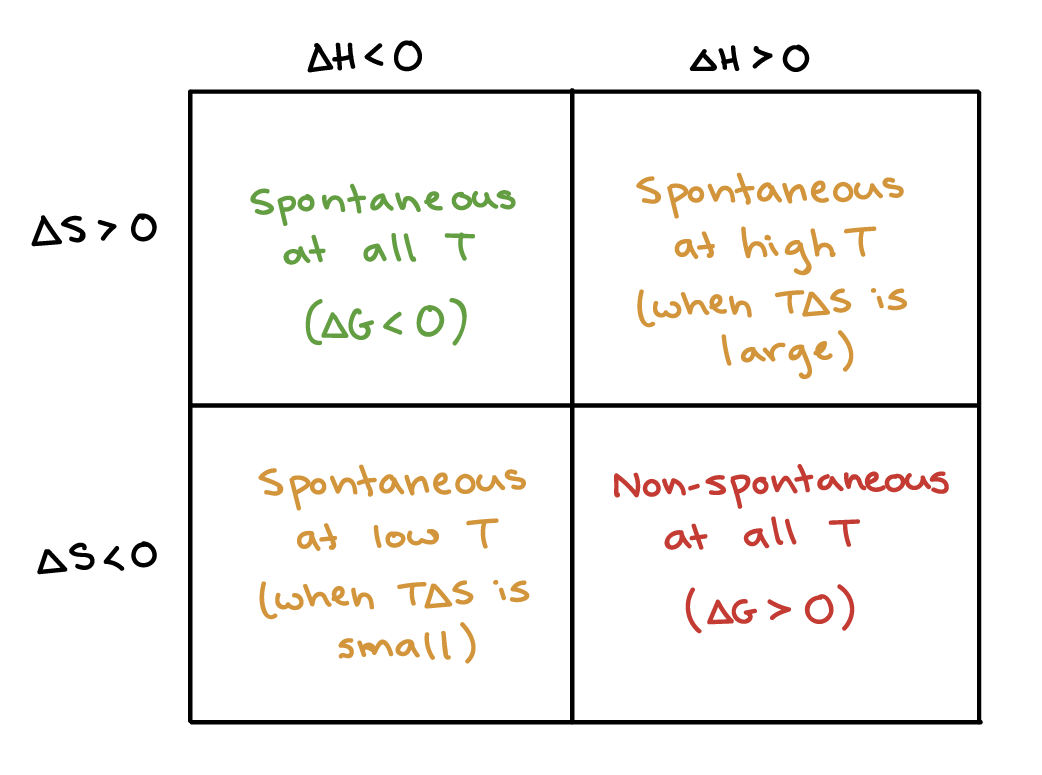
- Determine spontaneity when:
- ΔH and + ΔS
- +ΔH and - ΔS
- ΔH and ΔS have same sign
ΔG = ΔH - TΔS
- When ΔH is negative (exo) and ΔS is positive, always spontaneous
- When ΔH is positive (endo) and ΔS is negative, never spontaneous
- When ΔH and ΔS are both positive, it will be spontaneous only at high temp
- When ΔH and ΔS are both negative, it will be spontaneous only at low temp
- When one is favorable and one isn’t, spontaneity will depend on temperature. When both are unfavorable, not spontaneous at any temperature.
What is the third law of thermodynamics?
- The entropy of a perfect crystal at 0K, is 0.
- But that doesn’t exist. So all compounds in the world have positive molar entropy values

What is standard entropy,
and how do you calculate entropy change in a reaction?
- The standard molar entropy, S°, of a substance is the entropy gained by converting 1 mol of it from a perfect crystal at 0 K to standard state conditions at the specified temperature.
- All substances have positive entropy values at temperatures above 0 K. Tabulated.
- Calculate standard molar entropy of a reaction by balancing chem equation, and plugging into Sproducts - Sreactants.
- Don’t forget to multiply entropies by stoich coefficients, and look at the correct phase

Give an example of a process that is:
- Exothermic and spontaneous
- Endothermic and spontaneous
- Exothermic and nonspontaneous
- Endothermic and nonspontaneous
- Exothermic and spontaneous
- iron rusting
- Endothermic and spontaneous
- cold pack - dissolving ammonium nitrate in water
- Exothermic and nonspontaneous
- combustion in engine, requires a spark
- Endothermic and nonspontaneous
- photsynthesis, requires sunlight
- So although exothermic processes tend to be favorable, they aren’t always spontaneous
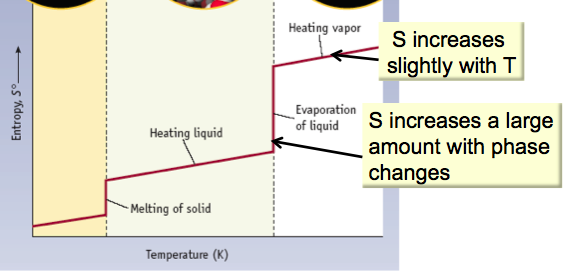
What is the entropy change at a phase change?
Graph and give equation.
- Entropy increases slightly with temperature
- At a phase change, the temperature is constant (isothermal process), but there is a big increase in entropy
- Calculate the entropy change for a phase change:
- ΔS = qrev/T or ΔS = ΔH/T
- ΔH for the phase change is tabulated. Plug in the T at which it’s happening.
- ΔS = qrev/T or ΔS = ΔH/T
If a reaction is spontaneous in the forward reaction,
it is _________ in the reverse.
nonspontaneous
(and vice versa)
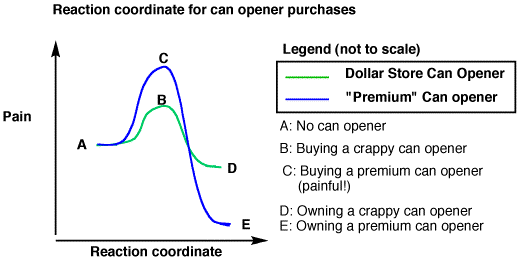
Kinetically favored vs.
thermodynamically favored
- The thermodynamically favored product is more stable, has lower energy
- The kinetically favored product has a lower activation energy, will form faster/more easily
- Ex graph: the premium can opener is thermodynamically favored. The cheap one is kinetically favored.
- Bottom line: thermodynamics has nothing to do with the rate of a reaction
- Even if something is spontaneous, it’s not necessarily going to be fast
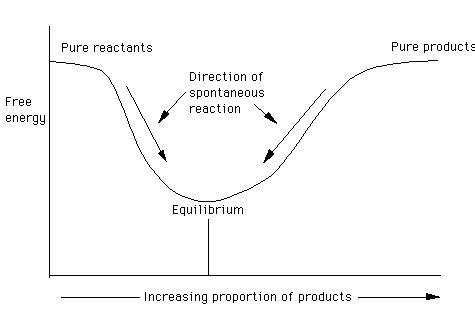
A reaction proceeds spontaneously towards what?
- Toward the minimum in free energy, which corresponds to equilibrium
- Hint: slide down the curve
Connect ΔG to equilibrium
- ΔG (not standard, any conditions) can be spontaneous in forward or reverse direction
- If it’s not spontaneous in either direction, you’re at equilibrium.
- When Q=K, you’re at equilibrium, which means that you are at the minimum of free energy that your system can be at, ΔG = 0
- At equilibrium, you can solve for K using this equation -

What is turnover temperature, and how do you find it?
How does this differ from phase change?
- Turnover is the temperature at which a reaction flips to spontaneous or nonspontaneous
- Using ΔG = ΔH - TΔS, set ΔG = 0
- The answer is given as a range - above or below a certain threshold
- For a phase change, also set ΔG = 0
- Can do this because during a phase change, equilibrium exists between phases, so G = 0
- However, the answer is just that particular temperature (not a range)
What does the sign of ∆G° indicate?
∆G° = – RT ln K
- The ratio of the amount of products to reactants at equilibrium
- The thermodynamic favorability of the reaction
- If ∆G° is negative at equilibrium, then we will have lots of products at equilibrium, K>1
- If ∆G° is positive at equilibrium, then we will have lots of reactants at equilibrium, K<1
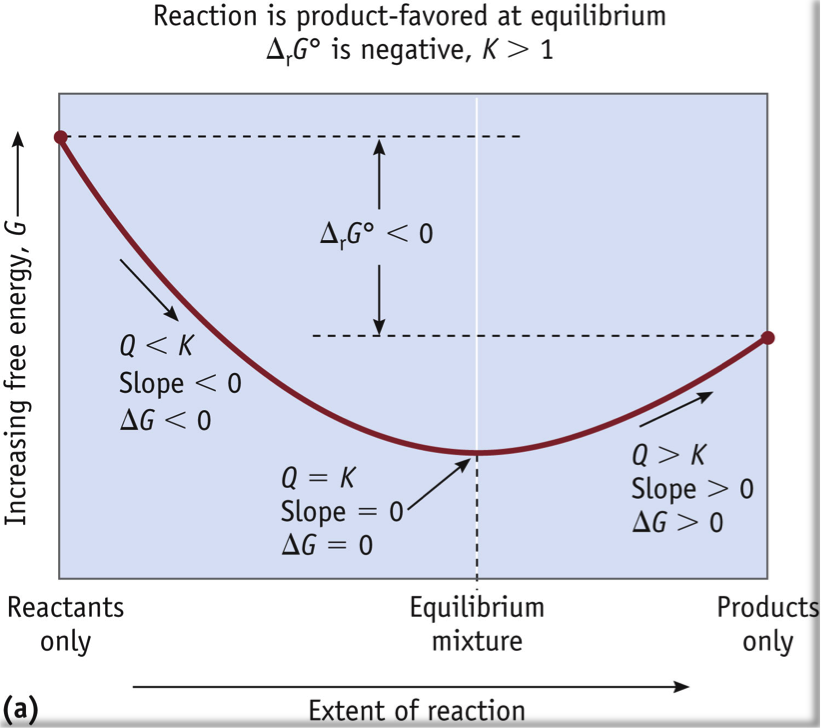
What does the sign of ∆G indicate?
- ∆G = 0 at equilibrium. (NOT ∆G°)
- Since with ∆G we are NOT necessarily at equilibrium, the sign of ∆G can be thought of as a predictor about which way the reaction will go.
- If ΔG is negative, the reaction is moving forward towards product. (Slide down, forwards.)
- If ∆G is positive, then to allow ∆G to reach zero, the reaction will need to from more reactants. (Slide down, backwards.)
- Look at the slope of the curve for the sign of ∆G, or use this equation to solve:
- ∆G = ∆G° + RT ln Q
For a pure element, what is ΔGoformation, ΔHoformation, and ΔSoformation?
- ΔGo and ΔHo are 0
- ΔSo has its own value
Enthalpy driven vs. entropy driven
- Enthalpy driven if exothermic
- Enthalpically favored
- Entropy driven if an endothermic reaction occurs because of a highly positive ΔS
- Entropically favored
- Ex: solution of ammonium nitrate
- Use the Gibbs equation to determine the relative importance of the enthalpy and entropy terms as driving forces behind a particular reaction
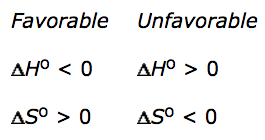




ΔGo is negative (the difference between all products and all reactants) so product favored



