What is a monoclonal antibody?
Recognises a single epitope on the antigen, less likely to cross-react.
What is a polyclonal antibody?
Recognises multiple epitopes on any one antigen, cheaper and quicker to produce
What is Western Blotting and when was the method invented?
- Introduced by Towbin et al. in 1979.
- Western blotting is a routine technique for protein analysis
- Involves separation of macromolecules by electrophoresis
- Followed by transfer (blotting) onto a adsorbent membrane and
- Detection with artificial antibodies specific to the protein of interest.
Cells and tissues need to be lysed to release the proteins of interest.
What key differences with DNA extraction need to be considered when preparing a sample for protein analysis?
- Some antibodies are only able to recognize proteins in their native, non-denatured form (i.e. epitope is only present in 3-D structure), thus the sample must not be denatured or heated.
- The epitope of other antibodies may only be exposed when the protein is unfolded.
- Some antibodies can only recognize proteins in their non-reduced form (i.e. oxidized), therefore reducing agents should not be used.
- The protein of interest may only be located in a specific organelle which requires specialist extraction.
What methods can be used to isolate proteins found in specific organelle?
Specific fractionation protocols are also used to retrieve specific cells fractions e.g.
- centrifugation
- velocity sedimentation on sucrose gradients
- equilibrium sedimentation on cesium chloride gradient
What is another major factor to consider when preparing samples for protein analyisis, and how is this dealt with?
- Protein degradation (proteolysis, dephosphosphorylation and denaturation) begins straight after lysis has occurred.
- Protease and phosphatase inhibitors. Cocktails of inhibitors and 4°C storage used to slow down.
When performing Western blotting for protein quantification what additional tests should be performed?
If blot is being used in a quantitative manner, total protein concentration of the sample should be determined e.g.
- Bradford
- Lowry
- BCA assay
What advantages does denaturing/unfolding the protein have and what is the most common methodology for denaturing proteins for analysis?
Dispulphide bridges can also be reduced to allow separation by true size.
- This is achieved by using a loading buffer containing an anionic denaturing detergent (SDS) and
- A reducing agent (b-mercaptoethanol or DTT) and
- Boiling prior to loading.
Describe how SDS treatmemnt leads to denaturation of proteins.
- Under denaturing conditions, the strong, negatively charged detergent SDS binds to hydrophobic regions of the proteins causing them to unfold and dissociate from other proteins.
What is the key property of a denatured protein that determines it’s migration pattern?
- SDS confers a negative charge to the polypeptide in proportion to its length.
- Therefore, when a current is applied, migration is determined by molecular weight (MW), not charge of the protein.
What is the key property of a _non-_denatured protein that determines it’s migration pattern?
Samples not exposed to heat or SDS are separated according to the
- net charge
- size
- shape of their native structure
Migration still occurs as most proteins carry a net negative charge in alkaline running buffers.
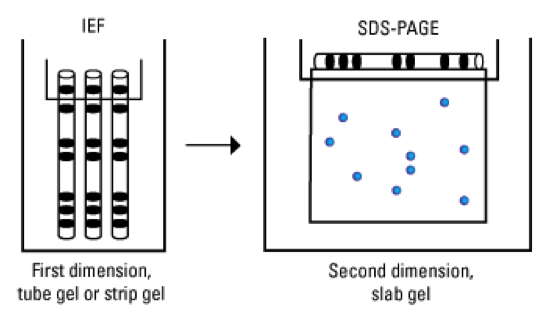
What electrophoresis methods are most commonly utilised when analysing proteins?
- Simple SDS-PAGE
- 2-D gel
- Isoelectric focusing
- SDS-PAGE
What alternative way can the proteins be visualised without requiring a blot?
- Post electrophoresis, visualization of all proteins in the gel can be achieved by staining the gel itself
- e.g. with copper stain (reversible), Coomasie Brilliant Blue or silver staining (both permanent – should only be used post transfer or if blotting not required).
If blotting is required what are the key stages of the blooting process after the electrophoresis is complete?
- Blotting - transfer of proteins onto a membrane
- Blocking - blocking non-target areas of membrane from non-specific antibody binding
- Detection - Visualisation of target peptides using antibodies in combination with x-ray or chemiluminescent based techniques
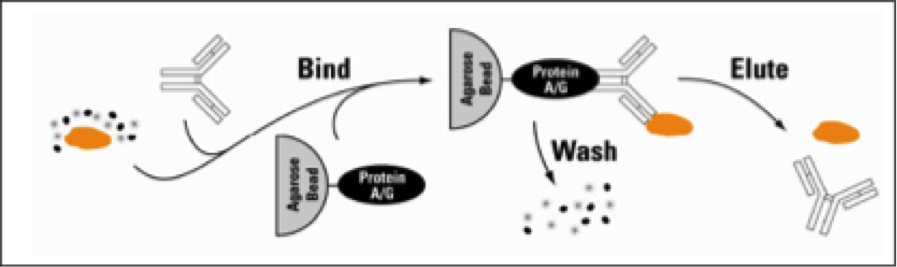
What is protein Immunoprecipitation (IP) and co-immunoprecipitation (co-IP)
- IP is a method used to enrich or purify a specific protein (or a group of proteins) from a complex sample using an antibody immobilized on a solid support (usually agarose resin beads).
- Purified antigens can then be quantified and analysed by Western blotting or other protein techniques such as ELISA.
- Novel isolated proteins can be identified by mass spectrometry.
- co-IP is designed to isolate the antigen (bait) along with any proteins or ligands bound to it (prey) e.g. co-factors, structural partners.
Describe the basic process behind IP.
- Antigen-containing sample (usually a cell lysate)
- Antibody and
- Beaded agarose affinity beads (usually Protein A or G)
- Allowed to bind together.
- Non-bound sample components are washed away
- Antibody and antigen are eluted with abuffer that disrupts the binding interactions.
What is a ‘pull-down’ assay and how is this different?
Similar in concept to Co-IP, but not based on antibody/antigen interaction.
The bait protein is captured to the solid support by a non-antibody affinity system i.e. covalent attachment or an affinity tag bound to the support.
What is a ‘Chromatin-IP (ChIP)’ assay and how is this different?
- Used to investigate the interaction between proteins and DNA in the cell
- DNA and associated proteins are cross-linked
- Specific antibodies are used to immunoprecipitate proteins of interest, along with its associated DNA.
- Allows insight into how genes regulation i.e. where proteins are binding in the genome
What are the primary application of protein-based assays in a genomics laboratory?
Investigation of protein stability, presence, activity, location and affinity can be a useful tool to assess the functional significance of unclassified genetic variants.
Although these assays are very informative, they are rarely amenable to routine screening.
Give some examles of protein-based assays being utilised in genomics laboratory?
- Investigation of protein half-life in BRCA1 and 2 proteins containing missense mutations (mutations influencing protein degradation)
- MMR proteins containing missense mutations in Lynch syndrome that can result in inactivation of enzymatic activity, defective protein-protein interaction, defective subcellular localisation.
- Co-immunoprecipitation of CDKN2A and CDK4/6 in familial melanoma is used to assess if a mutation interferes with CDK binding and thus cell-cycle arrest (Couch et al. 2008).
What is Immunohistochemistry (IHC)?
- Method for localising specific antigens (commonly proteins) in tissues based on antibody-antigen binding.
- This interaction is typically visualised using an antibody conjugated to an enzyme (e.g. peroxidase) that catalyses a colour-producing reaction (detectable via light microscopy), marking the sites of antibody binding.
- Provides information on the presence and localisation of proteins and tissue structure/cellularity
How does Immunohistochemistry differ from immunocytochemistry?
IHC uses sections of tissue (cells surrounded by architecture/matrix as in the body)
Whereas ICC uses intact cells with matrix removed (e.g. cultured cells/suspension/smear).
What type of specimen is used for IHC?
IHC most commonly performed on
- Frozen tissue
- Formalin fixed paraffin-embedded (FFPE) tissue
Fixation preserves protein antigenicity and morphological features of tissues (formalin is best for this).
What are some advantages of using IHC for protein analysis?
Fast turnaround time, cost-effective
Can visualise the protein location within the morphological features of the tissue - provides valuable informaiton for diagnosis




