A) What are surfactants?
B) What are their regions made of ?
C) How are they classified and give the advantages of one of the classifications
A)
- Amphipathic molecules: structure is characterised by having two distinct regions: hydrophilic & hydrophobic moieties
B)
Hydrophilic portion: cationic, anionic , non-ionic or ampholytic
Hydrophobic portion: Saturated or unsaturated hydrocarbon chains, or heterocyclic or aromatic ring systems
C)
- Anionic
- Cationic
- Non-ionic: R(OCH2CH2) n OH n=4-100
- Ampholytic
Advantages of non-ionic: stability, compatibility; less irritant; less toxic
What does the surface activity of surfactants dependent on? Give an example
Surface activity of a particular surfactant is dependent on the balance between its hydrophilic and hydrophobic properties
- Increase in hydrophilicity results in decreased surface activity
- an increase in the length of ethylene oxide chain of an non-ionic surfactant leads to an increase in both surface tension and the CMC
What is traube’s rule (Surface phenomena)?
Describes the relationship between hydrocarbon chain length and surface/interfacial activity of surfactants
- Each extra CH2 group in the hydrocarbon chain results in three times increase in surface activity of surfactants, therefore, only 1/3 molar concentration would be required to produce the same amount of surface tension reduction
What are some purposes surfactants are used for? (due to the hydrophilic and lipophilic properties they exhibit)
- Solubilising
- Emulsifying
- Wetting
- Detergent
What is the HLB system (hydrophile-lipophile balance)?
A measure of relative contributions of hydrophilic and lipophilic regions of the molecule
> Arbitary scale (0-20) calculated using empirical formulae
- High value = hydrophilic
- Low value = lipophilic

Different non-ionic surfactants can be blended to give any desired HLB. A required HLB can be calculated for a mixture of oily ingredients.
What are the steps required to calculate the required HLB? Provide an HLB calculation example
- Determine emulsion type w/o or o/w
- Calculate the total mass of the oily phase ingredients in the formula
- Using published values of HLB for oily phase ingredients, determine the total required HLB (RHLB) for the oily phase. This is a weighted mean determined by multiplying the proportion of each oily phase ingredient by its HLB and adding them together.

If you have a mixture of surfactants, the overall HLB can be determined using an algebraic mean:
x HLBA + (1-x) HLBB (x: fraction of surfactant A) (1-x fraction of surfactant B)
Q: Provide an example of a calculation using this formula

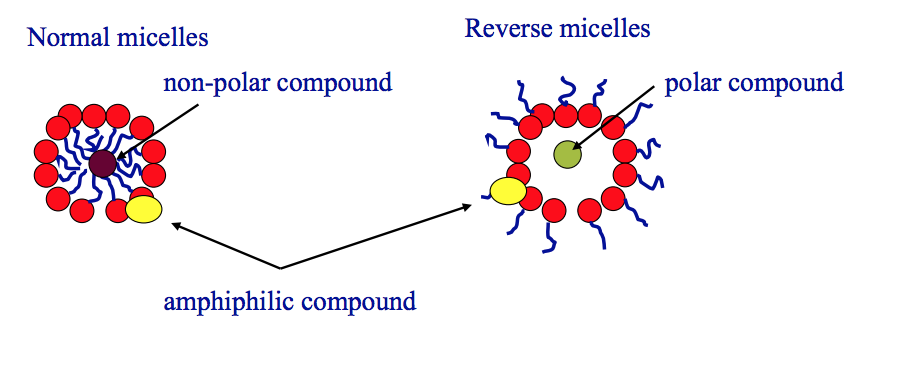
How do micelles form? What does this lead to?
- Micelles form after the surface (L/G interface) becomes saturated with soluble amphiphiles (surfactant)
- Micelles form in water with the hydrophobic portions of the surfactant molecules directed towards the centre of a sphere, or similar shape, shielding them from contact with water
- Micelles form spontaneously because their formation is energetically favourable. ie forming a micelle leads to a system of lower free energy
For water structure;
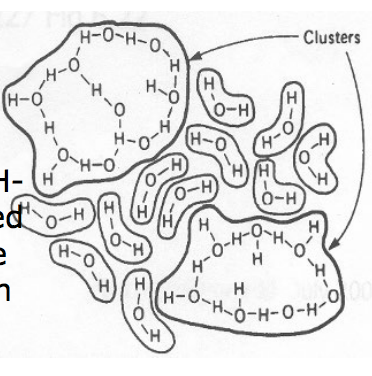
A) What is the flickering cluster theory
B) What happens when a non-polar molecule is added to water?
A)
- Water has areas of structure (ice-like clusters of multiple H-bonded molecules) interspersed with free water molecules. The clusters breakdown and reform –> flickering clusters
B)
- If a non-polar molecule is added to water, the non-polar part will seek out the ice-like structures where it can fit without breaking H-bonds. So hydrophobic portions of molecules in water are always surrounded by structured water
A) What is the gibbs free energy equation?
B) How does it relate to micelles being formed and the flickering cluster theory?
A)
ΔG = ΔH - TΔS (Describes energy available to do work –> determine spontaneity of reaction)
- ΔG : change of free energy
- ΔH: change of enthalpy
- ΔS: change of entropy
ΔS is the most important in determining ΔG in surfactant solution
B)
- As a micelle forms, the non-polar groups approach each other until they make contact.
- The formation of hydrophobic bonds between non-polar portion of molecules reduces the total number of H2O molecules in contact with non-polar groups
- Results in a loss of the ice-like structure which always surrounds the hydrophobic groups. Loss of structure = increased entropy
- Increased entropy = decreased ΔG (lower state of free energy)
A) What is the driving force for micellisation
B) How does the shape of micelles change as CMC changes?
A)
- This loss of structured water around hydrophobic groups is the driving force for micellisation
B)
- Micelles adopt a spherical or near spherical shape at CMC.
- At higher concentrations a gradual change in micellar shape may occur eg from spherical form to cylindrical or laminar form
A) Are micelles and monomers the same thing?
B) What type of micelles are formed in non-aqueous solution?
A) NO THEY ARE NOT
- Micelles are in dynamic equilibrium with monomer molecules in solution, continuously breaking down and reforming. (micelles –>–< monomoers)
- Micelles are not solid particles
B)
- In non-aqueous solution: “inverted micelles” may form, ie hydrophilic groups forming the micellar core are shielded from the non-aqueous environment by the hydrophobic chains
What are the 5 factors affecting CMC and micellar size? Describe how it does so for each one.
1) Structure of the hydrophobic group
- Hydrocarbon chains in micelles = hydrophobic groups
- Longer the hydrocarbon chain of a surfactant = lower CMC and a larger micellar size
- Compounds with rigid aromatic or hetero-aromatic ring structures (eg purines, many dyes) associate by a non- micellar process. They do not exhibit a CMC.
2) Nature of hydrophilic group
- Ionic hydrophilic = ionic heads oppose micelle formation due to electrostatic repulsion between adjacent charge groups –> higher CMC, fewer molecules in micelle.
- Non-ionic hydrophilic = lower CMC values than ionic. CMC is affected by hydrophilic chain length (eg polyoxyethylene chain
- Longer hydrophilic chain –> the higher the CMC
3) Nature of counter-ions
- the more weakly hydrated a counterion, the larger the micelles formed by surfactand (can be absorbed more readily in the micellar surface to reduce charge repulsion between polar groups)
- Cl- < Br- < I- ; Na+ < K+ < Cs+ (Chlorine being the weakest)
- Organic counter-ions produce a lower CMC and a greater increase in micellar size than inorganic counter-ions.
4) Addition of electrolytes
- For ionic surfactants: decrease CMC and increase micellar size
- For non-ionic surfactants: little effect.
5) Effect of temperature
- Ionic surfactants: temp has smaller effect
- Non-ionic surfactants: heating causes solution to become turbid at a characteristic temp called the cloud point (reversible)
> Turbidity is due to separation of solution into two phases
A) What is solubilisation?
B) What is its relationship with micelles?
A)
- water-insoluble or partly soluble substances are brought into aqueous solution by incorporation into micelles
- Centre of each micelle is an ideal environemnt to dissolve a non-polar drug
B)
SOLUBILISATION DOES NOT OCCUR UNTIL THE MICELLES ARE FORMED
- Surfactant should be of correct HLB value (15-18)
- Concentration of surfactant must be > CMC
- Above CMC = amount of substance solubilized increases as the surfactant concentration increases (ie as the no. of micelles increased)
A) How is solubility of a poorly soluble compound increased?
B) What is the maximum additive concentration (MAC)?
C) What is equilbirum
A)
- Increases as a result of solublisation in the micelles
B)
- Max amount of solubilisate that can be incorporated into a given system at a fixed conc of surfactant
C)
Free drug –>–< Drug/Micelle –>–< Free surfactant
For Solubilisation;
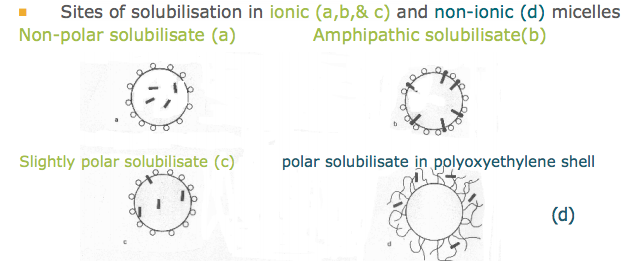
A) What are the sites of solubilisation in ionic and non-ionic micelles
B) What is aggregation no?
C) Does the size of micelles change as more solubilisates are in the core? Why?
A)
See attached image
B)
- No of surfactant molecules per micelle
C)
- Yes, size of micelles increase because of enlarged core & increased aggregation No
What are the factors affecting solubilisation?
- Nature of surfactant
- CH2 chain length: Solubilisate located deep within the micelle, solubilisation capacity increases with increase in alkyl chain length, within limits
- Ethylene oxide chain length: aggregation number decreases with the chain length; micelle size decreases while total number of micelles increases.
> Total amount solubilised/mole surfactant increases although solubilised molecule per micelle decreases
- Effect of temperature
- Amount solubilised increases as temp. increases (increase in micellar size with some non-ionic surfactant)
- drug solubility also changes; non-ionic surfactants undergo clouding on heating
- pH
* Alters the equilibrium between ionised and unionised drug;
> affects the drug solubility in water
> modifies the partitioning of the drug between the micellar and the aqueous phases
What are some disadvantages of drug solubilisation by surfactants?
- Conc. of surfactant must be maintained above CMC level
> Caution with dilution
- Biological influence of surfactants on drug absorption and bioavailability


