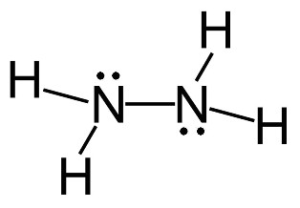
Hydrazine
N2H4
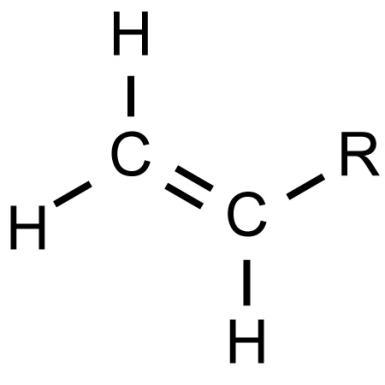
Vinyl
Define observed rotation of a molecule
- What determines it?
degree to which a sample rotates PPL
- determined by:
- []
- length of tube, etc.
Why are alcohols soluble in water?
They can H bond with water
Cycloalkanes and ring strain
- How much ring strain do big, small, bicyclic & monocyclic cycloalkanes have?
- Cycloalkanes w/ LESS than 6 C’s have increasingly more ring strain
- Really BIG (10+) rings have enough freedom to approximate 109.5º
- BICYCLICring systems haveMOREring strain thanMONOcyclic rings
If you see C6H5, what is it?
a BENZENE!
- not a straight chain
- since it has π bonds, it is unsaturated
1º,2º,3º OH groups have 1,2, and 3 ___ ___ which do what to charge?
What does this mean?
Have 1,2, and 3 donating groups to destabilize charge
this is why 3º is less acidic than 1º
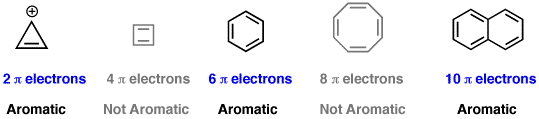
Hückel’s Rule (for aromaticity)
to exhibit aromaticity
a ring must have EXACTLY __+__ electrons
to exhibit aromaticity
a ring must have EXACTLY: 4n+2π electrons
From what I’ve gathered, “n” is just 1 (at least it is for benzene…( 4(1) + 2π= 6 )
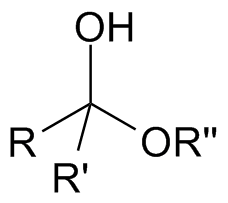
Hemiketal
Physical properties of alkanes
- Soluble/Insoluble in water?
- Low/High density?
- polar/non-polar?
- What increases BP?
- What decreases it?
- What increases MP?
- insoluble in water
- very low density
- non-polar
- BP increases with increasing chain length and/or MW
- BP decreases with increased branching (decreased symmetry)
- MP increases with increasing chain length and/or MW
Cyclic rings create ___ ___ and make bond angles do what?
- ring strain
- make bond angles deviate from preferred 109.5º
Alkenes are _____s!
Why?
Nucleophiles!
- π bonds are e’ dense and will attach E:philes
- forming a new bond to one of the C’s
- and leaving a CCI on the other
- CCI then attacked by a Nu:
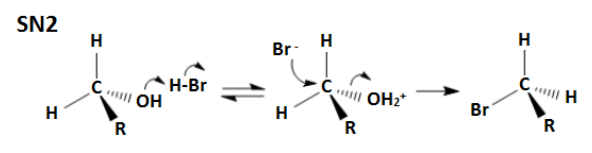
What does an SN2 rxn look like?
Rank BOND REACTIVITY for:
- Single bonds
- Double bonds
- Triple bonds
triple>double>single
more reactive bc of π bonds
3º ROHs can only be oxidized into ___?
PSYCHE!
They cant be oxidized at all!
1º ROH can be oxidized into?
1º ROH⇒Aldehydes⇒Carboxylic Acids
Alcohols are less/more acidic than water
- Rank alcohol acidity
LESS acidic than water
- ROH acidity increases from:
- (most acidic) 1>2>3 (least acidic)
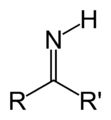
Imine
Hybridization:
sp3, 1 LP
Trigonal Planar
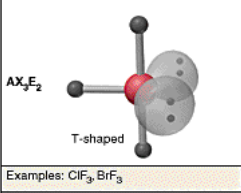
Hybridization:
sp3d, 2 LPs
T-shaped
What is a racemic mixture a mixture OF?
50/50 mix of R and S enantiomers
Anomers
- If anomeric OH/OR group and CH2OH group are on opposite sides of the ring, what kind of anomer is it?
ALPHA anomer
on the “Alpha-site side”
Alcohols behave as either: ___s or as a ___
1) Nucleophiles
* (the lone pair on oxygen acts as a Lewis base)
2) Lewis acids
- ..when they are oxidized to carbonyl groups
- the oxygen ACCEPTS A PAIR OF ELECTRONS (Lewis Acid) from the O-H bond as the proton is abstracted
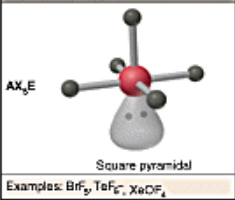
Hybridization:
sp3d2, 1 LP
Square Pyramidal