Reagent vs. reactant
- Reagent is “off the shelf”.
- A reactant is a substance consumed in the course of a chemical reaction.
Solvent vs. solute
- Solvent - more of it. Assume that it is an aqueous solution and that the solvent is water.
- Solute - have less of it, can be a solid, liquid or gas.
Solubility
- The # grams of solute that can dissolve in 100g of solvent at room temperature, to make a saturated solution
- When a compound has reached it’s maximum solubility and there is solid is present in the solution, the solution is SATURATED.
Coulomb’s Law
- Force of attraction or repulsion is directly proportional to the magnitude of the charges, and inversely proportionally to the distance between the particles. So as ion charges increase, the force increases. As distance increases, the force decreases.
- The sign of the charges matter for showing us the direction - attraction or repulsion
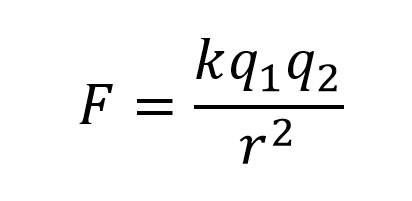
What happens to ionic compounds in water?
- The compound dissolves, dissociates. The ions separate break up into cations and anions. The ions move freely in the water, and the solution is able to conduct electricity.
- The electrolytic solution can be strongly acidic or strongly basic
Strong electrolyte vs. weak electrolyte vs. nonelectrolyte
- A strong electrolyte is an ionic compound that 100% dissociates in aqueous solution.
- A weak electrolyte partially dissociates.
- When a nonelectrolyte (like sugar) dissolves in water, molecules remain intact. The solution carries no electrically charged particles.
What is the formula for the dissociation of calcium chloride?
CaCl2 (s) –> Ca2+ (aq) + 2Cl- (aq)
Write the formulas of the ions separately, to show that they are independent of each other.
Describe the 3 types of equations for a reaction between solutions of electrolytes
- Molecular equation keeps the molecules together.
- Ionic equation separates the ions (except for individual polyatomics) and shows their charges
- Keep in molecular form if the ionic substance is insoluble
- Weak acids and bases are always written in molecular way, never in ionic way
- To be balanced, need same net charge on both sides of equation
- Net ionic equation eliminates the spectator ions, focus on the chemical change
ionic compounds vs. molecular compounds
- Ionic compounds are between metals and nonmetals or between metals and polyatomic ions or between polyatomics. When an ionic substance is dissolved in aqueous solution, it ALWAYS ionizes and the ions always have a charge.
- Ex: sodium chloride NaCl, magnesium nitrate Mg(NO3)2, ammonium sulfate (NH4)2SO4.
- Remember to never split apart a polyatomic ion. NH4NO3 ionizes into NH4+ and NO3¯.
- Molecular compounds involve only nonmetals. Molecular substances never ionize in solution, they exist as complete molecules and never have a charge.
- Ex: sulfur trioxide (SO3), carbon dioxide (CO2), water (H2O) and glucose (C6H12O6).
Solubility rules for ionic compounds in water:
What are the rules for soluble compounds?
- If one ion from the Soluble Compound list is present in a compound, the compound is water soluble.
Write in dissociated form:
- All compounds of the alkali metals (Group 1A) are soluble.
- All salts containing NH4+ , NO3- , ClO4 - , CLO3- , and C2H3O2 - are soluble.
- All chlorides, bromides, and iodides (salts containing Cl- , Br-, or I-) are soluble EXCEPT when combined with select transition metals: Ag+ , Pb2+ and Hg22+
- All sulfates (salts containing SO42-) are soluble EXCEPT those of Pb2+ Ca2+, Sr2+, Hg22+ and Ba2+
Solubility rules for ionic compounds in water:
What are the rules for insoluble compounds?
- Most salts of carbonate CO32-, phosphate PO43-, sulfite SO32-, sulfide S2-
- EXCEPT for salts of NH4+ and the Group 1 alkali metal cations
- Most metal hydroxides and oxides
- EXCEPT for those of Group 1A, Ca, Sr, Ba
Dissolve vs. dissociate vs. ionize
- To dissolve is to become part of a solution. (Any soluble compound.)
- To dissociate is to break up into ions which already exist in a compound. (Ionic compounds only.)
- To ionize is to produce new ions by gain or loss of electron. (Covalent compounds or neutral metals only, since they didn’t have ions before.)
What does is mean to be soluble?
Soluble means it dissolves
and in the case of electrolytes, dissociates
(breaks up into its ions).
Different from miscible.
What are strong and weak electrolytes?
- An electrolyte is an ionic compound that conducts electricity in aqueous solutions because it dissolves and dissociates in water.
- Strong electrolytes dissociate completely.
- Soluble salts, strong acids, strong bases.
- Ex: CuCl2 (s) –> Cu2+ (aq) + 2Cl- (aq)
- Weak electrolytes dissociate only to a small extent
- Weak acids, weak bases, partially soluble salts
- Ex: acetic acid. CH3CO2H (aq) ⇄ CH3CO2- (aq) + H+(aq)
- Nonelectrolytes dissolve, but do not dissociate
- Ex: insoluble compounds.
Salt
An ionic compound that is neither an acid nor a base
Acid
Acids are ionic compounds that increase the concentration of H3O+ in water.
Base
- Bases are ionic compounds that increase the concentration of OH- in water. Proton acceptor.
- When you see OH, be careful to notice what its attached to. If it isn’t attached to a cation, it isn’t a base.
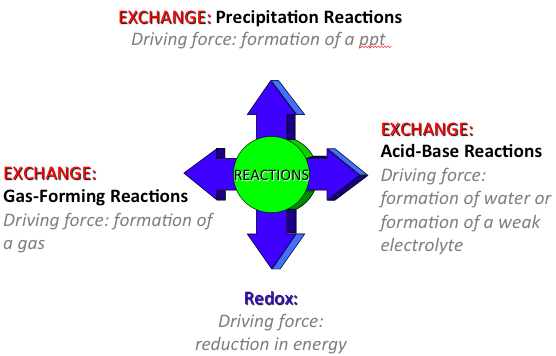
What are types of reactions?
- Exchange (aka double displacement, displacement, metathesis): ionic reaction in which cations and anions change partners.
- Precipitation formation (Due to low solubility - use solubility rules to predict)
- Acid-base neutralization
- Gas formation
- Combustion (compound with C and H + O2 –> CO2, H2O
- Single replacement from lab
- Magnesium reacts with hydrochloric acid according to the equation: Mg (s) + 2 HCl (aq) –> MgCl 2 (aq) + H 2 (g) This demonstration can be used to illustrate the characteristic reaction of metals with acid, a single replacement reaction, or to demonstrate the generation of hydrogen gas.
- Redox (oxidation-reduction) reactions
- Ionization reaction - ions form where they didn’t exist before. (Covalent compounds or neutral metals only)
- Ionic reaction - a reaction between ions
When is there no reaction?
- If everybody is soluble, nothing happens in the beaker. The ions just keep floating around.
- Write no reaction “ –> NR”.
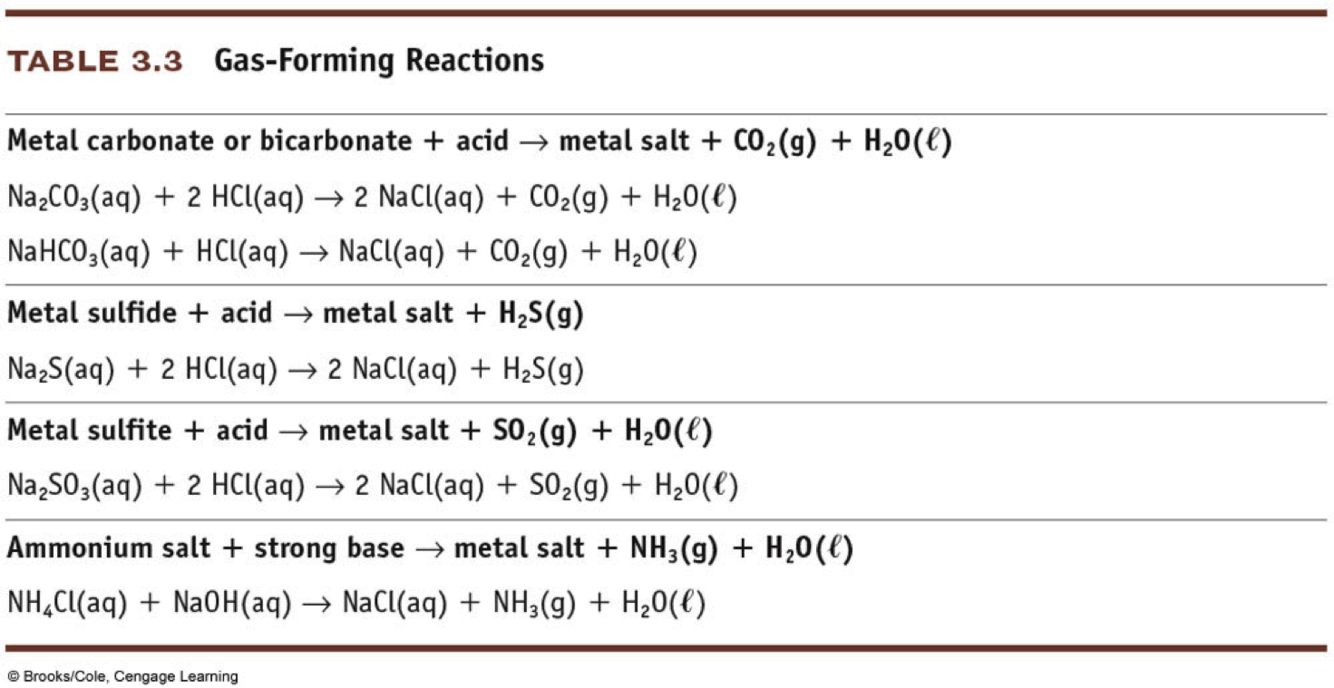
What are gas forming reactions?
- Strong driving force to create gas.
- Do exchange reaction and recognize gas products: CO2, NH3, SO3, H2S, HCN
What is molarity, and its relevant equations?
- Molarity (M) is how we define concentration in aqueous solution
- Molarity = moles of solute / liters of solution
- moles = Molarity • Volume
- M1V1 = M2V2
- Molaritystock • Volumestock = Mdilution • Vdilution
What are the important pH equations?
- pH = -log[H+]
- interchangeable with [H3O+]
- [H+] = 10-pH
- pH + pOH = 14
- [H+][H3O+]= 10-14


