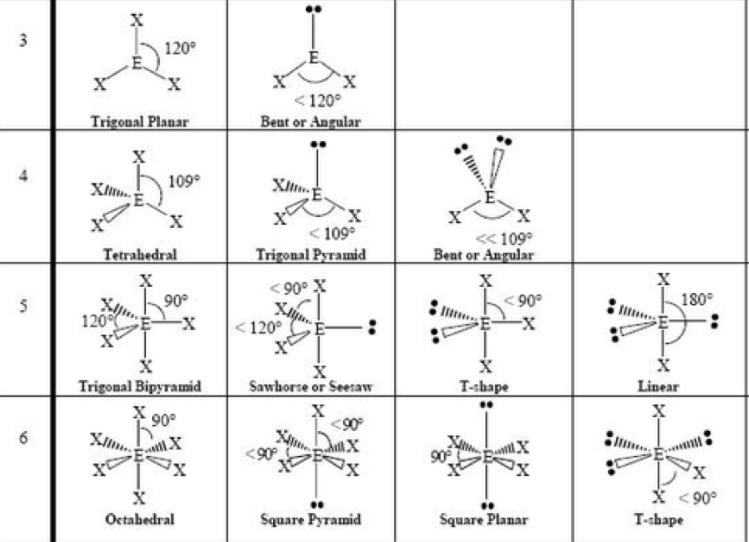
What is the steric number?
- Looking at central atom: # of atoms bonded to it + # of lone pairs
If there are no lone pairs, what are the shapes of steric numbers 2-6?
SN = 2 is linear (180°)
SN = 3 is trigonal planar (120°)
SN = 4 is tetrahedral (109.5°)
SN = 5 is trigonal bipyramidal (90° and 120°. Trigonal planar with linear through it.)
SN = 6 is octahedral (90°)
How do lone pairs affect the angles in a molecule?
- They take up more space than a bond
- So whatever main shape they’re based on, they squish the angles slightly.
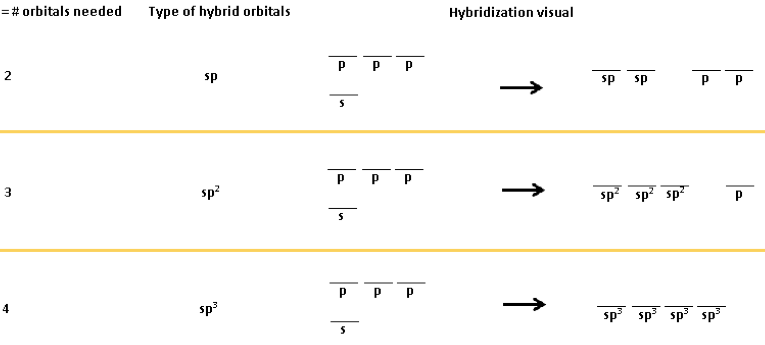
What are the hybrid orbitals for steric numbers 2-6,
and when are there p orbitals left over for π bonds?
The steric number = the total number of hybrid orbitals.
- sp (with 2 leftover p orbitals)
- sp2 (with 1 leftover p orbital)
- sp3
- sp3d (with 4 leftover d orbitals that don’t matter now)
- sp3d2 (with 3 leftover d orbitals that don’t matter now)

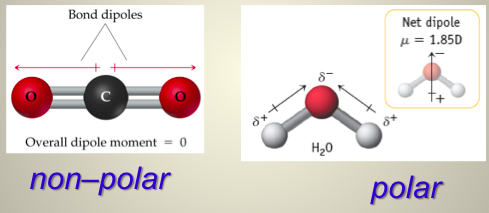
What does molecular polarity require?
- A molecule has a net dipole moment if it has at least one polar bond (a difference in electronegativity) AND
- If the bond dipoles don’t cancel, based on shape.
- If the atoms are the same and there are no lone pairs, nonpolar.
- All of the high symmetry molecular shapes (linear, trigonal planar, tetrahedral,trigonal bipyramidal, octahedral) yield non-polar molecules as long as all of the outer atoms are identical.
- If the central atom has lone pairs (aka nonbonding region) or if not all the atoms are the same, it’s polar. With the exception of SN 5 with 3 lone pairs (linear shape) and SN6 with 2 lone pairs (square planar)
- If the atoms are the same and there are no lone pairs, nonpolar.
Which theory do we use to determine molecular shape?
VSEPR
Valence Shell Electron Pair Repulsion theory.
(The most important factor in determining geometry is the repulsion between electron pairs.)
Bonding vs antibonding orbital
- A bonding orbital is constructive, adds stability, concentrates electron density between nuclei, and lowers the energy
- An antibonding orbital leads to nodes, and more energy
- So sigma* or pi* is higher in energy than sigma or pi
What do σ and π mean in the context of molecular orbital theory?
- σ = a molecular bonding orbital
- Formed by overlap of s orbitals
- σ* is antibonding and higher in energy
- π = a molecular bonding orbital
- Formed by overlap of p orbitals (specifically 2px and 2py orbitals that are perpendicular to the molecular axis)
- π* is antibonding and higher in energy
What do σ and π mean in the context of valence bonding theory?
- A sigma bond (σ) forms when two atomic orbitals overlap between the nuclei of two atoms (the internuclear axis).
- Pi bonds (π) occur when two atomic orbitals overlap above and below the internuclear axis.
- One π bond forms when two unhybridized p-orbitals overlap. Sandwich.
- Single bonds have one σ. Double bonds have one σ and one π. Triple have one σ and two π bonds.
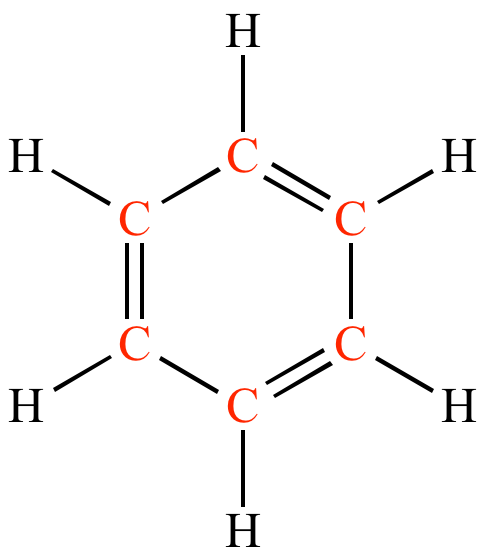
What is conjugation?
- Alternating single and double bonds = conjugation.
- The fractional bond order is the average between double and singles. So that is greater than a tabulated C-C bond. The average bond length will be shorter.
How can you predict molecule polarity?
- If the central atom has a lone pair, or if not all the atoms around it are the same, you can suspect polar
- Exceptions to lone pair tip: SN 5 with 3 lone pairs. Makes a linear shape that cancels out. SN 6 with 2 lone pairs. Makes a square planar shape that cancels out.
- Otherwise, the other shapes are symmetrical and nonpolar
- (Linear, planar trigonal, tetrahedral, trigonal bipyramidal, octahedral)
- Link for more flashcards: https://quizlet.com/181492688/ap-chem-shapeshybridization-flash-cards/
How can you tell if a molecule is paramagnetic or diamagnetic?
- Paramagnetic if has unpaired electron. Will be attracted to magnet.
- Safe to assume if there is an odd # of Valence Electrons
- But some even #’s can be paramagnetic too: ex O2
- Diamagnetic if no unpaired electrons.
- Need to look at the molecular orbital diagram, see if any unpaired
- (whereas for an element you could just look at the electron configuration)
What is the molecular orbital theory used for?
- Shows delocalized electrons in diatomics (as far as we’re concerned)
- Predicting magnetism
- paramagnetic if there are unpaired electrons
- Calculating MO bond order
- (# electrons in bonding orbitals - # electrons in antibonding orbitals ) /2
- Energy transitions color
- HOMO: Highest occupied molecular orbital
- LUMO: Lowest unoccupied molecular orbital
- Absorbance of light bumps an electron from HOMO to LUMO.


