State the equation of state for an ideal gas
PV = nRT
What is an ideal gas?
A gas that agrees with the ideal gas equation
What are the differences between ideal gases and real gases?
- Real gases are affected by high pressures and densities and low temperatures whereas ideal gases are not
- Ideal gases do not liquefy at low temperatures
What is absolute zero temperature?
A state at which all molecular motion has stopped, the lowest possible temperature: -273.15°C or 0 K
What is the Kelvin scale of temperature based on?
The behaviour of an ideal gas (pressure is proportional to the temperature)
Define system and surroundings
A system is the thing in question (in this case gases)
The surroundings are everything else outside of the system (environment)
What is the expression for the work involved in a volume change of a gas at constant pressure?
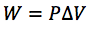
What is the first law of thermodynamics?
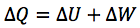
What is the first law of thermodynamics a statement of?
The principle of energy conservation
Draw the isochoric, isobaric, isothermal, and adiabatic changes of state of an ideal gas on P-V diagrams

Describe the isochoric change of state of an ideal gas
- There is no work done by or on the gas → no change in volume
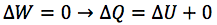
Describe the isobaric change of state of an ideal gas
- Any expansion or compression occurs at constant pressure
- Occurs when gases are allowed to expand freely
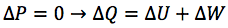
Describe the isothermal change of state of an ideal gas
- No change in the internal energy of the gas → temperature is constant
- The work done on the gas is all transferred away from the gas as thermal energy
- To be isothermal the change must be as slow as possible
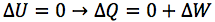
Describe the adiabatic change of state of an ideal gas
- No thermal energy is transferred between the gas and the surroundings
- All the work done by the gas is transferred from the internal energy → temperature decreases
- All the work done on the gas is transferred to the internal energy
- Adiabatic lines of P-V diagrams are steeper than isothermal lines
- The change must be as rapid as possible in a well-insulated container
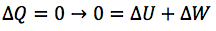
How is the work done calculated in a thermodynamic cycle?
By calculating the area enclosed by the cycle
What is the second law of thermodynamics?
Thermal energy cannot spontaneously transfer from a region of lower temperature to a region of higher temperature
What is entropy?
A numerical expression of the degree of disorder in a system
State the second law of thermodynamics in terms of entropy changes
In every process, the total entropy of any isolated system, or the universe as a whole, always increases
What happens to the total entropy of a system and surroundings if local entropy decreases?
It increases.
What do we know about overall energy and overall entropy?
Overall energy is always conserved and overall entropy always increases


