Leo the lion says Ger
- Loss of electrons = oxidatin
- gain of electrons = reduction
a redox reaction is one in which?
- the oxidation states of compounds change
what is oxidation state?
- a method for keeping track of how electrons are shared within a molecule
- a model of electron distribution where we simplify things by assigning the electron to the mroe electronegative atom in a bond
rules for calculating oxidation state:
- pure elements have an oxidation state of 0 and applies even if they are found in diatomic molecules such as O2
- the oxidation state of monoatomic ions is equal to their charge so Fe2+ ion has an oxidation state of +2 and Cl- ion has an oxidation state of -1
- the sum of the oxidation states of the components of molecules and polyatomic ions is equal to their charge (zero for neutral molecules and some integar value for polyatomic ions)
priority rules for individual atoms within a compound:
- the oxidation state of F is -1 because it is the most EN
- other halogens will usually have an oxidation state of -1 unless they are bonded to a more EN halogen, N or O
- the oxidation state of H is +1 except when it is bonded to a more electropositive element (NaH, NaBH4, and LiAlH4), in which case it will be -1.
- the oxidation state of O is usually -2 with some important exceptions, such as peroxides, in which it is -1
- the oxidation state of alkali metals (the first column in the PT is always +1 and that of alkaline earth metals (2nd column on the PT) is always +2
what are examples of non-redox reactions?
- the oxidation states do not change because the overall distribution of electrons remains the same
- acid base chemistry
- precipitation reaction
- many substitution reactions
- many double displacement reactions
what are examples of redox reactions?
- ‘classic’ redox (single displacement reactions)
- combustion
- co,bination reactins
- many metabolic reactions
how to balance redox reactions:
- split into half reactions
- balance the non-O and non-H atoms
- balance the O
- balance the H
- add electrons to balance the charge
- multiply so that both half reactions have the same number of electrons
- add and cancel like terms
- this is for acidic conditions, but if basic conditions applied, wen use OH- to balance out the oxygen molecules and H2O to balance out the H+ molecules
what is an oxidizing and reducing agent respectively?
- an oxidizing agent is a compound that you can add to a reaction mixture to cause another substance to be oxidized
- a reducing agent is a compound that you can add to a reaction mixture to cause another substance to be reduced
- this means that oxidizing agents are themselves reduced and reducing agents are themselves oxidized.
what are common oxidizing agents?
what are common reducing agents?
common oxidizing agents:
- CrO3
- Na2Cr2O7
- pyridinium chlorochromate (PCC)
common reducing agents:
- NaBH4
- LiAlH4
how do we measure reduction potentials (E°)?
- measured in volts and are defined relative to the standard hydrogen electrode which is defined as being 0V
- greater (more positive) reduction potentials indicate that a substance wants to be reduced more
- smaller (more negative) reduction potentials indicate that a substance wants to be reduced less and would prefer oxidation if possible
- reudction potentials start being useful when we compare them!
reduction potentials are not affected by?
- stoichiometric constants
how can oxidation potentials be found?
- flipping the sign on the reduction potentiral of the reduction half-reaction
the electrode where oxidation occurs is known as the?
the electrode where reduction happens is known as the?
mnemonic?
- anode
- cathode
- An Ox Red Cat
- therefore a surplus of electrons is generated at the anode (because electrons are lost during oxidation) and they travel to the cathode
in a galvanic cell/ voltaic cell, a spontanous redox reaction is used to generate?
- a positive potential difference that can drive current
the total standard potential generated by a cell, Ecell, can be calculted by:
- Ecell = E°cathode - E°anode
how to solve when you are given a table of standard half reduction potentials and then asked to calculate the potential of the galvanic cell that would be set up using 2 specific half-reactions:
- identify which half reaction has the higher reduction potential (this will be the cathode because it wants to be reduced more) and the other half-reaction will be the anode
- plug those values into the equation Ecell = E°cathode - E°anode
- understand that the actual half-reaction that happens at the anode will be the reverse of the one shown in the table of reduction potentials because oxidation happens at the anode
When there is a difference between the standard values of a parameter (which assumes Standard conditions) and the values of a paramenter in non-standard (real world) conditions, we use the Nernst equation which helps us account for how electrical potential of a cell is affected by conditions including temperature and the concentration of reactants:
- E’cell = E°cell - (RT/zF) ln Q
- E’cell refers to the actual cell potential under a given set of conditions
- E°cell is the standard cell potential
- R is the ideal gas constant
- T is temp in K
- z is the number of moles of electrons transferred
- F is the Faraday constant (1 x105)
- Q is the reaction quotient
- simplified version is E’cell = E°cell - (0.05916)/z) log10Q
what is a Daniell cell?
- prototype of a galvanic cell
- the half-reactions are carried out in 2 physically separated half-cells
- the electrodes are connected by a conductive wire and the half cells are additionally connected by a salt bridge
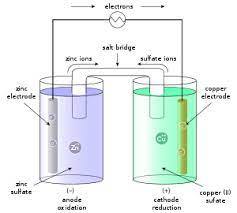
what is a common shorthand for denoting Daniell cells?
anode| anode solution || cathode solution| cathode
In Daniell cells, the cathode is on the ? and the anode is on the ?
- the cathode is on the right (where electrons are taken out of solution via a reduction)
- the anode is on the left (source of electrons)
in the Daniell cell, the 2 half-reactions are physically separated but it is possible to create a galvanic cell in which the 2 half-reactions take place in the same chamber. such cells are known as?
- concentration cells and they have to satisfy 2 conditions:
- there needs to be a concentration difference between 2 regions of the cell
- the electrodes need to be made out of the same material
- applications in pH and on each side of the cell membrane
what happens in an electrolytic cell?
- the idea is to apply energy to the system rather than to obtain energy from the system
- this means that tehr edox reactions will be carried out in the nonspontaneous direction
- the name reflects the fact that these cells are often used to break down compounds into their constituent parts through the application of electrical energy
- this means that tehr edox reactions will be carried out in the nonspontaneous direction
in electroyltic cells, the cathode (where reduction is carried out) is marked with a ?
while the anode (where oxidation is carried out is marked with a ?
- negative charge
- positive charge
- the opposite of what occurs in galvanic cells
- the convention used for an electrolytic cell is the same that is used for SDS-PAGE and isoelectric focusing so the Cathode is neg and the anode is pos (PANIC)
galvanic and elecrtolytic. ells are combined in?
- batteries
- discharge spontaneously (acting as a galvanic cell that produces current)
- recharged (acting as an electrolytic cell)


