Intrepret quantitve measures of risk (risk ratio, odds ratio, relative and absolute risk)

What is relative and absolute risk?
- *- Relative Risk:** the ratio of the probability of developing, in a specified period of time, an outcome among those receiving the treatment of interest or exposed to a risk factor, compared with the probability of developing the outcome if the risk factor or intervention is not present.
- *- Absolute risk:** The observed or calculated probability of an event in the population under study.
Absolute risk difference: the difference in the risk for disease or death between an exposed population and an unexposed population.
Absolute risk reduction (ARR): the difference in the rates of adverse events between study and control populations (ie: the difference in risk between the control group and the treated group: ARR=CER-EER)
- What factors influence perception of risk?
- We all interpret risk differently
- More willing to accept risks that are voluntary, controllable, familiar, non catastrophic, fair in
the way benefit and risk are distributed i.e. more risk = more general benefit. General anesthetic and surgery.
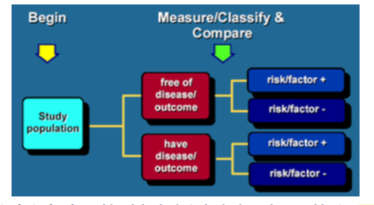
What is a cross-sectional study?
A study that examines the relationship between diseases (or other health related characteristics) and other variables of interest in a particular population at one time. Cross-sectional studies may be used to estimat ethe prevalence of disease, but not the incidence of disease.
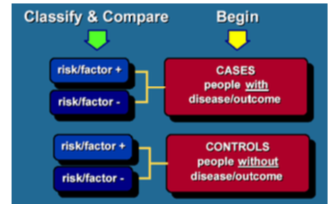
What is a case control study?
An epidemiological study design where subjects are recruited on the basis of the presence of absence of disease (case and controls) and exposure is measured retrospectively. In this way it is possible to estimate the risk of disease associated with expsoure usually by calculating an odds ratio.
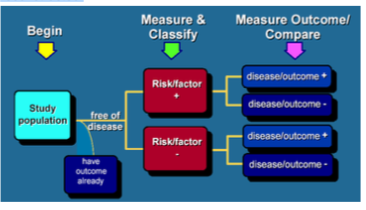
What is a cohort study?
An epidemiological study whereby a defined subset of the populationc can be identified and classified according to exposure status. The mean feature of a cohort study is that it can determine the incidence rate of disease amongst exposed and unexpsoed individuals. Common synonyms include longitudinal or follow-up studies.
What is occupational cohort.
The definition of the cohort is based primarily on a common occupational exposure e.g. workers in the nuclear power industry. In this way the risk of diease can be compared with the general population or other occupational groups to determine the occupational risk.
What is prospective cohort?
Healthy individuals are recruited, though some may already have the disease at baseline, and followed up for future disease occurrence, often for decades. Exposure is status is measured at baseline and repeat measures for change in exposure may be undertaken over the follow-up period
What is retrospective cohort?
Disease status for a defined subset of the population is ascertained at baseline but this is linked to pre-existing historical data on exposure either from routine records or an earlier research project so that the cohort’s experience of disease risk can be reconstructed.
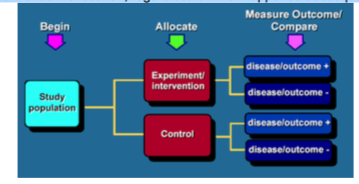
What is a randomised controlled trial?
- Randomised controlled trial: A study in which individuals are randomly allocated to two or more groups. Often, one of these groups will be the treatment group while the other will be a placebo group that receives no treatment other than standard care. The key elements of randomised controlled trials are:
The comparison of a group receiving the treatment (or intervention) under evaluation, with a control group receiving either best practice, or an inactive intervention.
Use of a randomisation scheme to ensure that no systematic differences, in either known or unknown prognostic factors, arise during allocation between the groups. This should ensure that estimated treatment effects are not biased by confounding factors.
Allocation concealment: Successful implementation of a randomisation scheme depends on making sure that those responsible for recruiting and allocating participants to the trial have no prior knowledge about which intervention they will receive. This is called allocation concealment.
Where possible, a double blind design, in which neither participants nor study personnel knows what treatment has been received until the “code is broken” after the end of the trial. This is achieved by using a placebo. If a double-blind design is not possible then outcome assessment should be done by an investigator blind to the treatment received.
An intention to treat analysis in which the treatment and control groups are analysed with respect to their random allocation, regardless of what happened subsequently.
What is a systematic review and meta-analysis?
A systematic review ‘does what it says on the tin’, it is an attempt to systematically review the research evidence on a particular topic – systematic means a defined methodology that could be repeated by someone else and retrieve the same publications and the same results. The results of the review might be expressed in words – sometimes it is also possible to mathematically combine the individual results of studies in the review to get a more precise estimate of what is happening. This mathematical combination is called meta-analysis. It isn’t always possible, or correct, to summarise the findings of a review as a meta-analysis – but in the area of randomised controlled trials it is an excellent way of getting an accurate and precise idea of how well a treatment works.
So bringing evidence together in a systematic way is always a good idea – sometimes this can be combined with meta-analysis.
- Focus group - An evaluation activity comprising of a semi-structure discussion with a group of people. Focus groups, comprising of stakeholders, are used to inform test-designers on the significance of each topic to be administered within a certification exam.
- Participant / non-participant observation:
- Participant observation - A research method involving direct participation of the
researcher in the events being studied. The researcher may either reveal or hide the true reason for involvement.
- Non-participant Observation – Researchers do not partake in the activity of the
study.
What is bias?
Bias is defined as ‘systematic error’ in measurement
- This is distinct from ‘random error’
- Think about when we ‘measured’ smoking in an earlier plenary …
- You were classified as smoker/non-smoker
- This will have been measured with error, some of you were misclassified
- Likely that (slightly) but systematically under-estimated smoking prevalence
Discuss the healthcare resource implications of orfgan transplantation and blood donation.
- How are blood and organs rationed and what are the issues surrounding payment for body parts?
- What is a market? - A market is a network of buyers and sellers who exchange goods and services. All markets are regulated by explicit and implicit rules (ethics)
- Exchange or trading may be based on money or may be based on other factors such as altruism and trust
- Altruism “Regard for others as a principle of action” – you may care for others as a fellow human, you may also expect reciprocity if you are in need
- Trust is a vital issue in the blood and organ donation sphere - Whether we use gifts or commercial mechanisms to create an adequate supply of blood and body parts we rely
on trust to ensure high quality and a timely supply
- The market for blood - Individuals have 8-10 pints of blood and can restore blood levels
after the loss of 2-3 pints with the aid of saline and iron tablets
- With greater blood loss than this transfusion is needed. The top demands for this come from:
general surgery, general medicine, cardiothorasics, orthopaedics and blood diseases
- Risks come from giving people the wrong blood group or from infections such as Hep B or
Hep C. The risk of Hep B is 1 in 900,000
- Supply of blood - The UK has a voluntary system of giving with 2 million people donating annually
- Donation, storage and distribution is publicly funded. I.e. it isn’t a free or costless system
- Stock levels are usually adequate but when there are significant local problems there are calls
to the public for more donations
- The case against a market for blood (Titmuss, 1970) - Represses altruism
- Erodes the “sense of community”
- Sanctions profits in hospitals and clinics and subjects medicine to marketplace rules
- Increases blood supply from the poor, unskilled and unemployed: redistributes blood from the
poor to the rich and may be risky
- The case for a market for blood (Cooper and Culyer) - Blood is no different from any other tradable product
- Supply can be increased by removing obsicles to donors
- This needs to be linked with either offering direct payment of exemption from payment in the
future on the basis of blood donated
- It is not clear from the evidence which method is cheapest and offers the highest supply
quality
- The market for used body parts - Kidneys can be obtained from cadavers and living
- Livers can be obtained from cadavers and from living donors
- Heart and heart and lungs
- Lung transplants
- Corneas
donors
- Alternatives to transplant - Mechanical maintenance for kidneys: dialysis and Continual Ambulatory Peritoneal Dialysis (CAPD)
- Costs and benefits: Quality of life after a transplant is significantly better than on dialysis I.e. a clinically affective transplant is more cost effective
- Younger spare parts give better outcomes: road traffic policy and dead from subarachnoid haemorrhage
- The problem is the lack of used parts to transplant
- Increasing the supply of used body parts - Altruism: Opt in with donor cards or opt out if you object. The use of transplant co-ordinators
- Financial incentives: as used in the blood market in the USA. May be issues of quality control and it doesn’t always help patients if doctors stand in the way because they find commerce ethically offensive
- The case for paying for donations of used body parts - Increases the supply
- Increases the rate of transplantation, which is a cost effective way of treating patients, adding
years to life and increasing quality of life
- Rationing - Rationing is a feature of all healthcare systems. The USA has 45million people
without insurance and a federal renal transplant program
- Rationing involves depriving patients of care from which they would benefit and which they
would like to consume
- The issues is not whether to ration care, but how to do so
- Methods of rationing - Ability to pay: this method favours the rich and is accepted in most markets though not usually in healthcare
- “Need”? but what is need? Ability to benefit as measured against cost. Therefore this method favours those who can benefit the most per unit cost
- How to select who benefits most - Age
- Behaviour: smoking, alcohol etc
- Family circumstances
- Social class (may also be related to behaviour)
- What else?
- The markets in summary - The supply of blood just about matches demand
- The supply of organse does not match demand and as such people are left to die from liver,
kidney, heart and lung failure
- The price mechanism would improve supply but societies prefer altruism
- Medical issues - Voltaire said “the role of the doctors is simply to amuse the patient while nature takes its course”
- The role of the modern doctor is not simple to treat the sick but to decide which patients can be treated cost effectively. I.e. to select who lives in what level of pain and to decide who dies. Whether this rationing is implicit or explicit it is unavoidable
- Conflicting perspectives on who to treat - The use of evidence of clinical effectiveness
- The use of evidence of cost effectiveness
- The individual Hippocratic ethic and the social ethics of economists and public health
physicians
- Conclusions - Altruism produces a supply of blood and spare parts
- The supply, particularly in the spare parts market, does not meet demand and so rationing occurs, even though transplantation may be cost effective
- Paying donors would probably increase the supply of parts but this is resisted on ethical grounds, thus creating avoidable mortality.


