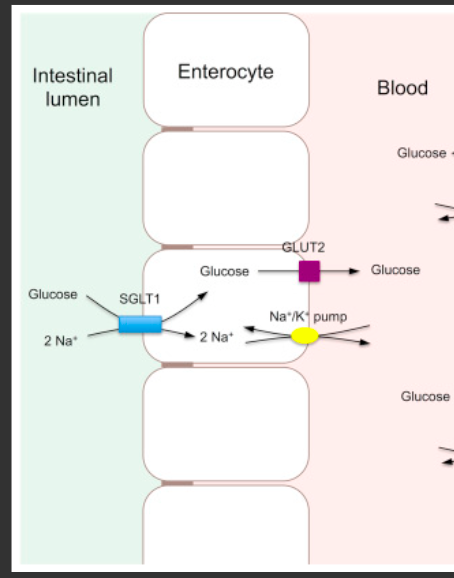
explain how glucose is absorbed from intestinal lumen into portal vein
aim: transport glucose from intestinal lumen -> portal vein
1. primary active transport: The sodium-potassium pump maintains the electrochemical gradient of living cells by moving sodium out and potassium in of the cell
2. secondary active transport: SGLT1 symporter brings glucose into the enterocyte / intetinal epithelium cell. this is driven by Na+ and glucose transported (through 2ry AT) from the intestinal lumen into the intestinal epithelium.. (Na+ goes in and glucose goes along with it).
- leads to high levels of glucose in intestinal epithelium. faciliated diffusion of glucose via Glut2 facilated diffusion transporter causes gluocse to move from intestinal epithelium into portal vein. (Glut 2 is only expressed on basal lateral membrane)
what is / how does DNA mismatch repair occur?
this type of repair, repairs insertion or delation mutations that occurs during DNA replication and recombination:
- DNA strand has an error
- A nick (break) occurs in strand of DNA that has the error in.
- This provides signals that direct mismatch proof reading proteins to DNA strand that has nick.
- proteins scan the strand until the nick is found
- proteins (MutS and MutL) remove the DNA strand from the error to the nick
- DNA repair synthesis occurs to fill in corrected DNA into strand
how does compaction of DNA occur?
1. compaction of histomes: (basic proteins that form subunit structures)
- 8 histomes form a nucleosome bead
- DNA wraps around histomes: 146 base pairs of DNA wraps around a histome
= v compact !
- further compaction of histomes into chromatin
- further folding of chromatin into chromosomes
what is PCR used for?
how does PCR work?
PCR: amplifies a region of interest within the human genome, using a template DNA and specific primers
- denaturation: heats DNA to over 90 degrees - opens DNA up
- annealing: 54 degrees. primer is added. DNA binds to primers
- extension: 72 degrees. add polymerase enzymes. builds DNA
repeat x lots
what is sanger sequencing?
(golden standard for genetic testing)
(Sanger sequencing is the process of selective incorporation of chain-terminating dideoxynucleotides by DNA polymerase during in vitro DNA replication)
uses dideoxyribonucleic acids (ddNTP) to terminate polymerization reaction of DNA.
can find every single base pair in a sequence!
Q what is next generation sequencing? what are the different types?
- whole genome sequencing
- whole exome sequencing - sequencing all exons in genome (~2% of genome). no info on mutations outside of exons.
RNA sequencing (RNAseq) - sequencing all expressed RNA and expression analysis. (RNA is direct result of exons to RNA, but also RNAseq gives info about which genes are expressed and if highly / lowly expressed).
why is molecular diagnosis and targeted therapy good?
what can it be used for? (x4)
good: offers personalised approach to care. can tailor to specific genetic conditions of patient
- selection of embryos prior to implantation in IVF (carry out genetic testing on embryos and screen for mutations)
- personalised chemotherapy (take a biopsy of cancer and sequence DNA. if have specific changes to target molecules, can target those molecules with chemo. e.g. herceptin)
- gene therapy for genetic conditions (v experimental)
- pharmacogenomics
describe 3 prenatal diagnoses that can be undertaken to test for aneuploidy
- amniocentesis: genetic testing of amniotic fluid. using needle to extract transabdominal. 15-18 weeks of preg (risk to miscarriage: 1/100). ultrasound guidance used. t
2. chorionic villus sampling: genetic testing of tissue from placenta (choroinic villi), ultrasound guidance used transabdominal or transcervical. 12-14 weeks
3. non invasise technqiues: ultrasound imaging of back of neck of embryo at 11-14 weeks. if depth of fluid at back of neck is 3.5-4.4 mm = 70% chance of delivering baby with no major abnormalities.
what is Xist?
- X-inactivation is assisted / coded by non coding RNA: XIST (X inactivion centre)
- Xist assocates closely with the X-chr from which it is expressed -> leads to chromatin changes and spatial reorganisation of chr. chr will condense into Bar > results in transcriptional inactivation in that chr.
(X chromosome inactivation (XCI) in females thus leads to similar transcription levels of X-chromosomal genes between males and females, who now both express genes from a single X chromosome. )
what is the Barker hypothesis?
thrifty phenotype: if a baby in-utero is subject to poor nutrients (because of bad maternal diet), the epigenome is programmed to expect this environment post-natally. means there are developmental changes in cellular energy metabolism, such as: glucose handling, lipid metabolism and mt biogenesis. if exposed to nutrient poor environment postnally: means pre-disposed to have a survival offspring.
BUT
if born and given a nutrient rich environment -> get increased risk of susceptibility to metabolic disease, such as type 2 diabetes, obesity and CHD
how does DNA methylation of cytosine occur?
- methyl groups can be added / removed from cytosine (the nucleotide base)
- methylation interferes with binding of transcriptional activators -> causes gene inactivation
- occurs when you get areas of DNA where cytosine and guanine are adjacent and repeated (CGCG / CpG islands). found in promoter regions.
- DNA methyltransferase (DNMT) is the enzyme
- methyl is added to cytosine to make 5-methylcytosine.
describe how chromatin architecture is changed (a factor than influences epigenome mechanism)
chromatin architecture modifcations
- changes shape of histone / DNA complex
- ATP-depending remodelling:
- complex of proteins sits on the histone nucleosomes. uses ATP. alters:
1. the contact between DNA and histones,
2. the path of DNA wound around histones,
3. the structure of nucleosomes.
normally an immediate effect
explain how histone modifications occurs (a factor than influences epigenome)
where do most modifications occur?
which enzymes used?
histone modifications
- post-translational modification of histone proteins: acetylation (acetyl functional group added), methylation M (addition of methyl group) , phosphorylation P, ubiquitylation (U)
- most of modifications are on lysine (K) and serine a.a. -> can make active genes by doing this. e.g. H3K4me3 (this is 3 methyl groups on K) OR make genes repressed (e.g. H3K27me3- adding 3 methyl groups to K)
can change histone modifcations using enzymes:
a) histone acetylase (HAT): opens DNA to make it accessible
b) Histone deactylase (HDAC) associated with closing DNA to make it inaccessible
explain non-coding RNAs
(a factor than influences epigenome / regulates epigenome):
- non-coding RNA (ncRNA). functional, but not translated into proteins = called interfering RNA.
- this ncRNA interferes with gene expression at transcriptional and post-transcriptional level. combine to RNA and prevent it from being translated. (involved in splicing, editing and mRNA stability)
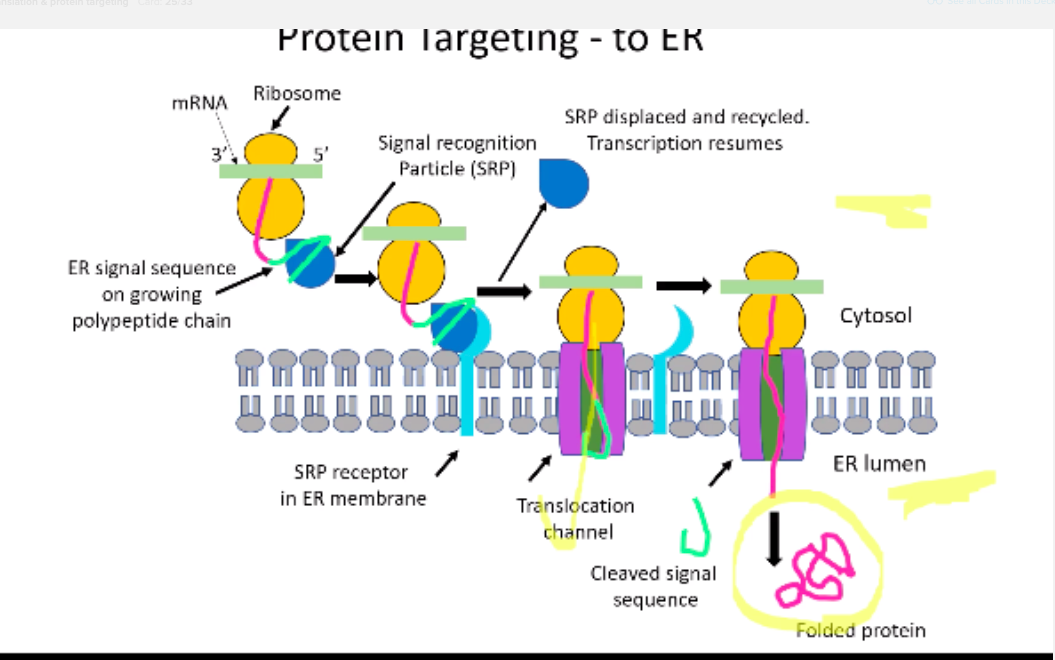
how does protein targeting to ER occur?
- polypeptide chain will have an endoplasmic reticulum (ER) signal sequence on it
- the ER signal sequence interacts with signal recognition particle (SRP)
- the SRP binds to receptor (on a translocation channel) within the membrane of ER
- the SRP leaves complex
- the newly synthesised protein can translocate through the translocation channel.
- SRP is cleaved
- leaves the new folded protein in correct place
describe how initiation of transcription works
- DNA unwinds close to a gene, RNA polymerase binds to a promoter sequence: how?:
- the promoter sequence acts as a template for the assembly of the multi component complex of proteins, called the pre-initiation complex, which brings pol II to gene
- once bound, the RNA polymerase II can then start trancribing the gene. (get transcription intition, elongation and termination)
- transcription always starts at ATG on Exon 1
what are the two apoptopic pathways?
2 pathways:
extrinsic apoptopic pathway
- enviroment around the cell could cause cell death.
- tumour necrosis factor (TNF) binds to death receptor. activates caspases
intrinsic apoptopic pathway:
- targets the cell’s mitochondria -> activates caspases
what are caspases? whats mechanism involved?
Initiator caspases
- pro-caspases get activated to active caspases (Caspase 2, 8 and 9)
- intiator caspases activate the executioner caspases
Executioner caspases
- again, activated from pro-caspase to activate caspase (Caspase 3, 6 and 9 -> effector caspases)
Cause:
nuclear fragmentation, cytoskeleton disruption, membrane alterations, organelle reduction
what is role of ACh on:
a) skeletal muscle - how?
b) cardiac muscle - how?
c) acinar cells - how?
- *skeletal muscle:**
- acetylcholine binds to skeletal muscle cell receptors called nicotinic acetylcholine receptors (nAChR)
- depolarisation occurs (action potential more likely to happen)
- contraction of skeletal muscle
- *cardiac muscle:**
- acetylcholine binds to acetylcholine receptor on heart muscle (called muscarinic receptor M2)
- muscarinic receptors are G protein coupled receptors that activate ionic channels via a second messenger cascade
- this causes hyperpolarisation and a decrease in cardiac activity
- *acinar cells:**
- acetylcholine binds to mAChr
- activates the secretion of digestive molecules
whats the mechanism of calcium ions being used as an intracellular messenger?
- calcium ions are stored in the ER
- calcium enters cytoplasm via transmembrane calcium channels or via channels in the ER
- calcium ions can bind to proteins and trigger events within the cell
- after events occurred, calcium signals are deactivated by pumping ions back into ER or out of cell
how are intracellular receptor proteins activated?
- hydrophobic signal molecules diffuse directly across plasma membrane and bind to intracellular receptor proteins
- e.g signal molecules include: steroid hormones, thyroid hormones, retinoids
- then, intracellular receptors bind to specific DNA sequences adjacent to the genes the ligand regulates
inactive receptor
- each receptor will bind to short DNA-binding domain
- a receptor protein in its inactive state is bound to its inhibitory proteins
active receptor
- ligand binds to receptor and causes the (ligand-binding domain of the receptor to clamp shut around the ligand) inhibitory protein to dissociate
- receptor is active
what happens with cell signalling when have a stroke?
- dying cells release large amounts of neurotransmitter: glutamate
- glutamate is toxic at high concs
- excitoxicity -> glumatate spreads outside area of initial damage = braindamage
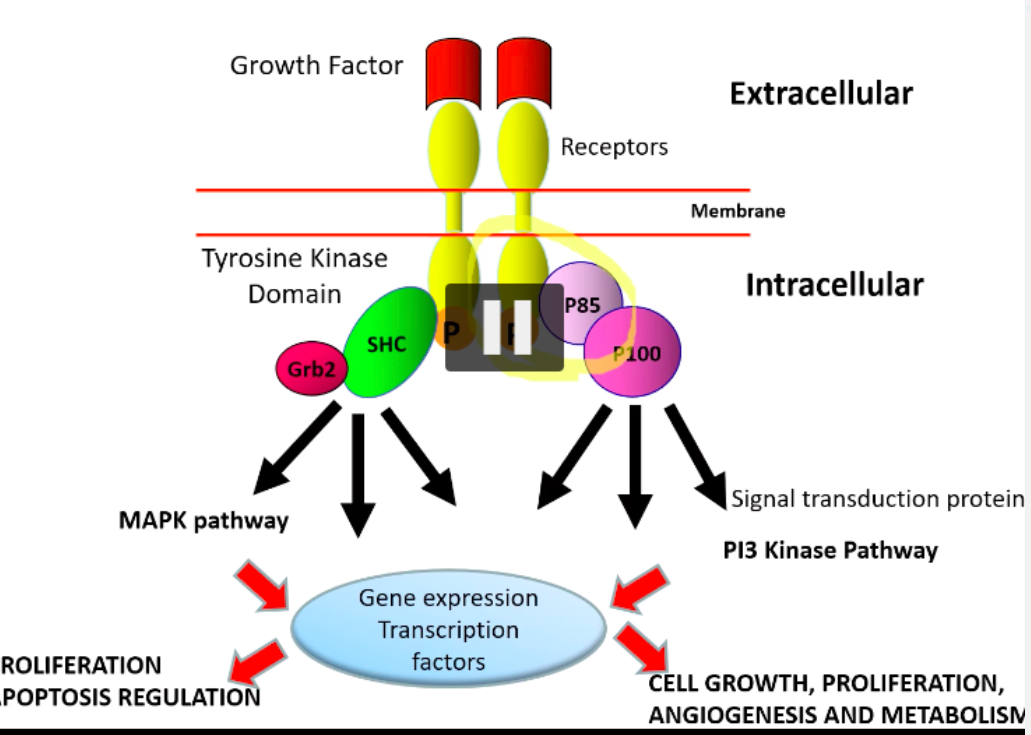
give over view of cell signalling in normal cell - growth factor -> activate gene expression?
- growth factor binds to receptor. two receptors interact (dimerization)
- intracelllar side: phosphorylation of tyrosine
- siganlling proteins bind to P-tyrosine
- causes cascade of phosphorylation events
- activates two pathways: MAPK pathway and PI3 Kinase Pathway
- once pathways are activated, activate gene expression and transcription factors occur.
explain the ras pathway and how mutation leads to kras cancer
- when ras switched on: (normally ras is switched on by binding to GTP) can switch on ERK and AKT pathways. (ras regulates the pathways by turning on / off the ERK and AKT pathways)
- mutated ras (kras): hydrolysis of bound GTP (first stage in pathway) occurs v slowly. GTP is bound to ras in unhydroloysed form - ras is permenantly switched on. continois proliferation and growth.



