Protein structures follow 6 themes. Name them.
- the three-dimensional structure or structures taken up by a protein are determined by its amino acid sequence.
- the function of a typical protein depends on its structure
- most isolated proteins exist in one or a small number of stable structural forms.
- the most important forces stabilizing the specific structures maintained by a given protein are noncovalent interactions
- amid the huge number of unique protein structures, we can recognize some common structural patterns that help to organize our understanding of protein architecture
- protein structures are not static
The conformations existing under a given set of conditions are usually the ones that are _____ the most stable—that is, having the lowest _____ _____ _____
- thermodynamically
- Gibbs free energy (G)
native proteins
- Proteins in any of their functional, folded conformations
- marginally stable
A given polypeptide chain can theoretically assume countless conformations, and as a result the unfolded state of a protein is characterized by a high degree of conformational ______. This ______, and the hydrogen-bonding interactions of many groups in the polypeptide chain with the solvent (water), tend to maintain the unfolded state. The chemical interactions that counteract these effects and stabilize the native conformation include ____ _____ and the ______ _____
- entropy
- entropy
- disulfide (covalent) bonds
- weak (noncovalent) interactions
Many proteins do not have _____ _____. Outside the cell, the environment is often more oxidizing, and _____ formation is more likely to occur. In eukaryotes, disulfide bonds are found primarily in secreted, _____ proteins (for example, the hormone insulin). They are also uncommon in bacterial proteins
- disulfide bonds
- disulfide
- extracellular
In general, the protein conformation with the lowest _____ _____ (that is, the most stable conformation) is the one with the maximum number of weak interactions.
free energy
_____ _____ generally predominate. When water surrounds a hydrophobic molecule, the optimal arrangement of hydrogen bonds results in a highly structured shell, or _____ _____, of water around the molecule. The increased order of the water molecules in the solvation layer correlates with an unfavorable decrease in the _____ of the water. However, when _____ groups cluster together, the extent of the solvation layer decreases, because each group no longer presents its entire surface to the solution. The result is a favorable increase in _____ which is the major _____ driving force for the association of hydrophobic groups in aqueous solution
- hydrophobic interactions
- solvation layer
- entropy
- nonpolar
- entropy
- thermodynamic
amino acid sequences of most proteins feature a significant content of _____ amino acid side chains (name five). These are positioned so that they are clustered when the protein is folded, forming a hydrophobic protein _____
- hydrophobic
- Leu, Ile, Val, Phe, and Trp
- core
there are limits to the solubility of even the most _____ molecules as their presence causes a net decrease in _____ _____ per unit mass. Therefore, a solvation layer also forms to some extent around _____ molecules.
- polar
- hydrogen bonding
- polar
important that any polar or charged groups in the protein interior have suitable _____ for hydrogen bonding or ionic interactions. One hydrogen bond seems to contribute little to the stability of a native structure, but the presence of hydrogen-bonding groups without partners in the _____ _____ of a protein can be so destabilizing that conformations containing these groups are often _____ _____.
- partners
- hydrophobic core
- thermodynamically untenable
hydrogen bonds often have an important role in guiding the _____-_____ process.
protein-folding
strength of a salt bridge increases as it moves to an environment of _____ _____ _____, ´ (p. 50): from the polar aqueous solvent (´ near 80) to the nonpolar protein interior (´ near 4). Salt bridges, especially those that are partly or entirely _____, can thus provide significant _____ to a protein structure
- lower dielectric constant
- buried
- stabilization
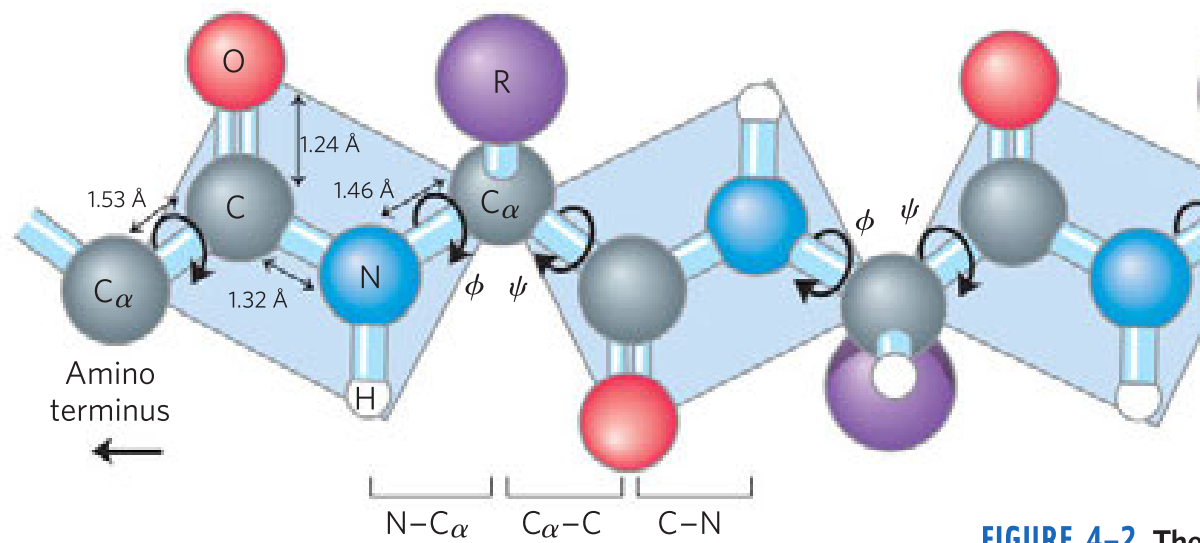
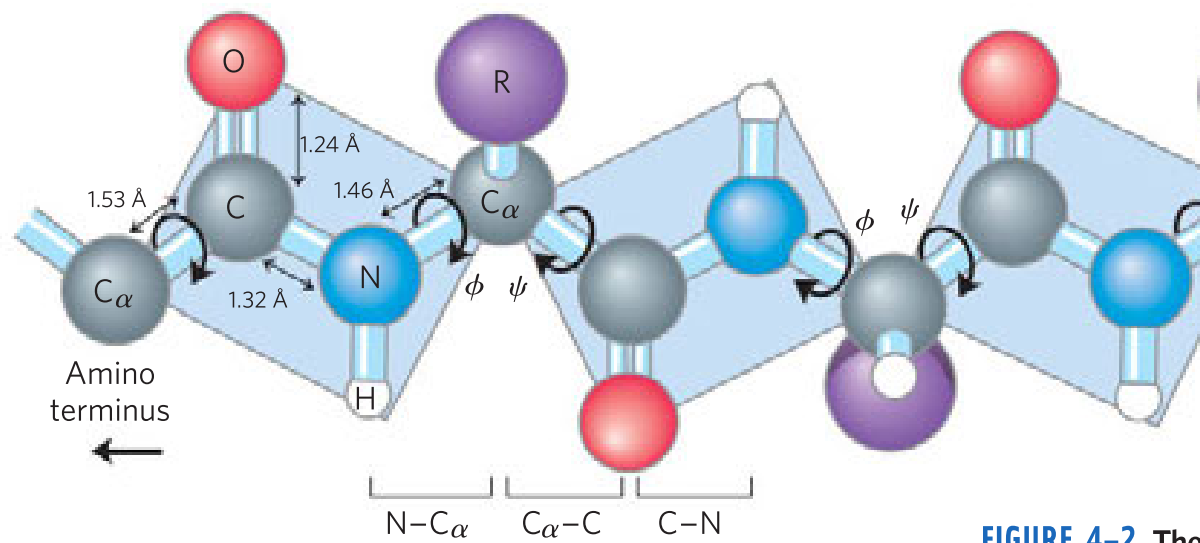
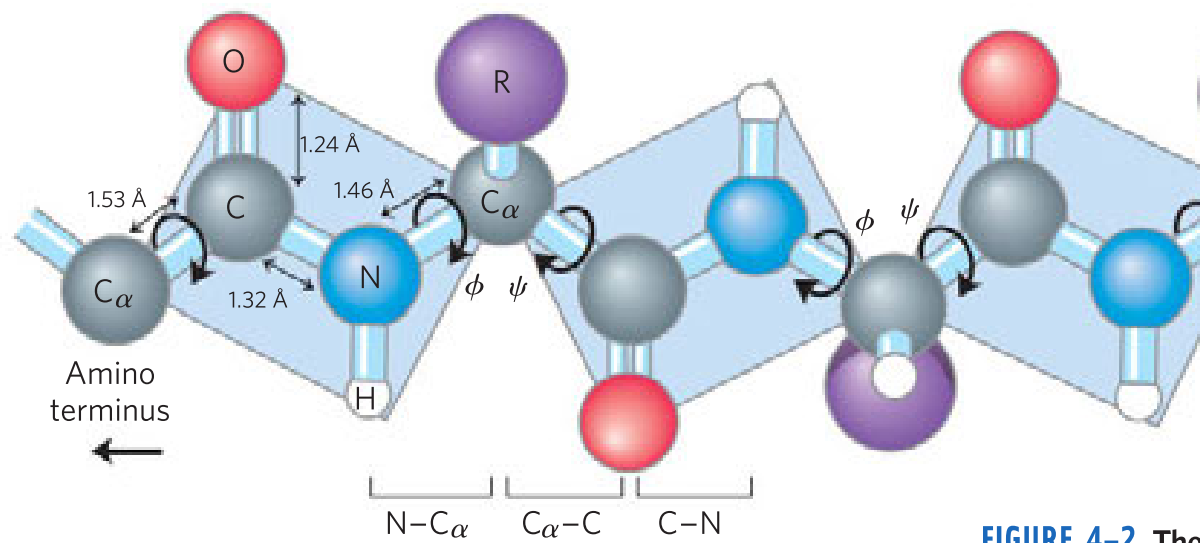
The Peptide Bond Is _____ and _____
- Rigid
- planar
The planar peptide group
- Each peptide bond has some _____-_____ character due to resonance and cannot _____
- Although the N atom is represented with a partial positive charge, bond orbitals and quantum mechanics show that N has a net charge that is
- double-bond
- rotate
- neutral or slightly negative
The planar peptide group
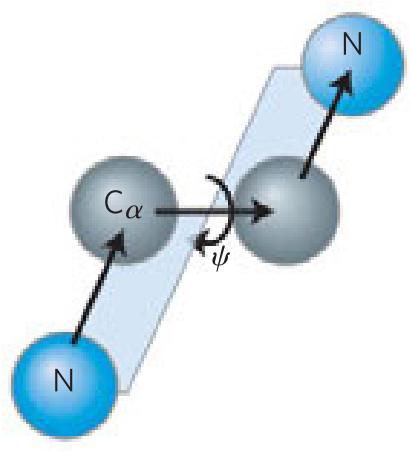
- _____ bonds separate sequential α carbons in a polypeptide chain
- _____ and _____ bonds can rotate, described by dihedral angles designated φ and ψ, respectively. _____ bond can’t rotate
- Other single bonds in the backbone may also be rotationally hindered, depending on
- Three
- N—Cα
- Cα—C
- C—N
- the size and charge of the R groups.
The planar peptide group
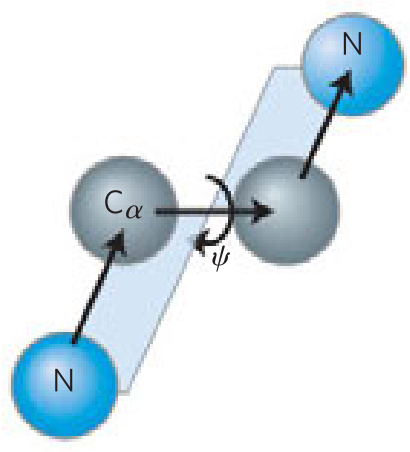
- φ and ψ are _____ or _____ when the first and fourth atoms are farthest apart and the peptide is fully extended
- looking along the bond undergoing rotation, the φ and ψ angles _____ as the fourth atom rotates clockwise relative to the first
- 180º
- -180º
- increase
secondary structure
- a segment of a polypeptide chain and describes the local spatial arrangement of its main-chain atoms
- ignores side chains or its relationship to other segments
- A regular secondary structure occurs when
- Examples of secondary structures are
- each dihedral angle, φ and ψ, remains the same or nearly the same throughout the segment
- the α helix and β conformations; another common type is the β turn
undefined or random coil
Where a regular pattern is not found in the secondary structure
α helix
- polypeptide backbone is tightly wound around
- R groups protrude outward from the helical backbone
- a single turn of the helix extends about 5.4 Å
- each helical turn includes 3.6 amino acid residues often deviate slightly
- right-handed α helix is the common form
- left-handed α helices are less stable; not observed in proteins
Why does the α helix form more readily than many other possible conformations?
- optimal use of internal hydrogen bonds
- stabilized by a hydrogen bond between the hydrogen atom attached to the nitrogen and the carbonyl oxygen atom of the fourth amino acid on the amino-terminal side
- every peptide bond (except those close to each end of the helix) participates in such hydrogen bonding
- At the ends there are always three or four amide carbonyl or amino groups that cannot participate in this helical pattern of hydrogen bonding
- exposed to the surrounding solvent
- they hydrogen-bond with water
- or other parts of the protein may cap the helix to provide needed hydrogen-bonding partners
What is the length of a polypeptide with 80 amino acid residues in a single contiguous α helix?
- α helix has 3.6 residues per turn
- rises 5.4 Å
- Thus rise for each amino acid residue is 1.5 Å (5.4 / 3.6)
- The length of the polypeptide is therefore
80 residues × 1.5 Å/residue = 120 Å.
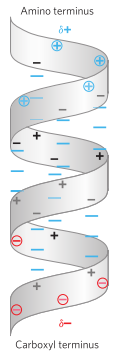
Hellix dipole
- transmitted through the intrachain hydrogen bonds, resulting in an overall helix dipole
- In pic, the amino and carbonyl of each peptide bond are indicated by “+” and “-“ symbols
- Non–hydrogen-bonded amino and carbonyl near each end of the helical region are circled and shown in color
Proline occurs infrequently in α helices because
- have the least proclivity to form helices
- nitrogen atom is part of a rigid ring making rotation about the N—Cα bond is not possible which introduces a destabilizing kink
- nitrogen atom of a Pro residue in a peptide linkage has no substituent hydrogen to participate in hydrogen bonds with other residues












