What are the pharmaceutical applications of surfactants?
- Wetting
- Stabilisation
- Emulsifiers
For Wetting agents (HLB 7-9)
A) How does it work?
B) What are some pharmaceutical applications of wetting?
C) What is the most important action of the wetting agent? What is the contact angle?
D) How does it increase the dissolution rate of a solid drug?
E) Does it alter the solubility of a solid?
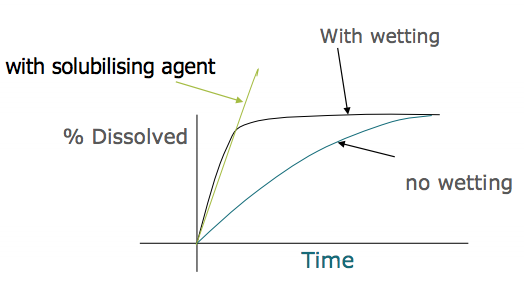
F) Draw a graph that shows % dissolved of a drug vs time (with solubilising agent, with wetting and with no wetting)
A)
- adsorb at L/V & S/L interfaces & reduce interfacial tension
- lowers contact angle
- Aids in displacing an air phase with a liquid phase at the surface of a solid
B)
- Displace air from surface of sulfur, charcoal and other powders for dispersing drug powders in liquid vehicles
- Cotton pad and bandages
C)
- Most important action of wetting agent is to lower the contact angle between the surface and wetting liquid
- Contact angle θ: Angle between a liquid droplet and surface over which it spreads –> describes wettability of a powder
- Decrease contact angle = more wettable (θ = 00 complete wetting)
- Increased contact angle = no wetting (θ =1800 no wetting occurred
D)
- Aiding penetration of fluid
- Reducing the tendency of particle aggregation, effectively increases the surface area of solids exposed to fluid
E)
- Wetting agent does not alter solubility of a solid
F)
See attached image ( can see wetting agent does not alter solubility of a solid)
What are some examples of solubilsing agents?
polysorbates in aqueous formulation of water-insoluble vitamins (Vit A, D E & K); iodophores: iodine- surfactant
What are some examples of emulsifying agents?
Eg cetomacrogol emulsifying wax used in lotion or creams
What are some examples of suspension stabilisers?
eg polyoxyethylene-polyoxypropylene used for steric stabilisation and controlled flocculation
What are some examples of detergents
Cetrimide shampoo
What are some examples of disinfectants?
Ampholytic surfactant such as Dodicin, derivatives of long chain N-substituted amino acids
For stabilising drugs;
1a) Does solubilisation of the drug by micelles improve its stability? What affects this?
For Base-catalysed hydrolysis;
2a) What do anionic micelles do?
2b) What do cationic micelles do?
1a)
- Yes, solubilisation of drug by micelles can improve the stability of a drug
- The deeper into the hydrocarbon core of a micelle the drug is located, the more likely it is to be better protected eg benzocaine vs homatrophine
2a)
- Anionic micelles give an enhaced protection due to the repulsion of the attacking OH- groups
2b)
- Cationic micelles were found also to be protective, suggesting the positively charged head groups hold the OH- groups thus blocking their penetration into the micelle.
> Eg micellar solution of penicillin G is 2.5 times as stable as monomeric solution
- Surfactants are not inert
- They are capable of increasing, decreasing or exerting no influence on bioavailability of drug
- They can also affect biological activity of drug by influencing drug metabolising enzymes or drug binding to receptors
What is the effect of surfactants on drug absorption;
A) When surfactants are at low conc?
B) When the surfactants are above CMC level ?
C) When the surfactants are at a very high level?
D) What are some thing that occur when surfactants are included in the formulation?
A)
- Low conc –> disrupt cell membrane –> enhancing drug penetration and absoprtion of drug across GI barrier
B)
- Drug partitioning between micelle and aqueous phase
- For soluble drug: inhibition of absorption
- For poorly soluble drug: dissolution and absorption rates are increased
C)
- increased amount required for solubilisation
- Decreasing chemical potential of drug
- Decreased drug absorption
D)
- Wetting effect
- Preventing aggregation (phenacetin + tween 80)
- Increasing drug solubility
What are some factors that dictate the ability of a surfactant to have influence?
- Properties & conc. of surfactant
- Nature of drug
- Type of biological membrane is involved
- Surfactant may exert drug action itself (quartenary ammonium compounds)
For Emulsions;
A) What two phases do they have? What do they consist of?
B) Why are emulsions thermodynamically unstable?
A)
- Disperse phase (inner) and continuous phase (outer)
- Emulsions consist of droplets of one phase (oil or water) dispered throughout another phase (water or oil)
B)
- Large SA due to small size and large number of liquid droplets
- Interfacial tension between inner (disperse) and outer (continuous) phases. (Cohesive force between molecules of each separate phase are greater than the adhesive forces between the two liquids)
How can the stability of emulsions be improved?
Stability can be improved by introducing an emulsifying agent (mixtures of them)
> Form films at the interface beween phases (protective layer)
- Surfactants: adsorbed at o/w interfaces to form mono-molecular films
- Hydrophilic colloids: form a multi-molecular film around the dispersed droplets of oil in an o/w emulsion
- Finely divided solid particles: adsorbed at the interface between two immiscible liquid phases and form a film of particles around the dispersed globules
How do emulsifying agents stabilise emulsions?
- reduction of interfacial tension –> reduces surface free energy
- provision of electrostatic repulsion –> if emulsying agent is charged
- steric stabilisation –> prevents doplets from approaching each other
- Some surfactants can also increased the viscocity of emulsions –> impede movement of droplets
What are the 3 emulsions stabiliastion theories? Describe how they work
1.
- Surface tension theory: Surface active agents lower interfacial tension of two immiscible liquids; as a result, they facilitate the break up of large droplets into small ones and maintain their dispersion.
- Interfacial film theory: Emulsifying agents adsorb at the interface between oil & water, surrounding the internal phase as a thin layer of film. The film, acting as a physical barrier, prevents the contact and coalescing of the dispersed phase.
- Oriented-wedge theory: Surfactant emulsifying agents form a monomolecular layer which curves around the droplet of the internal (disperse) phase of the emulsion. The emulsifying agents orientate themselves in a manner reflective of their solubility in the particular phase. If it is more water soluble, it would be embedded more deeply in water phase
For Surfactants (Emulsifying agent)
A) How does it work?
B) What properties should it have
C) What determines if a surfactant will stabilise a emulsion? Give an example
coalescence: two or more droplets merge to from one droplet
A)
- Reduce interfacial tension
- Form a coherent monolayer (interfacial film) at the surface of droplets to reduce the possibility of coalescence – the main factor for emulsion stabilisation
B)
- Film should be tough, flexible, closely packed and reforming rapidly if broken or disturbed
C)
- Depends on the type of film formed at o/w interface
- Sodium dodecyl sulphate (SDS) forms a gaseous film at o/w interface and does not form a stable emulsion on its own
- However SDS + Cetostearyl alcohol form condensed film –> stable emulsion
Ionic surfactants provide electrostatic repulsion
What is Bancroft’s Rule? How does HLB affect what type of emulsion occurs?
Bancrofts rule: The external phase is the one in which the emulsifying agent is most soluble.
- High HLB (hydrophilic) –> O/W emulsion (water soluble) = SDS
- Low HLB (lipophilic) –> W/O (water insoluble
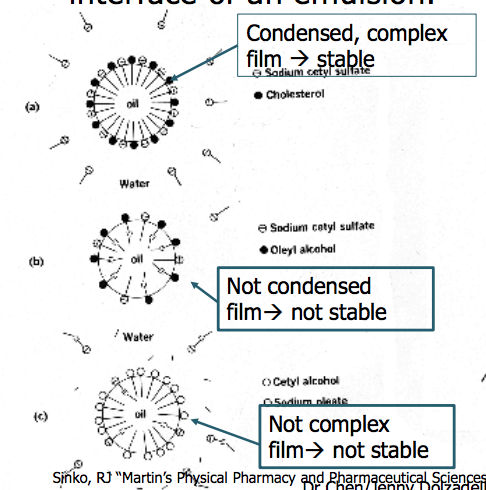
Combinations of emulsifying agents are used frequently, how do they work together? Give an example.
- A hydrophilic emulsifier in the aqueous phase and a hydrophobic agent in the oil phase to form a complex film at interface.
- Eg combination of sodium cetyl sulfate(SCS) and cholesterols
Complex film formed is of high viscosity, flexible, resists rupture and gives an interfacial tension lower than that produced by either component alone
Give some examples of emulsifying waxes (combinations of emulgents)
- Anionic Emulsifying Wax : 10% SDS, 90% cetostearyl alcohol
- Cationic emulsifying wax: 10% cetrimide,90% cetostearyl alcohol
- Non-ionic emulsifying wax: 20% cetomacrogol 1000, 80% cetostearyl alcohol
What are the combinations of emulsiying agents at O-W interface of an emulsion?
See attached image
For Hydrophilic colloids (emulsifying agents)
A) What are some examples?
B) How do they work
C) What are some advantages?
A)
- proteins (eg gelatin) or polysaccharides (eg Acacia, methylcellulose
- Used in liquid paraffin emulsions
B)
- Adsorb at the o/w interface but does not cause lowering of interfacial tension
- Exhibit little surface activity
- Form multilayers or multi-molecular film at the interface - mechanical barriers to coalescence.
C)
- If HC contain ionic group (e.g. protein and acacia), they provide electrostatic repulsion as an additional barrier
- Tend to promote the formation of o/w emulsion (hydrophilic nature)
- Increase the viscosity of an emulsion to improve its stability
For solid particles in an emulsion stabilisation (emulsifying agent)
A) How does it work
B) Where does it act on
C) How do O/W emulsions form as a result?
A)
- Finely divided solid particles that are wetted to some degree by both oil and water can act as emulsifying agents.
B)
- They are concentrated at the interface and form a particulate film around dispersed droplet to prevent coalescence
C)
- Those wetted preferentially by water form o/w emulsions (aluminium and magensium hydroxides are wetted by water –> form stabilised o/w emulsions)
- On the other hand, carbon black is more readily wetted by oil thus stabilises w/o emulsions


