when thinking about phase changes, we should be thinking about?
- intermolecular forces
- stronger intermolecular forces tend to be associated with solides
- weaker intermolecular forves with gases
- liquids are in between
what is a solid?
- a structure with a tightly packed organization of atoms, such as ice, NaCl, and most metals
- they are characterized by a fixed volume, meaning that they do not expand and are not compressible and have a fixed shape and do not flow but their particles do vibrate in place
- a solid can be crystalline or amorphous
what is a crystalline vs amorphous solid?
- crystalline solids enhibit a regular arrangement of atoms (lattice structure like NaCl) which is extremely difficult to disrupt
- the lattice energt of an ionic solid refers to the amount of energy required to separate the solid into its component cations and anions (large amount of energy)
- amorphous solids are solids that do not have a regular crystal structure (ex. glass)
what are liquids?
- have a fixed volume, meaning they are not compressible
- do not have a fixed shape
- have viscosity- the resistance of the liquid to deformation by certain forces
what are gases?
- not in a condensed phase
- lack a fixed shape or volume so density of a given gas is not constant
- if the gas is forced into a smaller container, its density will increase as its particles pack more closely together
what is it called when a substance goes from solid to liquid?
- melting or fusion
What is it called when a substance goes from liquid to solid?
- freezing
What is it called when a substance goes from liquid to gas?
- evaporation or boiling
What is it called when a substance goes from gas to liquid?
- condensation
What is it called when a substance goes from solid directly to gas?
- sublimation
What is it called when a substance goes from gas directly to solid?
- deposition
what are the 6 phase changes?
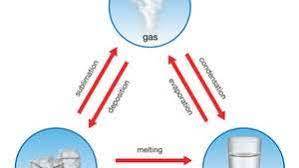
what are the 3 endothermic phase changes that require input of heat?
- melting, evaporation, and sublimation
- breaking of bonds or intermolecular forces
what are the 3 exothermic phase changes that release heat into the environment?
- freezing, condensation, and deposition
- bond formation or an increase in intermolecular forces
what is the heat of fusion?
- the amount of heat that was used solely to disrupt these interactions and melt the ice
the heat required to convert liquid to gas at constant temperature is?
- the heat of vaporization
both the specific heat of vaporization, which describes how much heat energy is necessary to boil a substance and the specific heat of fusion, which describes how much heat energy is necessary to melt a substance are given in units of?
- energy per unit of mass or per mole
what is specific heat capacity?
- the amount of heat required to raise the temperature of one unit mass (typically one gram or kg) of a substance by one degree
- the specific heat capacity of water is 4.184J/g•°C
specific heat capacity can be used in the following equation that related the heat applied to (or released by) a system to the temperature chage:
Q = mc deltaT
- Q is the heat (usually in J) applied or released by the system
- m is the mass
- c is the specific heat capacity of a substance
- deltaT is the change in temperature in ºC
- at phase changes, use heat of fusion or vaporization
- between phase changes, heat will be related to temperature change so use Q = mc deltaT
- try practice problem:
Imagine that we want to take 250g of ice from a -20°C freezer and turn it into water at 30°C under standard conditions. how much heat will this process equire?
- 3 steps
- heating the ice. we can use Q = mc delta T and approximate cice as 2 J/g•°C so Q = (250g)(2 J/g•°C)(20°C) = 10,000J
- melting the ice. we need the heat of fusion of water. Q = (250g)(334 J/g) = 83,500J
- heating the water. Q = mc deltaT and we can estimate cwater as 4 J/g•°C so Q = (250g)(4 J/g•°C)(30°C) = 30,000
- add all the joules up to gice 123,500J
the phase of a substance is affected by temperature and pressure. these relationships are illustrated using?
- phase diagrams, which are usually drawn with pressure on the y-axis and the temperature on the x-axis
- in phase diagrams, phase changes occur across the solid lines
- an increase in pressure or decrease in temperature can convert gas to solid (deposition) gas to liquid (condensation), or liquid to solid (freezing) depending on the temperature
- a decrease in pressure or increase in temperature can promote solid to gas (sublimation), liquid to gas (boiling), and solif to liquid (melting)
- solid to liwuid changes are depicted by green lines. for most substances, the solid phase is denser than the liquid phase so an increase in pressure causes liquids to form solids, ice however is less dense than liquid water due to its unique crystal structure so an increasing pressure can melt ice into liquid water so the phase change line between solid and liquid tilts to the left for water
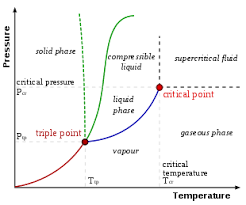
phase diagrams also have a triple point and a critical point which mean what?
- the triple point is the point at which solid, liquid and gas are in equilibrium
- the critical point is the point that represents the end of the liquid-gas interface. above this point, matter exists as supercritical fluid, which possesses properties of both liquid and gas
what is the concept of vapor pressure?
- describes what happens when a vapor, or gas, is in thermodynamic equilibrium with the liquid phase (or theoretically, the solid phase too)
- there is always some interchange between the liquid and gas phase at the boundaries of a liquid
- defined as the pressure exerted by the molecules of that substance that are in gas form in a closed system at a given temperature
- vapor pressure increases with temperature, and the point at which the vapor pressure of a liquid is equal to that of the surrounding atmospheric pressure corresponds to the boiling point of a substance
- there is always some interchange between the liquid and gas phase at the boundaries of a liquid
since gases are compressible, their density is affected by pressure and temperature, this means we have to specify certain pressure and temperature conditions in order to be able to compare density in a meaningful way. in general gases will specify conditions of standard temperature and pressure (STP) of:
- temperature = 0°C (273K)
- presure is 1 atm or 760 mmHg or 760 torr


