Alkynes has the prefix _______
yne
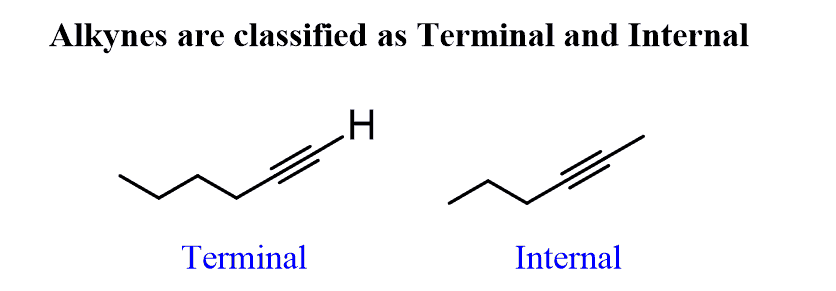
Terminal alkynes
Monosubstituted acetylenes
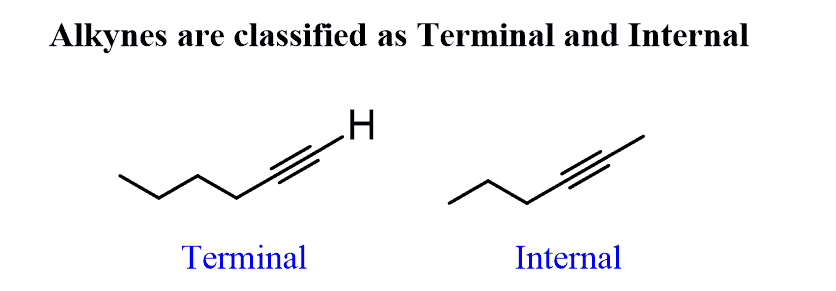
Internal Alkynes
Dissubstituted acetlyenes
What are the bases used to protonate an acetylene or terminal alkynes?
- CH9-
- H2N
- H-
- Acetylide ion
When preparing alkynes they can be formed from dihalides via ___________
Strong base
Alkyl dihalides can be turn into alkynes via _______ twice
E2 reactioon
Geminal dihalide
Both halogens are connected to the same carbon
Vicinal dihalides
Two halogens are connected to adjacent carbons
_________ & __________ are the bases used for turning alkyl dihalides into alkynes
NaNH2, NH3
A terminal alkyne can be prepared with ___________ soidum amide
Excess (xx)
Sodium amide
NaNH2
Alkynes can undergo catalytic hydrogenation & also can use a poisoned catalyst to covert alkyne into a _______ alkyne
Cis Alkyne
What are the types of poisoned catalyst that can be used to form a cis alkene?
Lindlar’s Catalyst
What is the sterochemistry of catalylic hydrogenation involving alkynes?
Syn Addition
______________ reduction can turn alkynes into trans alkenes
Dissolving metal reduction
What is the reagents used in a dissolving metal reduction?
Na in NH3
What is the mechanism for dissolving metal reduction?
- Nucleophilic attack
- Proton transfer
- Nucleophilic attack
- Proton transfer
A ____________ arrow shows the movement of one electron
Half arrow (Fish hook arrow)
To produce an alkane an alkyne can be treated with _____ & a metal catalyst like Pt,Pd, or Ni
H2
To produce a Cis alkene an alkyne can be treated with H2 & a _________ catalyst
Poisoned Catalyst (like Lindlar’s Catalyst or Ni2B)
To produce a trans alkene an alkyne can be treated with ______ in _______
Na in NH3
Alkynes can undergo hydroboration oxidation, what is the regiochemistry & stereochemistry?
- Regiochemistry is anti-markonvikov addition
- Stereochemistry is syn addition
What is the mechanism for hydrohaolgenation with alkynes?
- Proton transfer
- Nucleophilic attack
When terminal alkyne is treated with ____________ during hydrohalogenation an anti-Markovnikov addition is obsverved where the Br is positioned at the terminal position which produces E & Z isomers
HBr, ROOR (Radical addition can only happen with HBr (Not HCl or HI))


