What are the forms of energy?
- Kinetic energy
- Potential energy
What is kinetic energy?
The energy that an object or a system possesses due to its motion
Whats kinetic energy and its formula?
What is it related to?
For a particle with mass, m, and a velocity, v, we can evaluate the kinetic energy as:
Ekin= 1/2 MV2
Kinetic energy is related to temperature
Whats potential energy?
The stored energy in an object or system due to its position or configuration
Whats chemical bond energy?
Chemical bond energy is the energy stored in chemical bonds
Tell me if the following release or require energy?
- bond formation
- bond cleavage
Bond formation releases energy
Bond cleavage requires energy
What are the 3 forms of system?
- open system
- closed system
- isolated system
Whats an open system?
Both matter and energy can be transferred between the system and the surroundings
Whats a closed system ?
Only energy can be transferred between the system and the surroundings
Whats an isolated system ?
Neither matter nor energy can be transferred between the system and the surroundings
Whats the simplest system?
Isolated system is the simplest system
Energy and contents in isolated system is constant
Whats the 1st law of thermodynamics?
The sum of energies of a system and its surroundings remain constant
- energy cannot be created or destroyed
- energy can only be transferred between different forms
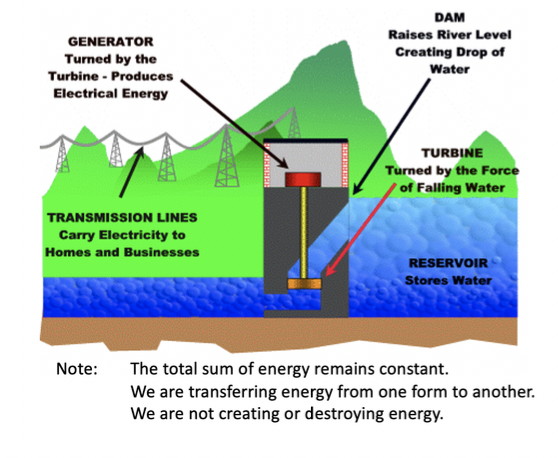
Give a real world example of the 1st law of thermodynamics?
Real world example of this:
- Dam (or waterfall). Raises the water level of the river to create falling water. The reservoir that is formed is, in effect, stored energy.
- Turbine. The force of falling water pushing against the turbine’s blades causes the turbine to spin. The turbine converts the kinetic energy of falling water into mechanical energy.
- Generator. Connected to the turbine by shafts. When the turbine spins the generator will also spin, converting the mechanical energy from the turbine into electrical energy
stored (potential) energy → kinetic energy → mechanical energy → electrical energy
The energy derived from pressure and volume change is given by what?
E= pV (a type of work)
Define enthalpy, H?
H= U + pV
Whats the formula for the change of enthalpy of a system?
∆H= ∆U + ∆(pV)
since in biology most measurements are made at constant pressure, p=pconst we can assume there is no change in p. Therefore…?
∆ (pV) = p∆V (for constant p)
Whats the formula for internal energy?
How can work be described? With this define ∆H?
∆U = w + q
w= -∆(pV)
w= p∆V
therefore;
∆H= w + q + p∆V
∆H= -p∆V + q + p∆V
∆H= q
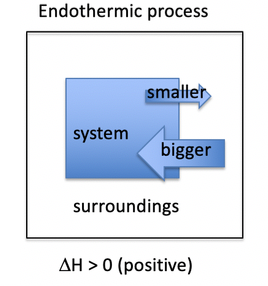
Whats an endothermic reaction?
- Heat flowing into a system from its surroundings is defined as positive, (+q)
- A process that absorbs heat from the surroundings is called endothermic.
- So, the system gains heat, the surroundings cool down.
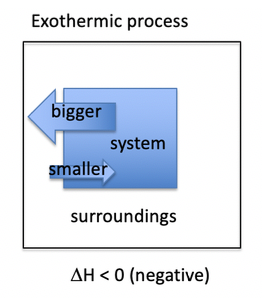
Whats an exothermic reaction?
- When the heat flows out of the system into the surroundings, the heat flow is given a negative value (-q)
- A process that gives heat to the surroundings is called an exothermic process.
- The system loses heat, whereas the surroundings heat up.
Give an example of a real world example of an endothermic and exothermic reaction?
Ice melting= endothermic process
Explosion= exothermic process
Whats the 2nd law of thermodynamics?
In an isolated system the entropy can only ever increase.
If i have a closed system, I need to evaluate the entropy of the system and the surroundings
What are the quantities describing a system and examples?
Macrostates: Temperature, Pressure, Volume
Microstates: Kinetic energy, Force, Velocity
e.g. assign mass, velocity, position for any particle in a system at any time
What can macrostates be described as?
A collection of microstates














