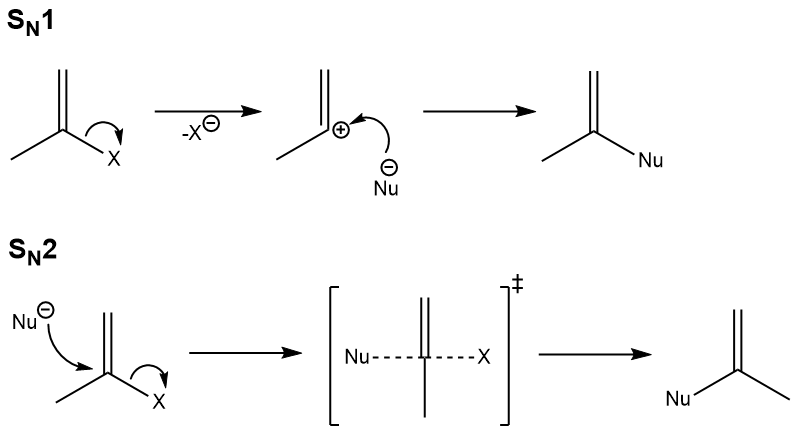
SN1 vs SN2 reactions in nucleophilic substitution:
‘key’ to the mechanism: carbocation stability | steric hindrance
rate law: unimolecular (substrate concentration only) | biomolecular (substrate and nucleus concentrations)
number of steps: 2 | 1
substituents on carbon: 3 > 2 > 1 > 0 | 0 > 1 > 2 > 3
strength of nucleophile: doesn’t really matter | strong and non-bulky
solvent: polar protic | polar aprotic
stereochemistry: not preserved (racemization) | inverted backside attack)
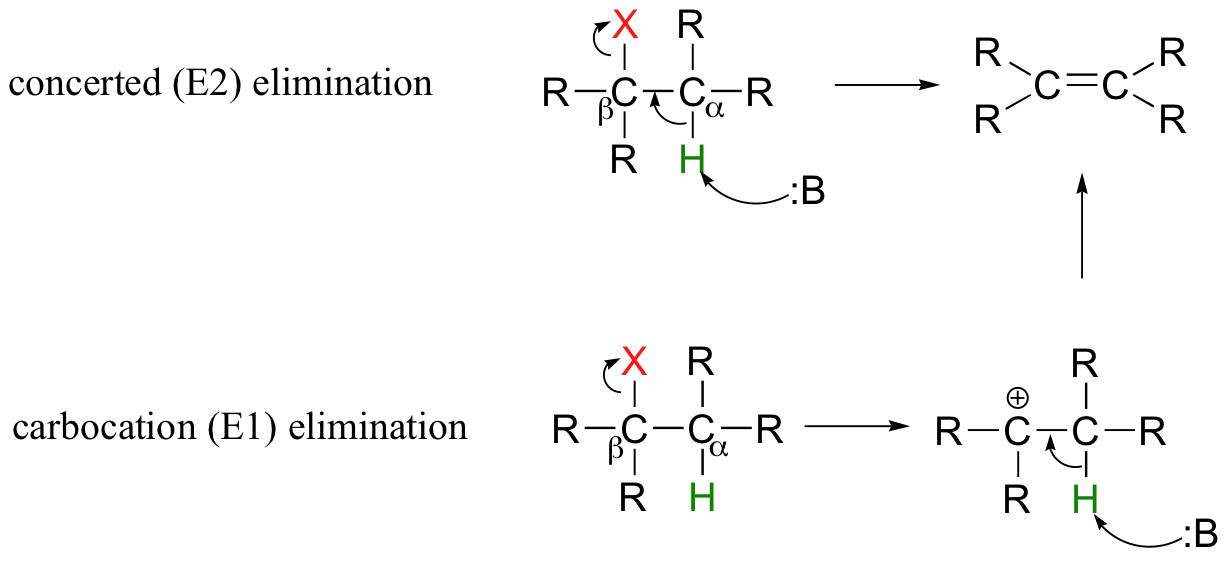
in a reaction with a nucleophile present, alkyl halides can also undergo a type of reaction in which the halide is kicked out, but the nucleophile doesn;t actually add itself as a substituent; instead a double C=C bond is formed. this type of reaction is known as?
- elimination (E1 and E2 mechanisms)
- E1 mechanism is similar to the SN1 mechanism in that it has a unimolecular rate law and is driven by carbocation formation. the difference is that isntead of attacking the carbocation once it is formed, the nucleophile pulls off a proton from an adjacent carbon, which allows a double bond to be created on the target molecule
- The E2 mechanism is also similar to its SN2 counterpart in that it has a bimolecular rate law and is driven by nucleophilic attack.. the difference is that in an E2 reaction, a storng base attacks a proton adjacent to the carbon with a halide substituent. once that proton is removed, a double C=C bond if formed, which causes the leaving group to be kicked off
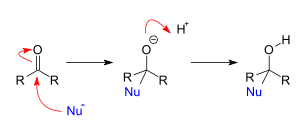
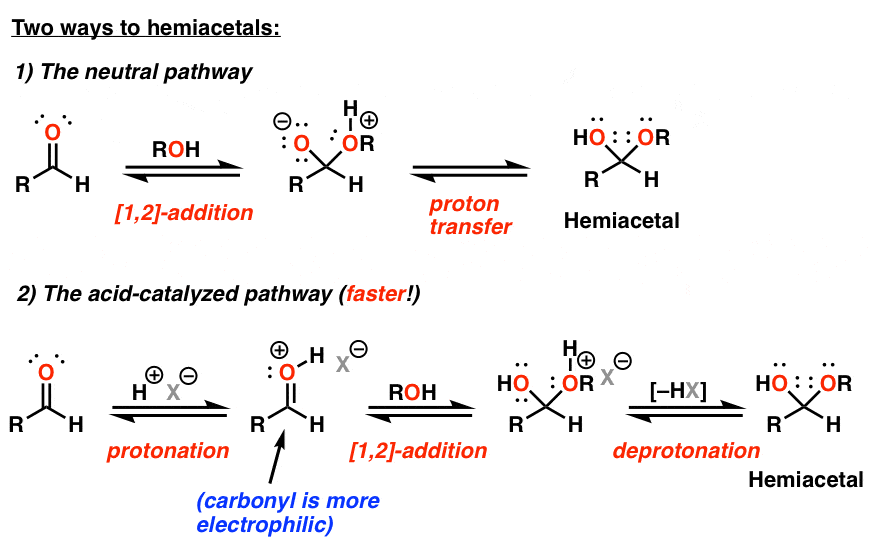
what is nucleophilic addition?
- if an aldehyde undergoes this reaction, the resulting structure is known as a hemiacetal, whereas a ketone that undergoes this reaction results in a hemiketal
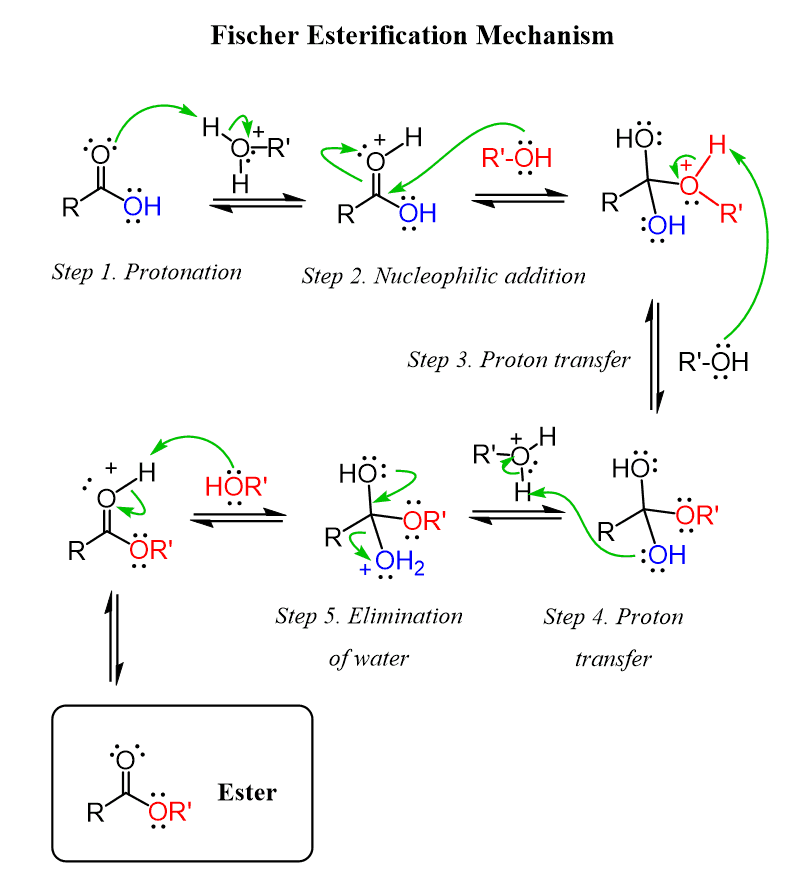
a prototypical example of a nucleophilic subtitution at a carboxylic acid group is provided by a process known as?
- Fischer esterification
- an acid-catalyzed, highly practical technique for turning a carboxylic acid into an ester (replacing the -OH group with the OR functional group characteristic of an ester)
- to do this, a carboxylic acid is mixed with an alcohol under acidic conditions
- Fischer esterification can be reversed through hydrolysis (a compound breaks down through the addition of water) and is a form of nucleophile subtitution
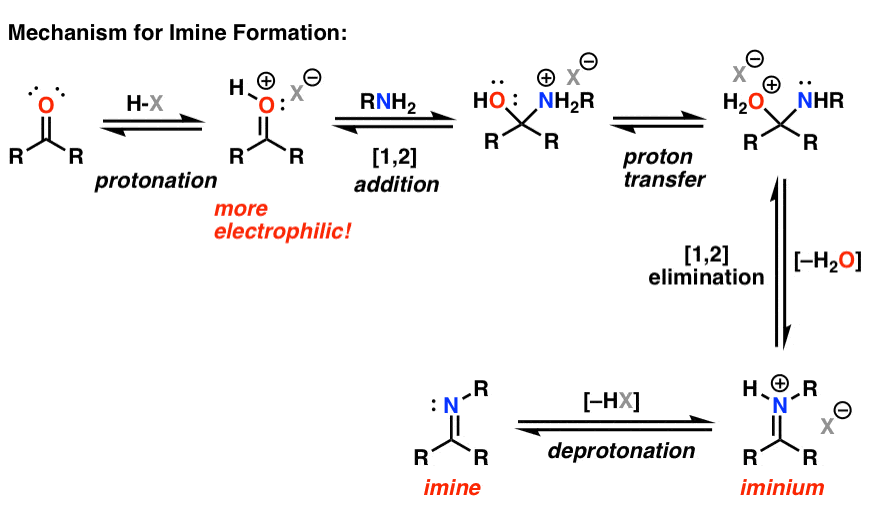
another important nucleophilic substitution reaction, imine formation:
- takes place at a carbonyl carbon
- the basic point of this reaction is to replace the carbonyl carbon with a nitrogen creating a C=N double bond that defines an imine
hemiacetal formation under acidic and basic conditions:
- if the initial molecule is a ketone, rather than an aldehyde, we have hemiketal formation
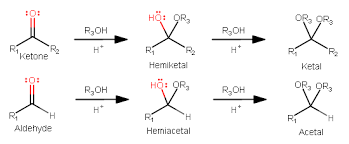
If there is an ecess of alcohol, the reaction can keep going, resulting in?
- acetals and ketals, which have 2 -OR groups instead of one -OR and one -OH group
- when a glycosidic bond is formed between 2 monosaccharides to form a disaccharide, a hemiacetal or hemiketal is converted into an acetal or ketal
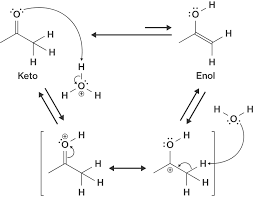
mechanism of keto-enol tautomerism:
- describes the interconversion between ketone and enol forms
- the interconversion between these 2 forms is generated by proton transfer that can be accomplished in either acidic or basic conditions
- in aqueous solution, the keto form generally predominates over the enol form
- if there is no alpha-hydrogen, tautomerization cannot take place
- the interconversion between these 2 forms is generated by proton transfer that can be accomplished in either acidic or basic conditions
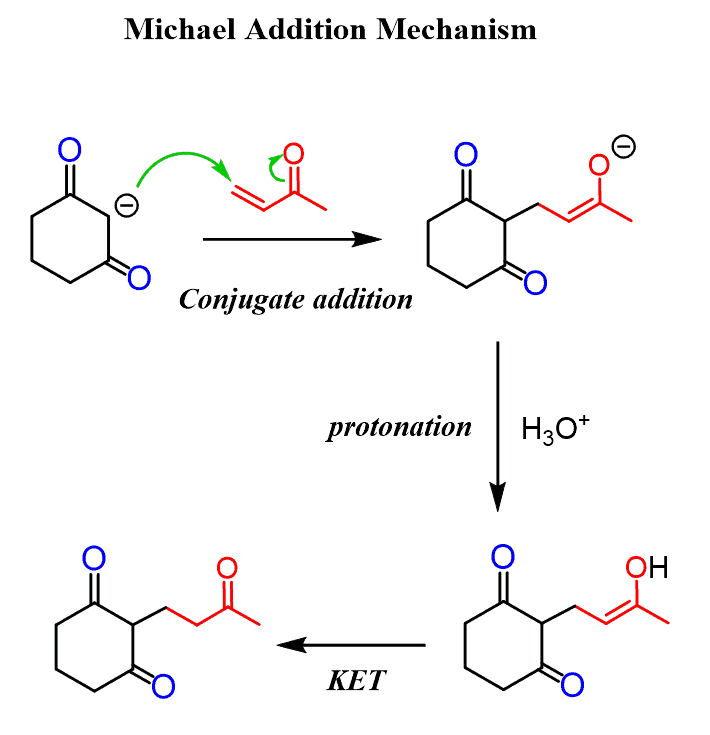
what is Michael addition?
- an enolate attacks the beta-C of an alpha,beta-unsaturated aldehyde/ketone
- in this mechanism, the double C=C bond is removed, initially resulting in a negative charge on the alpha-C. this negative charge is addressed by a quick protonation step, yielding a characteristic structure with 2 carbonyl groups that are separated by 3 C atoms, a 1,5-dicarbonyl structure
- key point is fundamentally that the beta-C in alpha,beta-unsaturated aldehydes/ketones is another potential site for the nucleophilic addition
- in this mechanism, the double C=C bond is removed, initially resulting in a negative charge on the alpha-C. this negative charge is addressed by a quick protonation step, yielding a characteristic structure with 2 carbonyl groups that are separated by 3 C atoms, a 1,5-dicarbonyl structure
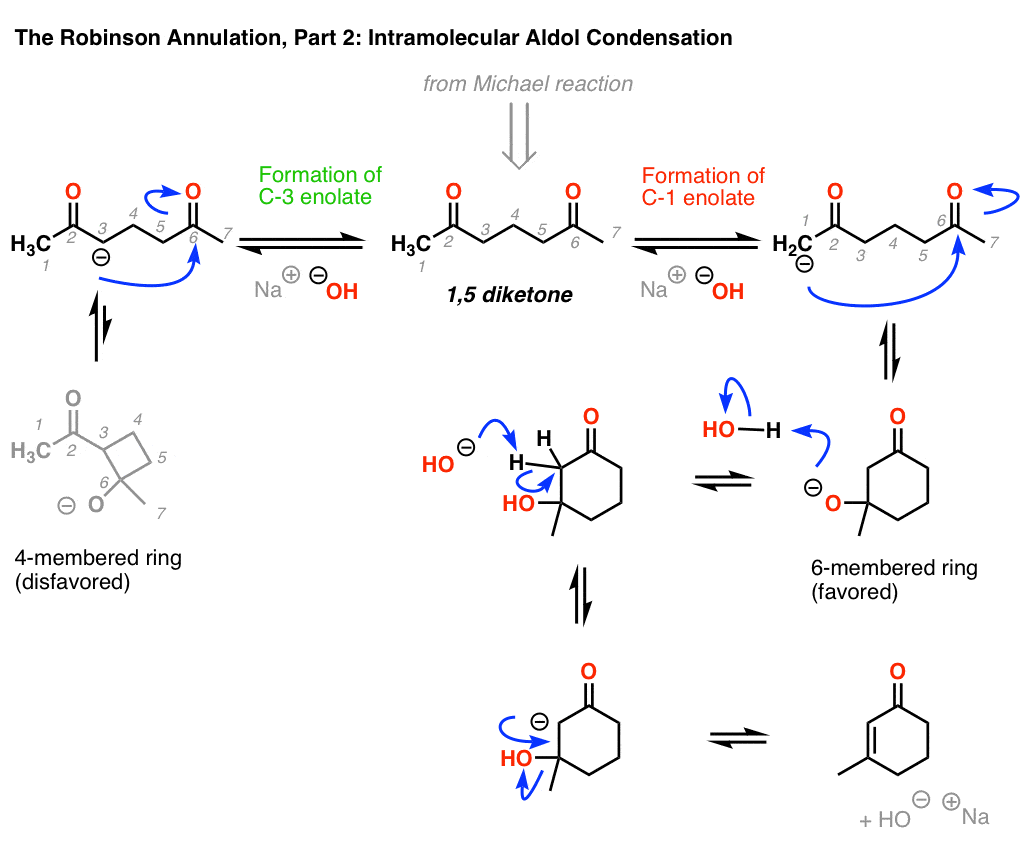
what is Robinson annulation?
- involves Michael addition followed by an aldol condensation
- limiting ourselves to basic conditions, it follows that we must form an enolate. this is possibly by using a strong base to remove a hydrogen from one of the alpha-carbons. more specifically, in order to form a stable ring structure without undue strain, we’ll need fo remove a hydrogen from an alpha-carbon at the end of the molecule. we label that alpha-carbon as C1. it acts as a nucleophile and attacks the carbonyl structure at the end of the molecule, which we label C6. just in standard aldol condensation reactions, a dehydration reaction pushes off the -OH group via an elimination mechanism, resulting in a new C=C bond, this is an alpha,beta, unsaturated cyclic ketone
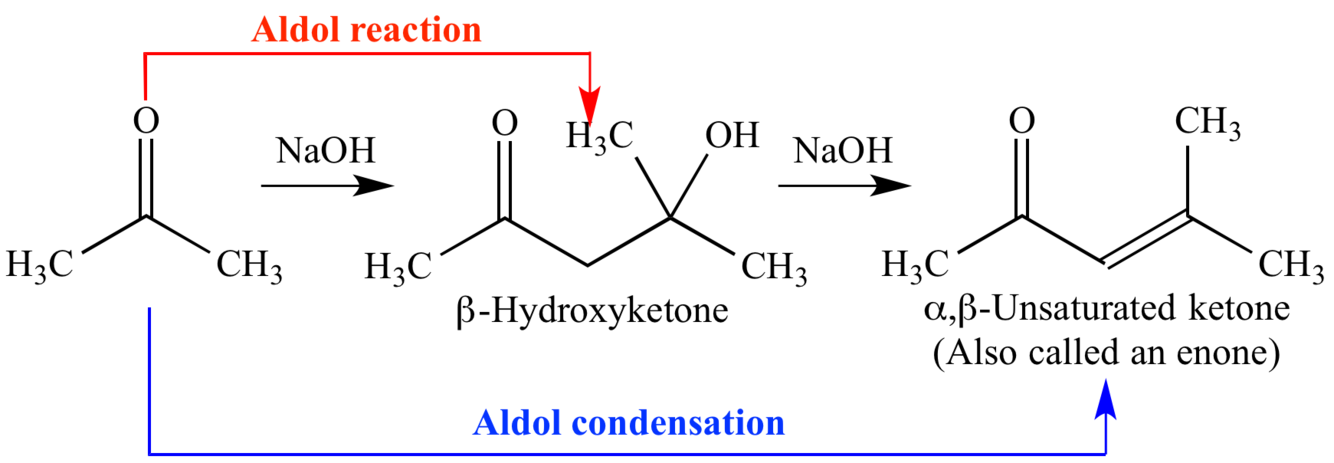
what is aldol condensation?
- base and acid catalyzed