Distinguish between a molecule, a compound, and a binary compound.
- Molecule – 2 or more atoms bound together.
- Compound – a molecule that has at least two different elements.
- Binary compound – compound that has precisely 2 elements.
- Ex: CO2 is a triatomic molecule, binary compound. O3 (ozone) is a triatomic molecule.
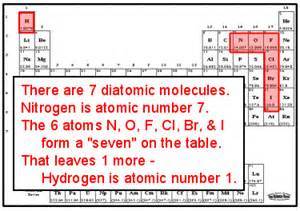
What are the seven diatomic molecules?
- hydrogen, nitrogen, oxygen, fluorine, chlorine,bromine, iodine
- They never exist alone. Instead they double up to form nonpolar covalent compounds
- Exist as gas (g)
Categorize elements by groups.
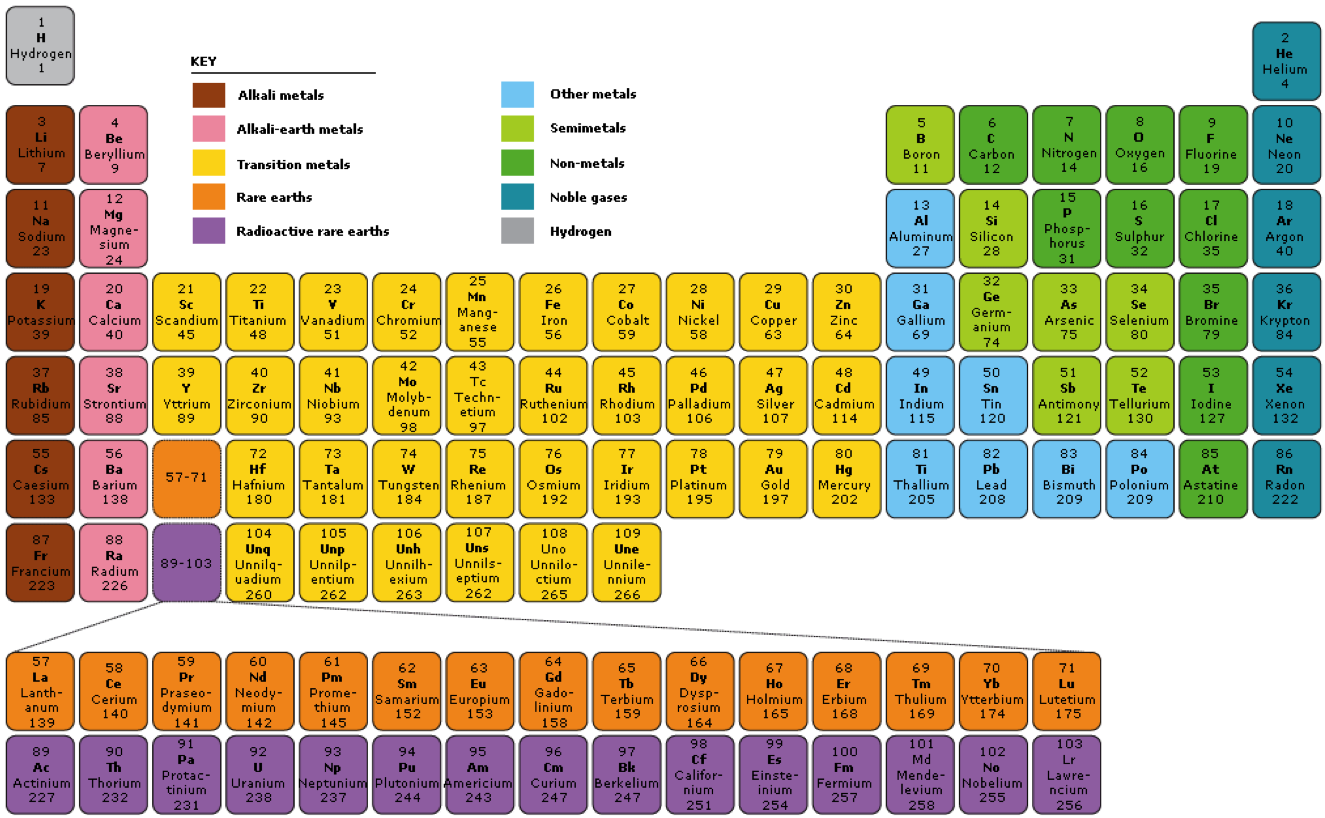
- Red: Alkali metals (when they combine with water, they create an alkaline solution)
- Pink: Alkali earth metals
- Yellow: Transition metals.
- Orange, Purple: Inner transition (aka rare earth, lanthanides and actinides)… but they aren’t used in general chem.
- Light blue: other metals.
- Pea green: Live on a staircase. Semi-metals aka metalloids.
- Dark green: Non-metals.
- Dark blue: Noble gases aka inert gases.
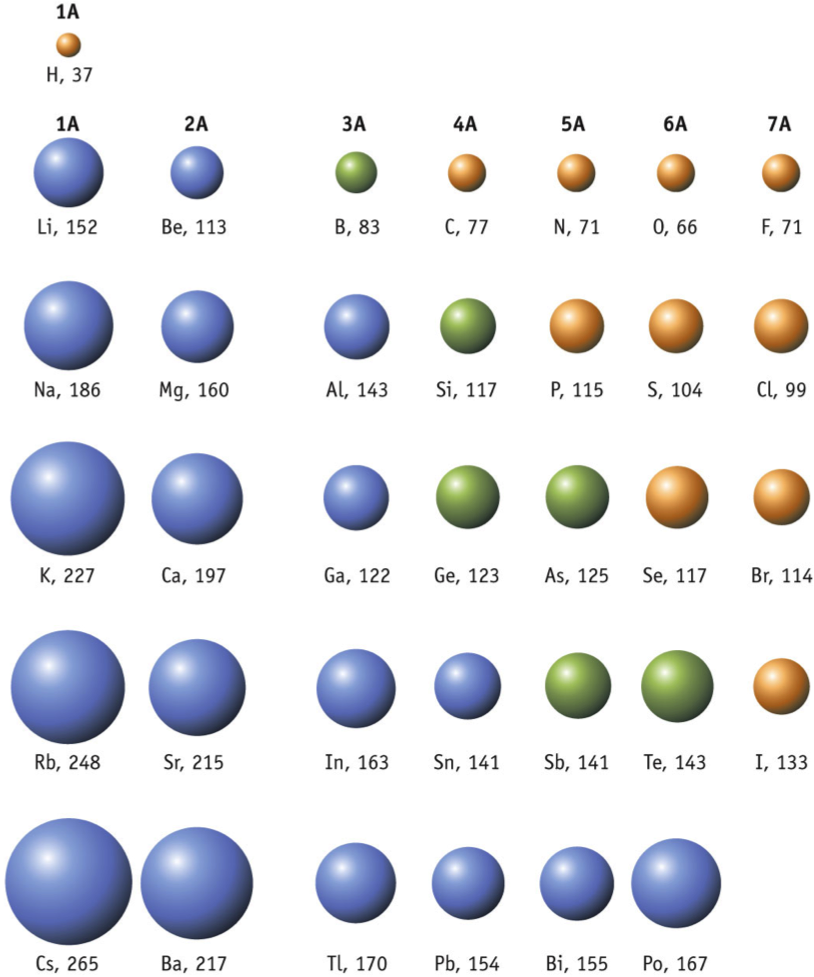
How do atomic radii change, looking at the periodic table?
Larger as you move down a group. Smaller as move across period.
Plum pudding model vs. Rutherford’s planetary model vs. Bohr’s model?
- The plum pudding model: We used to think that electrons were little negative spots of charge within a ball of diffuse positive charge. This persisted until Rutherford’s experiment.
- Rutherford aimed alpha particles (positive charge) at layer of gold atoms. A few of the alpha particles deflected greatly. Led to understanding of a nucleus with a positive charge, and electrons orbiting nucleus.
- Bohr’s model: Electrons don’t live in orbits. They exist in specific allowed energy levels.
What is the difference between these?
- atomic number
- mass number
- atomic mass or molecular mass
- molar mass or atomic weight
- Atomic number (Z) = # protons (dictates the element)
- Mass # (A): Whole number. The number of protons and neutrons of that particular atom. If you aren’t given the # of neutrons, just choose the closest whole number to the atomic mass.
- Atomic mass (A) = Weighted average of all the naturally occurring isotopes. Take the periodic table decimal. Unit: amu.
- Molecular mass = Weighted average like above, but for a molecule. Unit: amu.
- Molar mass aka atomic weight = Numerically the same as above, a decimal. But the unit is different: g/mol or u. It’s the average mass of all of the naturally-occurring isotopes of an element.
Explain this.
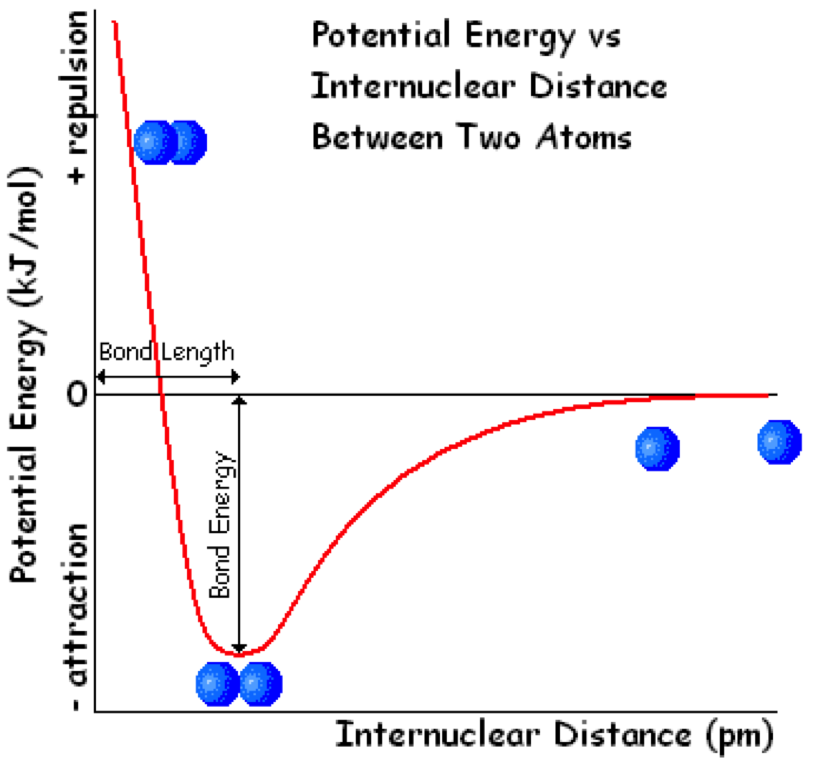
- Bonding has to do with stability. Attractions are stabilizing, repulsions are destabilizing. If two atoms are far apart enough from each other (internuclear distance “bond length” is large), there is no attraction or repulsion, zero energy of reaction. If the internuclear distance is smaller, then the electrons will also be attracted to the nucleus of the other atom, and they will become more stable, lower energy. “Bond energy” is the stabilizing energy you get from bonding, the lowest. But if you start pushing them very close together, there will be repulsion between the two protons.
- Negative energies are associated with stability, positive energies are associated with instability/breaking bonds.
What kinds of elements form covalent bonds, and what does this create?
- Occurs between nonmetals
- Involves sharing of electrons
- Creates molecular compounds, neutral
What kinds of elements form ionic bonds, and what does this create?
- Between a metal and a nonmental
- Involves electron transfer
- Creates ionic compounds, which are electrically neutral overall, but made of charged cations and anions
- The ionic bond itself is the electrostatic attraction between those ions. They’re attracted and often form lattice structures
- Ionic compounds are solid (s) when they are pure and not in water
- Ionic compounds are aqueous (aq) when water is around
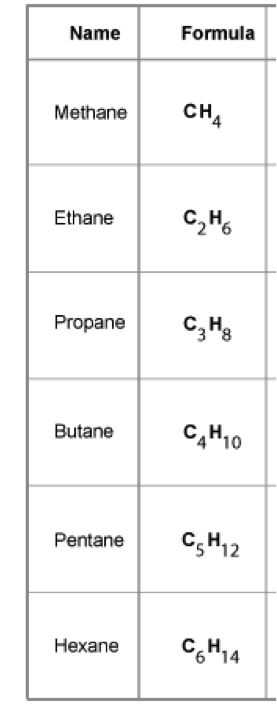
What are the first six hydrocarbons?
Pattern - carbon is n, Hydrogen 2n + 2
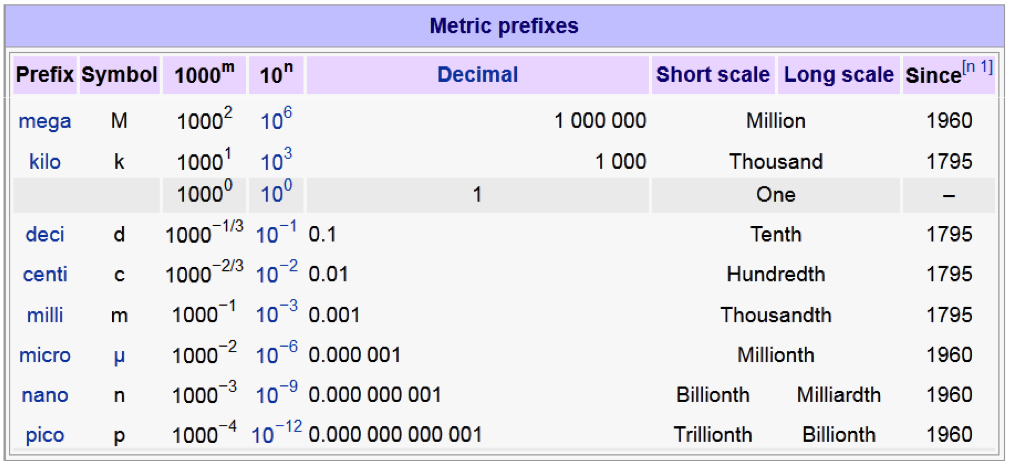
What are the main metric prefixes?
mega (M) 10^6 (million)
kilo (k) 10^3 (thousand)
deci (d) 10^-1 (tenth)
centi (c) 10^-2 (hundredth)
milli (m) 10^-3 (thousandth)
micro (u) 10^-6 (millionth)
nano (n) 10^-9 (billionth)
pico (p) 10^-12 (trillionth)
What are the ten prefixes used in nomenclature?
mono
di
tri
tetra
penta
hexa
hepta
octa, nona, deca
Avogadro’s number
6.02 × 1023
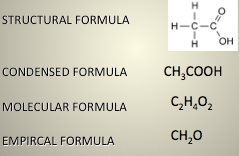
Distinguish:
- Structural formula
- Condensed formula
- Molecular formula
- Empirical formula
- Structural formula shows how all the atoms are attached to each other, with lines.
- Condensed formula condenses, it’s a map of the structural formula.
- Molecular formula (the most common) groups each of the elements. Order the elements in most metallic to least metallic (left to right, except if there is C and H, put C first).
- Empirical formula is the simplest whole number ratio of everything.
In a combustion equation, what are the reactants and products?
- Combustion as a reaction is the rapid combination of the reactant with oxygen.
- Common combustion products are CO2 and H2O, as long as sample has C and H in it.
- Because of the way combustion works, we know:
CxHyBlahz + O2 –> CO2 + H2O + (don’t worry about whatever else)
- We can convert between moles and grams to find the composition of the reactants
What is the equation for the reaction of an alkali metal with water?
*alkali* + water → *alkali* hydroxide + hydrogen
All the alkali metals react vigorously with cold water. In each reaction, hydrogen gas is given off and the metal hydroxide is produced. The speed and violence of the reaction increases as you go down the group.
Ex: 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
Ex: 2Li(s) + 2H2O(l) → 2LiOH(aq) + H2(g)
What is the equation for the reaction of an alkali earth metal with water?
Similar to the alkali metal + water reaction. Balances differently.
Ex: Calcium (and the metals below calcium in group 2) will react with cold water.
calcium + water –> calcium hydroxide + hydrogen
Ca(s) + 2H2O(l) Ca(OH)2(aq) + H2(g)
Ex: Magnesium will not react with water but will react with gaseous water (steam) to form magnesium oxide and hydrogen gas.
magnesium + steam –> magnesium oxide + hydrogen.
Mg(s) + H2O(g) –> MgO(aq) + H2(g)
What is the difference between intensive and extensive properties?
Intensive properties don’t change with size – extensive too.
Ex: Density, boiling point, color, etc don’t change regardless of size. Volume and mass do.
What are the signs of a chemical change?
color chage, formation of a precipitate, formation of a gas, temperature change


