List the Non-covalent (NC) Intermolecular Forces:
- Protein folding
- Protein-protein interactions
- DNA structure formation
- Protein-DNA interactions
- Enzyme-substrate interactions
- Drug interactions with protein targets
Key Noncovalent Interactions
- Ionic interactions
- Hydrogen Bonding
- Van der Waals forces
Force between oppositely charged species
Ionic interactions
A type of weak noncovalent force of attraction between a H atom attached to a N, O, or F aton and a N, O, or F aton on another molecule (or part of the same molecule)
Hydrogen Bonding
Weak interaction between uncharged but polarizable groups
Van der Waals forces
A _________can form between residues of opposite charge in protein
salt bridge
The balance between attractive and repulsive forces in van der Waals forces determines the optimal _____________
VdW contact distance—“van der Waals radius”
_____ is an indirect effect of the high polarity of water
The hydrophobic effect
Water is ________ when __________ or groups cluster together
less ordered (entropy is higher); hydrophobic molecules
Know as the “helix breaker”
Proline
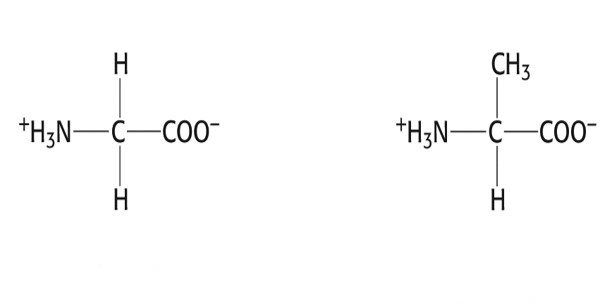
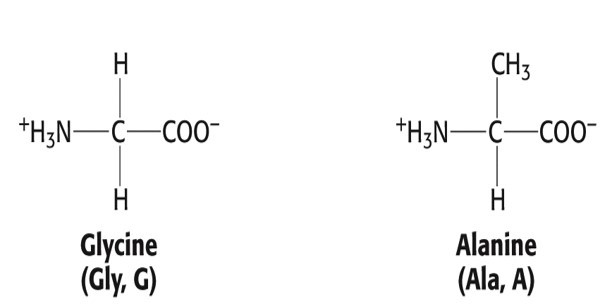
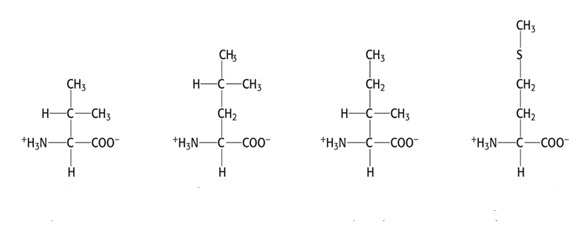
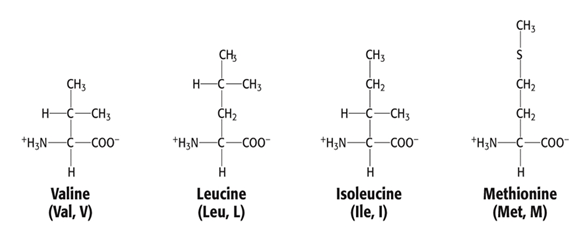
Group 1 AAs: Nonpolar, aliphatic R group
- Glycine, Gly, G
- Alanine, Ala, A
- Valine, Val, V
- Leucine, Leu, L
- Isoleucine, Ile, I
- Methionine, Met, M
- Proline, Pro, P
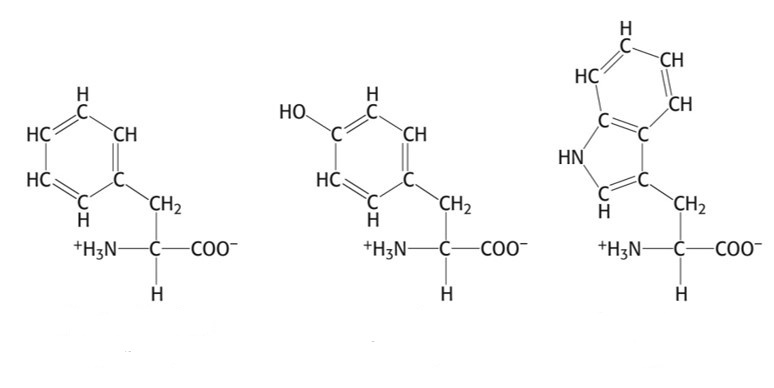
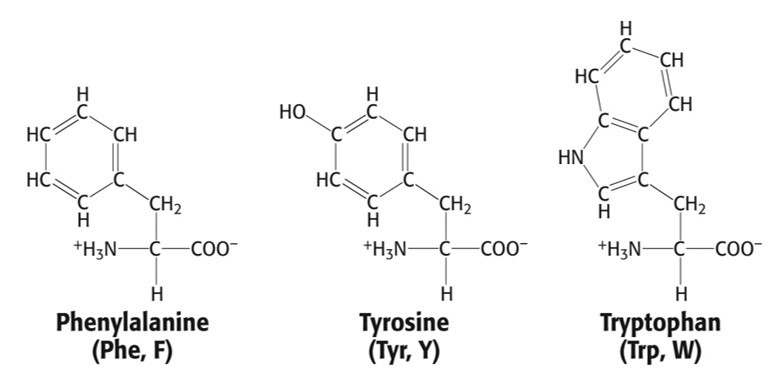
Group 2: Aromatic Side Chain
- Phenylalanine, Phe, F
- Tyrosine, Tyr, Y
- Tryptophan, Trp, W
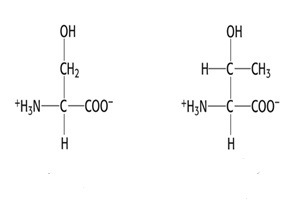
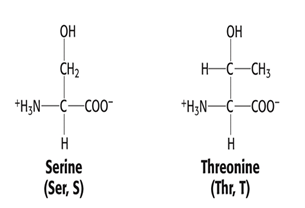
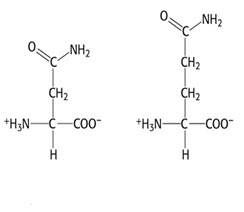
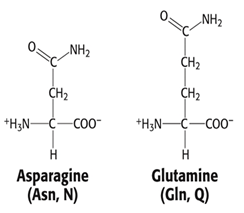
Group 3: Polar, uncharged side chain
- Serine, Ser, S
- Threonine, Thr, T
- Asparagine, Asn, N
- Glutamime, Gln, Q
- Cysteine, Cys, C
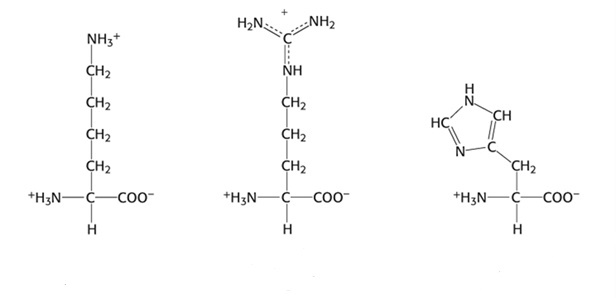
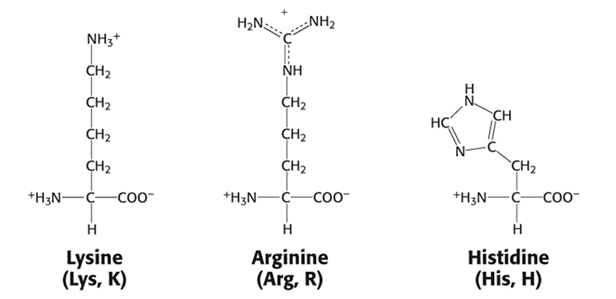
Group 4: Positively charged Side Chain (Basic)
- Lysine, Lys, K
- Arginine, Arg, R
- Histidine, His, H
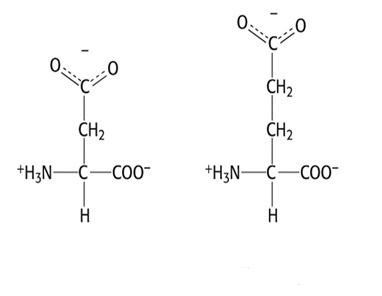
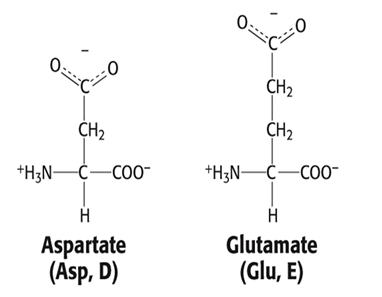
Group 5: Negatively Charged Side Chain (Acidic)
- Aspartate, Asp, D
- Glutamate, Glu, E
Has an imino group
Proline
Aromatic Side Chain that absorbs the most light
Trp




