What are colloids?
Colloids consist of particulate matter of certain size range (a dispersed phase) distributed throughout a continuous or dispersion phase
- Colloidal systems have particle sizes that are between molecular (solution) and coarse (suspension) dispersions
A colloid is one type of disperse system;
How do you classify disperse systems based on particles size?
ClassParticle Size Range
Molecular dispersion (solution) <1.0 nm
Colloidal dispersion 1.0nm to 1.0um
Coarse dispersion (suspension) > 0.5 um

What special properties do colloid exhibit?
Large surface-to-volume ratio of the dispersed particles
What are some characteristics of disperse systems? (i.e. what tests can be done for them)
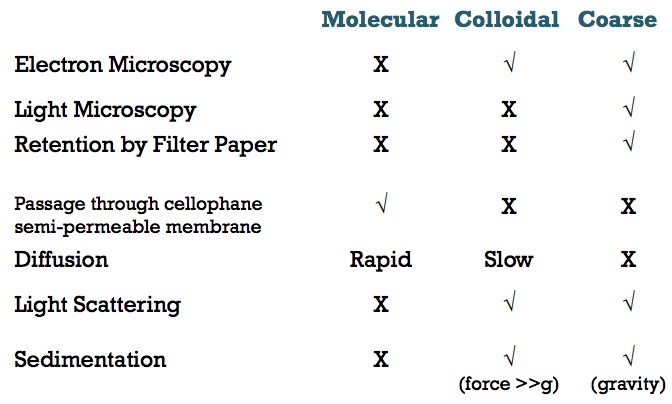
Can colloids pass through the pores in a cellophane membrane?
- No they cannot
- Small molecules such as electrolyes or ions can
What are the shape characteristics of colloids? How does the shape change in repsonse to a dispersion medium?
- Colloidal particles may present in different shapes
- Shape will afect behaviour and property eg shape of proteins
> Shape depends on medium;
- In a hostile medium particles will ball up
- In a medium with which they have affinity, particles will open out and increase surface area
How does the shape of colloids change when in;
A) Kaolin
B) Microcrystalline cellulose
A) Kaolin: plate-like
B) Microcrystalline cellulose: needle-like
How does the method of prepraration influence shape?
- Mechanical disintegration –> usually irregular shapes
- Condensation –> regular shapes (eg colloidal silicon dioxide – spherical)
- Emulsification –> spherical droplets
How does the effect of hydrophilic polymer in water and non-solvent change the shape of the colloid
- Hydrophilic polymers in water – high interaction -> open structure
- Hydrophilic polymer in non-solvent –-> balls up
What are the types of colloidal systems?
Colloids are commonly classified according to their interaction with the medium in which they are dispersed
- Lyophilic (solvent-loving) colloids
- Lyophobic (solvent-hating) colloids
- Association (amphiphilic) colloids
For lyophilic colloids (most interst in pharmacy);
A) What does lyophilic mean?
B) What are some examples?
C) What are the types? What are the differences between the dispersion medium?
D) What are water soluble polymers
E) Is dissolution energetically favourable?
A)
- general term for colloid with affinity for the particular solvent it is in (ie “solvent loving”)
- Dispersed phase has affinity for the dispersion medium
- Forms dispersions easily
B)
- eg proteins, acacia or gelatin in water
- or rubber, polystyrene in organic solvent
C)
- Hydrophilic colloid: water is the dispersion medium
- Lipophilic colloid: organic solvent is the dispersion medium
D)
- Water Soluble Polymers are hydrophilic polymers
- Afiinity of water allows them to be solvated
E)
- Yes, results in spontaneous dispersion
For Lyophobic colloids;
A) What is it?
B) What are some examples?
C) What are the methods used to prepare it?
A)
- Dispersed phase has little attraction for the dispersion medium
B)
Inorganic particles dispersed in water eg;
- Au, Ag or sulfur in water
- o/w, w/o nanoemulsions; nanosuspensions
C)
Dispersion method
- Colloid mills- shearing particles
- Ultrasonic treatment- collapse of cavities in medium break down particles
Condensation method
- Deposition from supersaturated solution as colloidal particles
What are clays ? How are they classified?
Some colloids are difficult to classify as hydrophilic or hydrophobic. Clays exhibit some properties of each, but usually referred to as hydrophobic
- Clays are particulate colloids that swell or break up in the presence of water leaving particles in the colloidal size range that are hydrated but not dissolved
E.g. bentonie swells and divides into particles <1nm thick and Kaolin forms thicker particles (40-200nm)
For Association (amphipillic colloids);
A) How are they formed and what are they made out of?
B) How does concentration affect thier formation
C) What is the significance of CMC
D) What are surfactants
E) Example of real life product?
A)
- Formed by grouping or association of amphiphiles
- Part hydrophilic, part hydrophobic
B)
- At low concentration amphiphiles exist separately (monomers – sub-colloidal) and gather at interface such that lyophobic portion is outside the medium
- As concentration increases, aggregation occurs –> Aggregates (50 or > monomers) are called micelles, whose diameters about 50 Å (5 nm)
C)
- Association colloids can only be formed by amphiphiles or surface active agents (surfactants) at concentration > CMC
- Formation of association colloids is spontaneous provided that the conc. of amphiphile exceeds the CMC
D)
- Molecules or ions with dual characteristics: i.e. one region is solvent-loving and the other is solvent-hating
E)
- Dettol (soap micelles containing chloroxylenol and terpineol
How are amphiphiles classified?
- Amphiphiles may classified as anionic, cationic, nonionic or ampholytic (zwitterionic)
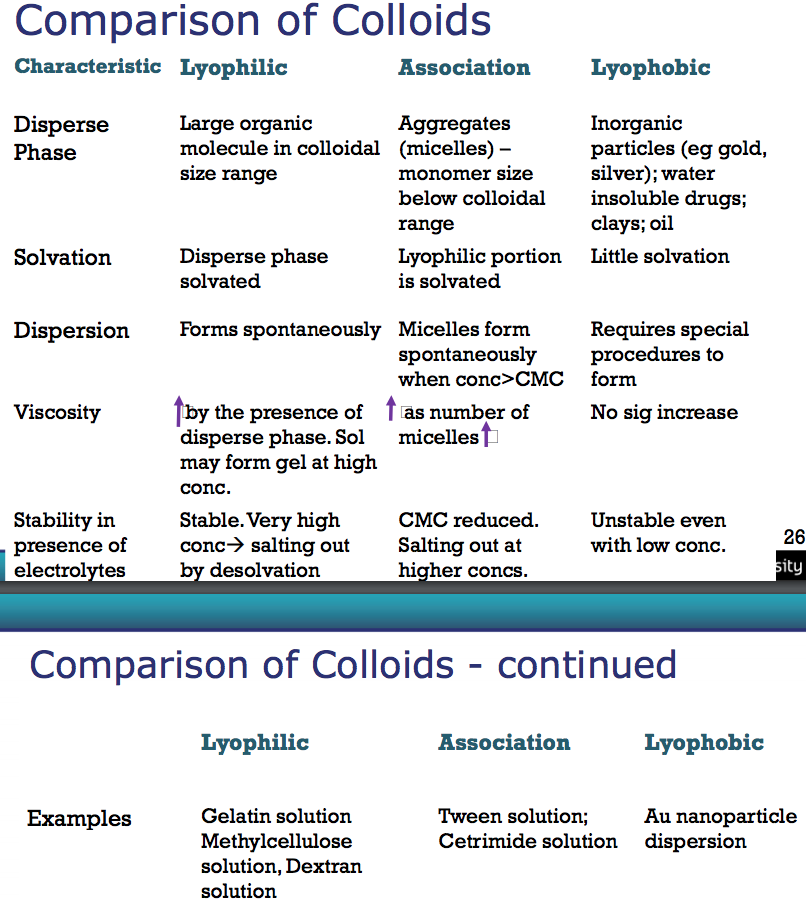
Compare the three types of colloids through;
- Disperse Phase
- Solvation
- Dispersion
- Viscosity
- Stability in presence of electrolytes
- Examples