Whata are the three major properties of colloids?
- Optical
- Kinetic
> Brownian motion
> Diffusion
> Osmotic Pressure
> Sedimernation
> Viscocity
- Electrical
For optical properties of colloids;
A) What does it involve?
B) What does it allow the measuremnt of?
C) How are colloids looked at/visualised?
A)
- Due to their size, colloidal particles are capable of scattering light
- Light scattering –> Turbidity
- Tyndall beam produced by shining light through a colloidal dispersion –> scattering of light gives rise to the appearance of turbidity
B)
- MW of colloids
- Shape and size of colloidal particles
C)
- Not visible with normal optical microscope
- Dark-field microscopy –> observable contrast (ultramicroscope)
> Particles are viewed against a dark background at a right angle to the beam of incidence light. Light scattering causes bright spots corresponing to the particles to be visible
- Electron microscopy –> resolution
What is Brownian motion (kinetic properties)? What decreases it?
Erratic motion (moving in zig-zag path) resulting from random collisions of colloidal particles with molecules of dispersion medium
- Colloidal particles are small enough to be affected by Brownian motion
- High viscosity decreases Brownian motion
What is diffusion? What are some equations that describe it ? How can the diffusion coefficient value be obtained?
Particles migrate from the region of high conc to that of low conc spontaneously. Direct result of Brownian motion. i.e. passive process
- Fick’s First Law
- Stokes-Einstein equation can be used to obtain the particle radius using the diffusion coefficient (D) value
- Diffusion coefficient can also be used to obtain the molecular weight of spherical molecules eg albumin & haemoglobin by an equation
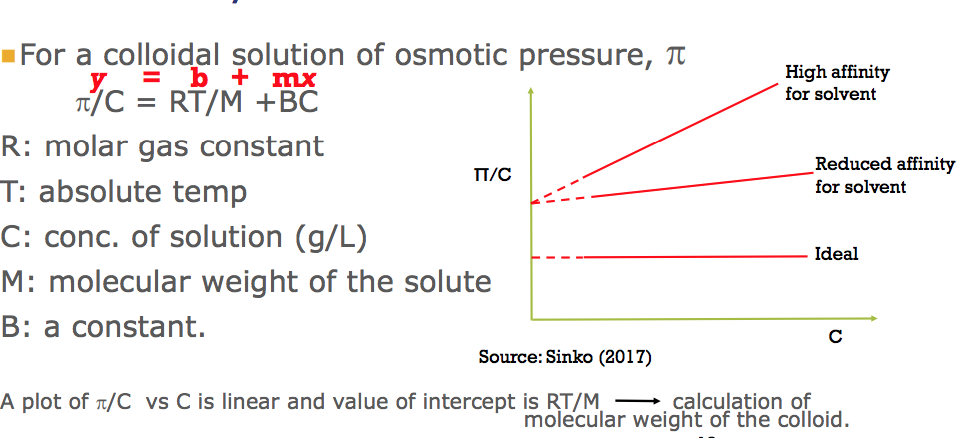
For osmotic pressure (kinetic property);
A) What is it?
B) What is it used for in colloids?
C) What does it provide?
D) Provide the graph for osmotic pressure
A)
- The pressure necessary to to balance the osmotic flow
- The pressure applied to Solution A to prevent solvent moving into Solution A to balance out concentration = OSMOTIC PRESSURE
B)
- Only colligative property with practical value in the study of colloidal particles
- The use of osmotic pressure is limited to a molecular weight range of about 104 –106
> Colligative properties: properties that are dependent on the No. of molecules in solution eg vapor pressure, freezing point and bp and osmotic pressure
C)
- Provides measurable effect
- Eliminates the effect of small molecule to which membrane is permeable
- For Mn< 104, the method won’t work due to the permeability of these molecules
- For Mn > 106, the osmotic pressure is too small to be determined accuratel
For sedimentation;
A) What is it?
B) Do colloidal particles sediment under force of gravity and what is its application
C) When does brownian motion become significant?
D) When does sedimentation require stronger force and what is its application?
A)
See attached image (bigger radius, less time to fall for stokes law)
B)
- Only applicable to the particles> (or equal to) 0.5 micron (μm) which are subject to the force of gravity
- Application: particle sizing
C)
- If particle size < (or equal to) 0.5 micron (μm), Brownian motion becomes significant
D)
- Requires stronger force - ultracentrifuge
- Application: study the micellar properties of compounds; molecular weights of substances

For Viscocity;
A) What is it
B) What is einstein equation?
A)
- an expression of the resistance to flow of a system under applied stress
B)
- Flow for dilute colloidal dispersion of spherical particles
- η = η0 (1+2.5φ)
- η0: viscosity of dispersion medium, η is the viscosity of dispersion medium containing φ volume fraction of particles
A) Compare the viscocity of linear and spherical particles
B) What makes a formulation more viscous
C) What are some applications of viscocity?
A)
- Linear particles – more viscous
- Spherical particles – reduced viscosity
B)
Temperature and pressure
> Increased temp = decreased viscocity
C)
- Means of detecting changes in the shape of flexible colloidal particles and macromolecules
- Avg. Mw of Blood plasma substitutes eg dextrans
- High viscosity leads to gel formation and can affect the performance/usage of the formulations or pharmaceutical products, eg injection, topical preparations
Discuss some general properties of viscosity?
- Hydrophilic polymeric colloids are hydrated and their chains entangle by Brownian motion. Hydration sheath allows the chains to slip past each other without bonding.
- If conditions change to reduce the amount of hydration, neighbouring chains will attract one another and form reversible bonds via Van der Waals’ forces or Hbonding.
- Depending on its type, the polymer may precipitate out, form a colloid-rich viscous layer (coacervate), or set into a semi-solid gel.
- Temperature, concentration, molecular weight and the nature of the polymer and solvent are factors that affect phase separation, precipitation and gelation
- Gelation is more likely with higher MW polymers, increased concentration and conditions that reduce solubility of the polymer


